Background and Objective: Diabetes mellitus (DM) is a rapidly growing global health challenge characterized by chronic hyperglycaemia and progressive metabolic dysfunction. Despite established clinical guidelines, conventional diabetes management remains limited by delayed diagnosis, fragmented data systems, poor glycaemic control, and high complication burden. Artificial intelligence (AI) has emerged as a promising solution to enhance early detection, risk prediction, monitoring, and personalized treatment. This review aims to comprehensively examine AI-based approaches in diabetes mellitus and evaluate their clinical applications, benefits, and future potential. Methods: A comprehensive narrative review was conducted using published research articles, systematic reviews, and meta-analyses focusing on AI applications in diabetes care. Literature addressing AI in early screening, diagnosis, glycaemic monitoring, complication prediction, personalized therapy, and digital health integration was analysed and synthesized. Results and Interpretation: AI-based models demonstrate improved accuracy in early diabetes risk prediction by integrating electronic health records, laboratory data, imaging, and wearable sensor inputs. Deep learning systems have achieved high diagnostic performance in diabetic retinopathy screening, while AI-driven continuous glucose monitoring and closed-loop insulin delivery systems significantly improve time in target glucose range. Predictive analytics further support early identification of nephropathy, neuropathy, and cardiovascular risk. Additionally, AI enhances personalized medicine by optimizing drug selection, insulin dosing, and lifestyle recommendations. Conclusion: AI-based approaches hold substantial potential to transform diabetes care by enabling precision medicine, early intervention, and scalable digital health solutions. However, robust validation, regulatory oversight, bias mitigation, and data privacy safeguards are essential for safe and equitable clinical implementation.
Artificial Intelligence; Diabetes Mellitus; Machine Learning; Deep Learning; Glycaemic Monitoring; Personalized Medicine.
Global Burden of Diabetes
Diabetes mellitus is acknowledged as a serious global health issue with a fast rising prevalence that places a heavy burden on the healthcare system, mortality, and morbidity globally (1,2). According to systematic assessments of epidemiological data from around the world, the number of persons with diabetes has significantly increased in recent decades. For instance, a thorough worldwide burden assessment found that the prevalence of diabetes increased steadily between 1990 and 2021, and if current trends continue, predictions suggest that this increase will continue through the middle of the century (1).
According to estimates from a global analysis conducted in 2017, there were 476 million people with diabetes worldwide, accounting for over 67 million disability-adjusted life-years (DALYs). Without effective intervention strategies, the number of cases is expected to increase to over 570 million by 2025 (2). Elevated body mass index, poor diets, and physical inactivity are metabolic and behavioural risk factors that are closely linked to diabetes and greatly increase the disease's incidence and related mortality (2,3).
Geographically, there are significant regional and socioeconomic group differences in both incidence and prevalence, which reflects the impact of urbanization, changing lifestyles, and demographic shifts as well as expanding health disparities (2). Type 2 diabetes is responsible for the great majority of cases and continues to be the leading cause of future burden globally, according to other systematic reviews (4). These epidemiological trends highlight the fact that diabetes is a widespread public health issue that affects people from a wide range of geographic locations and socioeconomic backgrounds in addition to being a clinically difficult condition (1,4).
Limitations of Conventional Diabetes Management
Even with decades of research and comprehensive professional recommendations, standard diabetes care strategies have significant obstacles to reaching the best possible results. Standardized treatment procedures, static risk models, and recurring clinical evaluations are the mainstays of conventional management techniques. However, the varied and dynamic nature of diabetes progression and control cannot be adequately captured by these techniques (5).
Conventional models are severely limited by their reliance on infrequent monitoring and simple risk variables, which do not take into consideration the intricate interactions between biological, behavioural, and environmental factors that affect individual glycaemic trajectories. Epidemiological risk models, for instance, tend to ignore more subtle but important indicators like physical activity patterns, psychosocial factors, continuous glucose dynamics, and concomitant disorders in favour of demographic measures like age or body mass index (5).
In addition, fragmented healthcare data systems, delayed feedback loops, and inadequate integration of patient longitudinal data frequently hinder rapid intervention changes and adaptive patient care in real-world clinical management. Long wait times between clinic appointments are a common problem for patients, which delays the identification of problems or deteriorations. Care discrepancies are further exacerbated by the fact that access to specialized diabetes care is still unequal, especially in low- and middle-income nations with limited resources (6).
