
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
SNJB’s Shriman Sureshdada Jain College of Pharmacy, Chandwad, Nashik, India.
Effervescence is the formation of gas bubbles from a liquid as a result of a chemical reaction. Oral dosage forms continue to be the most popular method of administering pharmaceuticals, despite some disadvantages when compared to other methods, such as the potential for slow medication absorption, which can be avoided by giving the medication in liquid form, possibly allowing for a lower dosage. This systematic review and meta-analysis sought to evaluate the effectiveness of effervescent tablets with alternative chemical and physical methods for cleaning and sanitizing detachable prostheses by quantifying the reduction in biofilm, microbiological levels, and material stability. Despite several drawbacks as compared to other approaches, oral delivery formulations are the most often utilized technique of administering pharmaceuticals. The potential for delayed drug absorption is one of these drawbacks; this can be avoided by administering the drug in liquid form, which probably allows for a lower dosage.Due to their quick and simple administration, effervescent pills are growing in popularity beginning of activity. Usually, they contain carbonates or bicarbonates that react with acidic compounds rapidly when water is present, producing carbon dioxide and enhancing the flavor and solubility of APIs hiding. The pharmaceutical industry has given effervescent drug delivery systems (EDDS) a lot of attention because of their distinctive traits and possible benefits over traditional dosing forms.
Pharmaceutical dosage forms that are extensively used because of their benefits are effervescent tablets. They can be used in nutritional supplements as well as pharmaceutical medications. Although oral administration is commonly recognized as the most widely used method for systemic medication delivery that is non-invasive, Patient compliance and ease of use are not the sole advantages of effervescence technology. For many years, oral medication administration has been recognized as the most frequently used route of administration out of all the routes that have been used to administer drugs systemically through several medicinal items in a range of dosage forms. The reasons the oral route became so well-liked could be partly ascribed to its simplicity of use.1-2 Verbal Limited resources make a sustained drug delivery system difficult to use durations of gastric residence [2]. Rapid GI transit can prevent full drug release in the absorption zone, reducing the effectiveness of the amount that is given. Effervescent tablets are designed to dissolve or disperse in water prior to administration. The tablet is rapidly broken apart by the internal release of CO2 in the water, and the CO2 reaction is created when tartaric and citric acids interact with alkali metal carbonates or bicarbonates in the presence of water. Effervescent tablets are uncoated pills that usually include acids and carbonates or bicarbonates. 3, 4. For drugs that damage the stomach or that are sensitive to the pH of the stomach, several products are useful. Moreover, effervescent pills may be utilized to provide medications that are frequently recommended in high dosages [4]. When effervescent pills are submerged in water or juice, they split and dissolve, forming a solution. According to the USFDA's revised definition, an effervescent tablet is one that is meant to dissolve or scatter in water prior to administration. Acids/acid salt, carbonates and hydrogen carbonates, flavoring, sugar, and other ingredients that produce carbon dioxide when mixed with water make up effervescent tablets. The following chemical process occurs in effervescent tablets:
C6H8O7 (aq) + 3NaHCO3 (aq) → Na3C6H5O7 (aq) + 4H2O + 3CO2 (g) ↑
Citric acid + Sodium bicarbonate → Sodium citrate + Water + Carbon dioxide
The presence of water causes the aforementioned reaction since it is one of the reaction products that speeds up the process and makes it more difficult to halt. Because of this, effervescent products are manufactured and stored with as little contact to water as possible [2].
Fundamentals of Effervescent:
Effervescent pills produce carbon dioxide when they come into contact with water, which facilitates their dissolution. The medication is ready in solution and the pills dissolve entirely in a matter of minutes. A weak organic acid (such as citric or tartaric acid) and a carbonate or bicarbonate salt (such as sodium bicarbonate) react chemically to produce carbon dioxide when water is present. The high concentration of carbonate salt in the pharmaceutical solution causes the stomach's pH to temporarily rise after consumption, which leads to rapid gastric emptying. The upper small intestine is then stimulated to absorb the medicine, which is the primary site for medication absorption. Usually, doing this can result in higher drug absorption and a faster onset of impact. Therefore, effervescence has been used to produce medications that are hard to digest, irritate the stomach or esophagus (like aspirin), are sensitive to moisture or pH (like antibiotics or medications with large doses that are hard to swallow, like those that contain more than 2 g of drug), or have a quick onset of action (like analgesics) [2].
Pros of effervescent tablets:
Effervescent formulations are widely used dosage forms that offer a number of benefits over alternative pharmacological delivery systems. Some benefits of effervescent formulations following:
Quicker start of action: Unlike other forms of medication, effervescent formulations dissolve rapidly in water and are absorbed by the body. This could result in a quicker onset of treatment and a quicker alleviation of symptoms [8].
More practical: Since effervescent formulations dissolve in water, they will be handier for patients who have trouble swallowing [8].
Improved flavor: Effervescent formulations usually taste good, which can improve medication adherence and patient compliance [8].