Furthermore, despite their potential effectiveness, traditional patient self-management programs frequently struggle to sustain long-term adherence because they lack real-time feedback systems and offer no personalized customization. Because of this, a large number of people go untreated or receive inadequate care, which raises the risk of serious consequences such retinopathy, neuropathy, and cardiovascular disease (5,6).
Why AI Is Needed in Diabetes Care
In light of these drawbacks, artificial intelligence (AI) presents a revolutionary solution to meet unmet demands in clinical decision support, risk prediction, diabetes diagnosis, and patient monitoring. AI includes deep learning (DL), machine learning (ML), and other computer techniques that may uncover significant patterns from sizable and intricate health-related datasets (7,8).
The capacity of AI techniques to combine heterogeneous data sources, including wearable sensor streams, continuous glucose monitoring (CGM) outputs, imaging data, and electronic health records (EHRs), to produce predictive models with greater sensitivity and specificity than conventional models is one of their main advantages (7,8). For example, by capturing subtle nonlinear interactions among indicators that traditional regression models frequently miss, AI-based risk prediction models have shown increased performance in identifying patients at elevated risk of developing diabetes or complications (8).
By modifying suggestions in response to real-time physiological data, lifestyle patterns, and individual risk profiles, AI can also facilitate individualized treatment techniques. AI-powered digital health treatments have the potential to improve long-term self-management outcomes, medication adherence, patient engagement, and behaviour modification (7).
Predictive analytics and pattern recognition automation can help doctors identify illness exacerbations earlier and customize treatment plans, which will ultimately improve patient outcomes. Furthermore, automated screening for diabetes-related problems (such diabetic retinopathy) is made possible by AI techniques like deep learning applied to medical imaging. This allows for scalable and effective diagnostic procedures, which are essential in underprivileged settings (8).
AI is a potent tool to overcome the static and fragmented characteristics of traditional diabetes management because of its ability to process large amounts of healthcare data and adjust over time. All things considered, the application of AI to diabetes care has the potential to address many of the fundamental issues with traditional management models by enhancing continuous monitoring, improving early detection, personalizing treatment plans, and supporting clinical decision-making at scale (7,8).
Definition of Diabetes Mellitus
A collection of metabolic disorders known as diabetes mellitus (DM) are characterized by persistent hyperglycaemia brought on by deficiencies in either insulin action or secretion, or both (9). If left untreated, persistent hyperglycaemia damages many organs over time and results in aberrant metabolism of proteins, fats, and carbohydrates (9,10).
Classification of Diabetes Mellitus
Diabetes is categorized according to its underlying aetiology rather than just its symptoms (10).
Among the most well-known kinds are:
- Type 1 Diabetes Mellitus (T1DM)
The autoimmune death of pancreatic β-cells causes type 1 diabetes, which results in complete insulin insufficiency. It accounts for 5–10% of all instances of diabetes.
Important facts regarding T1DM:
- It happens when β-cells are destroyed by an immune-mediated process.
- Patients need insulin therapy for the rest of their lives since they have little or no endogenous insulin.
- Although it can happen at any age, it typically manifests in childhood or adolescence (11).
- Type 2 Diabetes Mellitus (T2DM)
About 90% of instances of diabetes are type 2, which is the most prevalent kind (11). It is characterized by a relative insulin secretory deficiency and peripheral tissue insulin resistance (11,12).
Important facts regarding T2DM:
- Fat, muscle, and the liver are less sensitive to insulin.
- As β-cell function gradually deteriorates, insulin production falls short of demand.
- Genetic predisposition, obesity, and a sedentary lifestyle all raise risk.
- Lifestyle modifications and glucose-lowering medications are part of management; insulin is used when necessary (12).
- Gestational Diabetes Mellitus (GDM)
Glucose intolerance initially identified during pregnancy is known as gestational diabetes mellitus. Due to metabolic changes brought on by pregnancy, it typically manifests in the second or third trimester (13).
Important details of GDM:
- When hyperglycaemia is initially noticed during pregnancy, it is recognized.
- GDM raises maternal and lethal consequences (such as preeclampsia and macrosomia) if left unchecked.
- There is a greater lifetime risk of T2DM for women with GDM and their offspring (13,14).
- Additional Specific Types
Other uncommon types of diabetes consist of:
- Monogenic diabetes (e.g., MODY) from single gene mutations.
- Secondary diabetes caused by other diseases (e.g., pancreatitis) or medications (e.g., glucocorticoids) (10).
Pathophysiology
- Insulin Resistance (IR)
A decreased biological response to insulin in target tissues, primarily skeletal muscle, liver, and adipose tissue, is known as insulin resistance. Skeletal muscle has decreased glucose absorption in insulin resistance due to poor insulin-stimulated GLUT-4 translocation. Insulin resistance makes it difficult for insulin to inhibit gluconeogenesis in the liver, which raises fasting plasma glucose levels. (9, 12, 14)
Increased lipolysis and the release of free fatty acids (FFAs) into the bloodstream are caused by insulin resistance in adipose tissue. Increased FFAs affect insulin signalling by encouraging ectopic fat accumulation in the liver and skeletal muscle. Insulin resistance is greatly exacerbated by obesity, particularly visceral obesity, which raises inflammatory cytokines and causes an imbalance in adipokines. (9, 10, 14)
Because β-cells secrete more insulin to keep glucose levels normal, insulin resistance first causes compensatory hyperinsulinemia. Postprandial glucose rises first, followed by fasting hyperglycaemia, as the compensatory state becomes insufficient with advancement. Increased cardiovascular risk and dyslipidaemia (↑ triglycerides, ↓ HDL) are closely linked to insulin resistance. (10, 12, 14)
One important metabolic problem that frequently manifests years before type 2 diabetes mellitus (T2DM) is diagnosed is insulin resistance. (12,14)
- β-Cell Dysfunction
Impaired insulin production in relation to the level of insulin resistance is known as β-cell dysfunction, and it is crucial for the development of overt type 2 diabetes. β-cells first increase insulin secretion and occasionally β-cell bulk to make up for insulin resistance. Chronic metabolic stress causes β-cells to become less responsive to glucose, which lowers first-phase insulin production. (10,11,14)
Glucotoxicity, which is brought on by chronic hyperglycaemia, harms β-cells by causing oxidative stress, inflammation, and a decrease in gene expression. Increased β-cell death and decreased insulin secretion are the results of lipotoxicity, which is caused by elevated free fatty acids. Glucolipotoxicity is the term for the combined effect of elevated FFAs and chronic hyperglycaemia, which hastens β-cell failure. Pancreatic islet inflammation increases cytokine-mediated stress pathways, which in turn leads to β-cell malfunction. (10, 11)
Progressive β-cell loss in type 2 diabetes is associated with islet amyloid deposition (IAPP accumulation), which alters islet architecture. (13) Given that numerous T2DM risk genes impact β-cell activity and insulin secretion capacity, genetic susceptibility is significant. (11, 14) Gradually worsening β-cell dysfunction makes it difficult to meet insulin demands, which leads to chronic hyperglycaemia. (10,11,14)
- Relationship Between Insulin Resistance and β-Cell Dysfunction
Insulin resistance raises the need for β-cells to secrete insulin. Although β-cells initially adjust, prolonged metabolic stress results in β-cell fatigue and decreased insulin production. (10,11,14)
Because of the hyperbolic relationship between insulin sensitivity and secretion, a decrease in sensitivity must be offset by an increase in secretion in order to maintain euglycemia. This balance is described by the disposition index, which also forecasts the likelihood of developing type 2 diabetes. (14)
Insulin resistance and insufficient β-cell compensation lead to type 2 diabetes. (10,11,14)
Current Challenges in Diabetes Care
- Late Diagnosis
Although many people with diabetes go years without receiving a diagnosis, early detection is essential to preventing long-term problems. Because prolonged hyperglycaemia before diagnosis raises the risk of microvascular and macrovascular damage, undiagnosed diabetes is a significant hidden risk factor. (15)
Because glycated haemoglobin (HbA1c) may not accurately reflect glycaemic state in some populations, relying just on it for diagnosis can further postpone the identification of diabetes for years. (16)
These delays contribute to an earlier onset of problems at the time of clinical discovery by allowing persistent hyperglycaemia to remain. (15,16)
- Poor Glycaemic Control
Prevalence and Impact
Reducing the risk of microvascular and macrovascular diabetes consequences requires maintaining ideal blood glucose levels, such as a HbA1c of less than 7%. Poor glycaemic control is still common in type 2 diabetic individuals, though. (17, 18)
According to extensive systematic reviews, the prevalence of poor glycaemic control is significant worldwide, particularly in low- and middle-income nations, and can range from 45 to 90 percent, depending on the population and the method of assessment. (17)
Determinants of Poor Control
Poor glycaemic control is caused by a number of patient-level and system-level factors:
- Low health literacy and non-adherence to medication.
- Difficulties with lifestyle and poor diet compliance.
- Comorbid conditions and alcohol consumption.
- Inconsistent supervision and restricted access to healthcare. (17, 18)
In situations when healthcare resources and information are scarce, the intricacy of treatment regimens, particularly insulin therapy, can lower adherence. (17)
Consequences of Poor Control
Inadequate glycaemic control is strongly linked with progression of beta-cell dysfunction, increased glycaemic burden, and heightened risk of both microvascular and macrovascular complications. (17,19)
Long-standing hyperglycaemia can also impair β-cell function further, making subsequent control even more difficult a phenomenon known as glycaemic burden. (19)
- Complications Burden
Numerous long-term consequences linked to diabetes significantly increase morbidity, mortality, and healthcare expenses.
Traditional and Emerging Complications
Both microvascular (retinopathy, nephropathy, neuropathy) and macrovascular (coronary artery disease, stroke, peripheral vascular disease) problems are brought on by persistent hyperglycaemia. (20)
The burden of diabetic morbidity is expanding beyond traditional endpoints as people with diabetes live longer thanks to new consequences such functional disability, infections, liver disease, and cancer connections. (20)
Severity and Health Outcomes
Increased hospitalizations and mortality are closely correlated with the quantity and severity of diabetes complications. (21)
For instance, research employing complication severity indexes demonstrates that, in comparison to isolated difficulties, the risk of death and the burden on the health system are exponentially increased when there are several concurrent complications. (21)
Definition of Artificial Intelligence (AI) in Health care
The ability of digital devices or computers to carry out tasks like reasoning, learning, and decision-making that have historically required human intelligence is known as artificial intelligence (AI). This feature sets AI systems apart from traditional software, which merely complies with explicit instructions without "thinking" or adapting. AI systems can perform well on challenging tasks utilized in clinical practice and research because they use computational techniques to mimic parts of human cognitive functioning, such as perception and problem solving (22).
The use of these technologies to analyse and comprehend complicated medical and health-related data is especially referred to as artificial intelligence in the healthcare industry. AI has the potential to increase efficiency, accuracy, and patient outcomes in the healthcare industry by enhancing human capacities in diagnosis, treatment planning, patient monitoring, and healthcare delivery (23).
Types of AI Used in Healthcare
Artificial Intelligence is a vast science with several subfields, each with its own techniques and uses. Neural networks (NNs), deep learning (DL), and machine learning (ML) are the three main forms of AI utilized in healthcare. Much of the recent advancements in clinical decision support, predictive analytics, and medical imaging can be attributed to these methods.
- Machine Learning (ML)
A subfield of artificial intelligence called machine learning allows computers to recognize patterns and make choices or predictions without needing to be specifically programmed for each activity. ML algorithms can generalize from previous cases to new, unseen data since they perform better the more data and examples they are exposed to (24).
ML algorithms are frequently used in the healthcare industry to understand massive datasets, including medical imaging, laboratory results, and electronic health records (EHRs). Early disease identification, risk assessment, predictive modelling, and treatment outcome optimization are all supported by machine learning. ML models, for example, can analyse EHRs to find trends that foretell the development of a disease or the decline of a patient before clinical signs appear (25).
ML is separated into various learning paradigms:
- Supervised learning teaches the algorithm how to map inputs to desired outputs (e.g., illness vs. non-disease classification) using labelled training data.
- Unsupervised learning finds patterns or groups in unlabelled datasets without clear results.
- Reinforcement learning uses feedback loops and trials to learn the best course of action. Decision support systems and healthcare analytics make substantial use of these learning paradigms (26).
- Deep Learning (DL)
Multi-layered neural network topologies are used in deep learning, a specialized subset of machine learning, to model intricate correlations in data. Deep learning models automatically find hierarchical representations of raw data through layers of computation, in contrast to classical ML techniques that frequently call for manual feature engineering (27).
Deep learning has revolutionized medical image analysis in particular, making it possible to do extremely precise tasks like tumour detection, retinal disease classification, and anatomical structure segmentation on MRI or CT scans. For instance, deep learning models such as convolutional neural networks (CNNs) have shown human-level performance in tasks such as detecting anomalies in radiography pictures (28).
Deep learning differs from traditional machine learning (ML) in that it can automatically extract characteristics from unstructured data, such as text, waveforms, and pictures. This makes it useful for sophisticated diagnostic and prognostic applications in clinical procedures (29).
- Artificial Neural Networks (ANNs)
Computational models inspired by the composition and operation of the human brain are called artificial neural networks. Interconnected nodes, or "neurons," arranged in layers—an input layer, one or more hidden layers, and an output layer—make up artificial neural networks (ANNs). To enhance performance, each connection has a weight that is modified during training (30).
The basic building blocks of deep learning are neural networks. While more complex networks with several hidden layers constitute deep neural networks (DNNs) capable of collecting nuanced patterns in high-dimensional healthcare data, simple ANNs may perform tasks like classification or regression (23).
In medical applications, neural networks are widely used for:
- Medical image classification and segmentation
- Time-series analysis of physiological signals
- Predictive modelling of clinical outcomes
ANNs are an effective tool for automated decision assistance in healthcare contexts because of their design, which enables them to learn directly from examples (26).
Relationships Between AI, Machine Learning, Deep Learning, and Neural Networks
Healthcare is changing as a result of artificial intelligence through machine learning, deep learning, and neural networks, which allow:
- Automated diagnosis and identification of diseases
- Enhanced predictive analytics accuracy
- Effective handling of intricate imaging and clinical data
- Tailored treatment suggestions and monitoring systems
These AI methods can improve diagnosis accuracy, help physicians make decisions, and possibly lower healthcare costs by analysing vast amounts of medical data. Though AI has potential, safe and successful deployment depends on ethical factors such data privacy, bias in model outputs, and clinical validation (23).
Applications of AI in Diabetes Mellitus
- AI in Early Screening & Risk Prediction
Artificial intelligence (AI) helps in early screening and risk prediction of diabetes by analysing large-scale clinical data such as age, BMI, family history, blood pressure, lifestyle factors, and laboratory values to identify high-risk individuals before symptoms appear, enabling timely preventive interventions and reducing long-term complications (31).
- Prediction of Prediabetes and Type 2 DM
By identifying small trends in physiological, laboratory, and demographic factors, artificial intelligence (AI) use machine learning (ML) to analyse massive clinical datasets and identify people at risk of developing type 2 diabetes or prediabetes. By incorporating several characteristics, these models enhance risk classification beyond conventional statistical techniques, allowing for earlier intervention to stop the progression of disease (32).
- Risk Stratification Models
Based on thorough predictive analytics, risk stratification AI models classify people as having a low, medium, or high risk of developing diabetes in the future. These models enable individualized monitoring and customized preventive interventions by prioritizing preventive care pathways and quantifying individual risk through the integration of clinical, behavioural, and sensor data (32).
- AI in Diagnosis
Retinal Image Analysis for Diabetic Retinopathy
By learning visual characteristics linked to the severity of the illness, artificial intelligence (AI) systems, particularly deep learning-based convolutional neural networks, have demonstrated great accuracy in identifying diabetic retinopathy (DR) from retinal images. For referable DR screening, extensive research shows that automated AI methods perform diagnostically on par with human specialists (33, 34).
By facilitating the early diagnosis of visual alterations that pose a hazard to vision and streamlining referral processes, clinical applications of AI-based DR screening can assist environments with limited resources (33).
Example Tools from Google Health
The idea that AI can accurately classify retinal pathology is supported by a number of open studies that validate deep learning-based DR models with strong sensitivity and specificity in real-world settings, despite the fact that certain proprietary tools, like Google Health's DR detection model, are not publicly available as downloadable PDFs (33).
Automated Classification of DR Severity
By recognizing characteristics like microaneurysms, haemorrhages, and neovascularization in fundus images, AI systems automate DR severity grading schemes. By standardizing DR staging across healthcare settings and reducing human workload, these automated classifiers can enhance early disease treatment (33).
- AI in Glycaemic Monitoring
AI-Based CGM Data Interpretation
Dense time-series glucose readings are generated from continuous glucose monitoring (CGM) data. Advanced prediction of hypo- and hyperglycaemia episodes before they happen is made possible by AI systems' ability to anticipate glucose trends and detect glycaemic excursions. AI facilitates more sophisticated glycaemic control than static measures by identifying patterns in CGM profiles (35).
The use of CGM data by AI-based DSS to optimize insulin dosage techniques, modify glucose prediction models, and tailor insulin recommendations makes diabetes self-management more sensitive to real-time physiology, according to narrative reviews (36).
Smart Insulin Dosing Systems
AI-powered closed-loop insulin delivery devices automatically modify insulin supply in real time by combining CGM measurements with insulin pump control algorithms. According to meta-analyses, AI-driven closed-loop systems demonstrate efficacy in automated insulin dosage and glycaemic control while improving time in the glucose target range when compared to conventional therapy while retaining safety (37).
- AI in Complication Prediction
Diabetic Nephropathy
AI prediction models can identify people at risk of diabetic nephropathy by analysing clinical and laboratory data (such as blood pressure and albuminuria). AI assists clinicians in taking early action to stop the decrease of kidney function by measuring risk trajectories using longitudinal datasets (38).
Neuropathy
From clinical and sensor data, machine learning models have been constructed to identify patterns suggestive of diabetic peripheral neuropathy. By utilizing multi-feature inputs associated with nerve conduction and symptom profiles, these models facilitate earlier diagnosis than traditional evaluations (38).
Retinopathy
In addition to screening for the existence of DR, deep learning-based retinal analysis uses longitudinal imaging patterns to estimate progression risk. This helps anticipate future visual outcomes and prioritize high-risk individuals for treatment or referral (33).
Cardiovascular Risk Prediction
AI models stratify cardiovascular risk in diabetic patients by integrating multimodal data, including lab findings, wearable activity data, and EHR characteristics. Based on the intricate relationships between metabolic indicators and lifestyle factors, these algorithms are able to identify those who are more susceptible to heart disease (32, 34).
- AI in Personalized Treatment
Predicting Response to Metformin
AI algorithms can predict a person's likely reaction to first-line treatments like metformin by analysing patterns in baseline patient information (e.g., age, HbA1c, body mass index), allowing for customized drug regimens that optimize effectiveness and reduce adverse effects (32).
Optimizing Insulin Therapy
In order to improve glycaemic stability and minimize the need for manual changes, machine learning models that analyse CGM profiles and insulin delivery data can suggest optimal insulin dosages based on anticipated glucose patterns (36).
Individualized Lifestyle Recommendations
AI systems use information from activity trackers, food records, and wearables to customize lifestyle recommendations that promote better glycaemic control. Patients can adjust their behaviour according to their glycaemic response patterns by using personalized feedback systems (32, 35).
- AI in Drug Discovery & Development
Virtual Screening of Antidiabetic Drugs
Large chemical libraries are being screened using AI approaches, particularly deep learning and generative models, to find new antidiabetic drugs. By selecting promising compounds for additional testing, these techniques expedite the initial phases of drug discovery (32).
Dose Optimization
By simulating responses at various dosages, reinforcement learning and optimization algorithms assist in identifying optimal dosing schedules for novel medications, potentially saving time and money throughout clinical development stages (32).
Pharmacovigilance & Post-Marketing Surveillance
Following market approval, AI systems can improve drug safety monitoring by mining real-world safety data (such as EHRs and registries) to identify adverse drug responses faster than with conventional methods (32).
- AI-Based Devices and Digital Tools in Diabetes
Smart Glucometers
In order to automatically upload glucose levels and feed data into AI models that can identify patterns and notify patients of out-of-range glycaemic episodes, smart glucometers connect with mobile apps (35). This helps with self-management and clinician evaluation.
Mobile Health Apps
In order to improve adherence and self-care behaviour patterns, mobile applications combine AI analytics with self-reported food consumption, activity, and CGM/glycemia data to offer predicted insights and individualized assistance (32).
Wearable Biosensors
Wearable biosensors, such as chest patches and smartwatches, continuously gather physiological information that AI models utilize to forecast risk events and glucose dynamics. According to reviews, wearables with AI capabilities can facilitate tailored therapies and increase monitoring accuracy (35).
Closed-Loop Insulin Delivery Systems
AI-powered closed-loop systems, commonly referred to as "artificial pancreas" technologies, automatically combine insulin pump control and CGM data to maintain ideal blood glucose levels. According to meta-analyses, these systems have a clinically significant advantage over standard care methods in that they increase duration in target range and lower the incidence of hypoglycaemia (37).
Advantages of AI in Diabetes Care
1. Early Detection
Large and complicated datasets (such as EHRs, retinal scans, and CGM data) can be analysed by AI-enabled technologies to find early indicators of diabetes, prediabetes, and associated problems before significant clinical symptoms manifest. By identifying trends that conventional approaches would overlook, these prediction algorithms let physicians take action sooner and prevent the development of severe disease states (39, 40).
AI models, for instance, can recognize small patterns in retinal imaging and other diagnostic inputs that suggest early disease development and possible risk of progression, allowing for prompt preventive intervention and lowering the possibility of irreversible sequelae (40).
2. Improved Accuracy
High levels of diagnostic accuracy and dependability in the treatment of diabetes have been shown by artificial intelligence models, especially those that employ machine learning and deep learning. Through sophisticated analytics of multidimensional datasets, AI systems routinely outperform conventional risk stratification and diagnostic methods, improving categorization and decision-making accuracy (39).
Compared to traditional methods, AI-powered diagnostic tools have demonstrated better performance in detecting diabetes subtypes, screening for sequelae (such as neuropathy and retinopathy), and forecasting risk profiles with higher sensitivity and specificity (39, 40).
3. Personalized Medicine
The capacity of AI to enable precision or customized medicine by customizing care based on patient data, including glucose trends, clinical history, lifestyle, and biomarkers, is a significant benefit in the management of diabetes. By combining these many inputs, AI models can improve risk assessment and treatment suggestions to maximize results for every patient (39,41).
This allows for the customization of treatment regimens that include insulin dosage, dietary modifications, and preventative measures, increasing efficacy and decreasing needless or ineffective interventions (39).
4. Reduced Healthcare Burden
Healthcare systems and providers have less work to do because to AI-driven automation and remote monitoring. To save time and clinical resources, automated algorithms that are connected with CGM, clinical decision support systems, mobile health apps, and closed-loop insulin delivery, for instance, eliminate the need for manual data interpretation and frequent in-person clinical visits (39, 41).
AI lowers hospitalizations, ER visits, and overall care expenses related to diabetes morbidity by spotting early warning signals of complications, forecasting the course of the disease, and assisting self-management tools (such as telemedicine and AI-powered coaching) (39).
5. Better Patient Adherence
Artificial intelligence (AI) solutions encourage better patient participation by offering ongoing feedback, prompts, and tailored advice. By making treatment recommendations more pertinent and useful, AI-enhanced solutions like smart CGM integration, smartphone apps for glucose management, and decision support systems can guide patients toward improved self-management, medication adherence, and lifestyle behaviour modifications (39,41).
Better long-term glycaemic control and fewer problems come from patients' improved adherence to medication, dietary modifications, and monitoring routines when they receive personalized insights and real-time support (39).
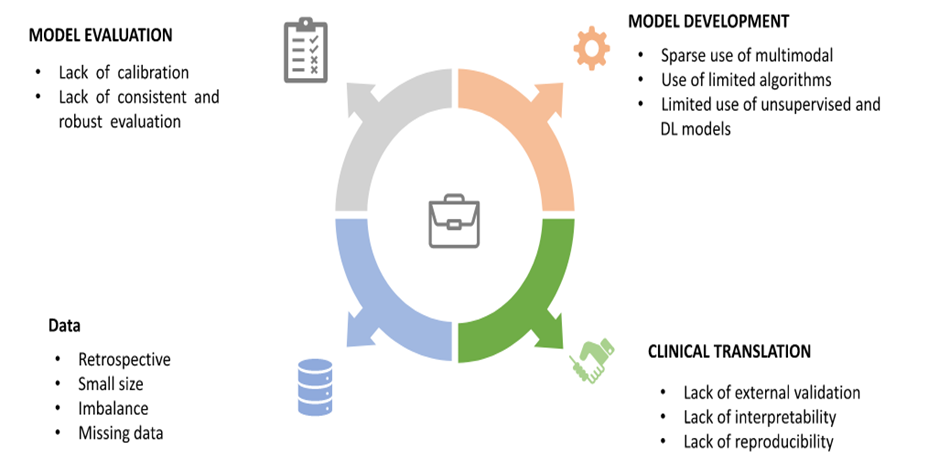
Limitations and Challenges
- Data privacy & security
- Bias in AI algorithms
- Lack of Indian population data
- Regulatory approval issues
- High cost of implementation


 Komal Chavan*
Komal Chavan*
 Dr. R. V. Shete
Dr. R. V. Shete
 Dr. M. M. Ghaisas
Dr. M. M. Ghaisas
 Tejas Digole
Tejas Digole
 10.5281/zenodo.19142746
10.5281/zenodo.19142746