Decreased gastrointestinal irritation: Effervescent formulations may mitigate the gastrointestinal discomfort caused by certain medications by buffering the stomach acid. Enhanced portability: Due to their small size, effervescent tablets are simpler to store and move around than liquid dosage forms [8].
Enhanced palatability: To improve the flavor of effervescent tablets and boost patient acceptability, flavoring chemicals are commonly included during manufacturing. Children and elderly individuals who may have difficulty swallowing standard tablets or capsules may find this to be extremely beneficial [8].
Good stability: In general, effervescent pills exhibit good stability. This results from the tablet packaging protecting the active ingredients from the elements, keeping them from coming into contact with moisture or oxygen, which can cause some drugs to degrade and lose their effectiveness [8].
Better absorption: Formulations that are effervescent have been made to dissolve quickly in water, which may facilitate the easier absorption of the active ingredients. This is to ensure that the drug is distributed more evenly and has a greater surface area throughout the effervescence phase, which will facilitate the body's absorption of the drug [8].
Cons of effervescent tablets:
Larger tablets: Effervescent tablets are frequently bigger than regular tablets, requiring special packaging [8].
Complex process: in contrast to conventional tablets, this need for the ideal humidity and temperature levels, this makes it a challenging and costly process [8].
Sensitive packaging: a desiccant must be included in the sensitive packaging because these pills are prone to hygroscopicity [8].
Required disintegration time: it may take up to five minutes for pills to disintegrate. The temperature of the water and the active ingredients present mostly dictate this. Off-notes are characteristics of certain active substances that cannot be covered up by sweets or flavorings. This will lead to a subpar final product [8].
Ideal Drug Candidates for EDDS
COMPOSITIONS OF EFFERVESCENT TABLETS
Active Pharmaceutical Ingredients (APIs)
Effervescent tablets are adaptable and may hold a variety of APIs, effervescent tablets can be used for a number of medicinal applications. Examples of popular APIs and the justification for their choice are provided below [12]:
Table 1: Popular Active Pharmaceutical Ingredients [6,12]
|
Category |
Examples |
Rationale for selection |
|
Analgesics and Antipyretics |
Aspirin, Paracetamol, Ibuprofen |
Fast onset of action due to rapid dissolution and absorption. |
|
Vitamin Supplement S |
Vitamin C, Vitamin B complex |
Improved stability and bioavailability, convenient for patients with swallowing difficulties. |
|
Electrolyte Replacement |
Sodium, Potassium salts |
Rapid replenishment of electrolytes for dehydration or sports recovery. |
|
Antacids |
Calcium carbonate, Magnesium hydroxide |
Quick relief from acidity and heartburn via rapid effervesces. |
|
Expectorants |
Bromhexine, Acetylcysteine
|
Enhanced solubility and faster action in respiratory conditions. |
|
Mineral Supplements |
Iron, Magnesium Zinc
|
Improved taste masking and absorption of essential minerals. |
|
Antibiotics
|
Amoxicillin, Ciprofloxacin
|
Suitable for pediatric or elderly patients requiring liquid |
Key Considerations for API Selection:
Excipients Used in Effervescent Tablets
Acid materials
Three primary sources of acidity are necessary for effervescence. Commonly used food acids include ascorbic, citric, and tartaric acids. These are selected because of their pleasant taste, mild odor, affordability, and ease of usage [6].
Citric acid
Its great taste and high solubility make it the preferred acidic ingredient in effervescent products. It is a white, crystalline powder that has no color. Coarse, medium, fine, and powder (for the anhydrous version) are the available particle sizes. Citric acid dissolves well in ethanol and water [6].
Tartaric acid
It is very soluble in water and hygroscopic in nature. Tartaric acid reacts with sodium bicarbonate similarly to citric acid, resulting in considerable effervescence. Tartaric acid, a diprotic acid with two acidic hydrogen atoms, requires more quantities to establish proper stoichiometric proportions than citric acid, a triprotic acid. Tartaric acid compresses similarly to citric acid [6].
Ascorbic acid
When it is crystallized, it appears white, and when it is finely powdered, it appears pale yellow. Because it is less hygroscopic than other acids, handling it during manufacturing is made easier. It is very soluble in pure ethanol and water (about one gram dissolves in three milliliters of water). 30 On the other hand, it gradually darkens when exposed to light. The rate at which carbon dioxide is released when sodium bicarbonate is used in an effervescent process is similar to that of other acids, including citric and tartaric acid [6].
Acid salts
Acid salts such sodium dihydrogen phosphate, amino acid hydrochlorides, and acid citrate salts are frequently found in effervescent formulations. Because of their rapid reaction with alkaline sources and water solubility, these acid salts were chosen. They act as pH buffers during the delivery of medications when combined with one of the previously stated acids. This buffering mechanism reduces any potential gastrointestinal negative effects while increasing the absorption of the therapeutic ingredient [6].
Carbon dioxide’s sources
Carbonate salts are commonly used to generate effervescence in formulations. Bicarbonate forms of carbonate salts are selected for their increased reactivity and enhanced effervescence action [6].
Sodium bicarbonate
With a yield of roughly 52%, sodium bicarbonate is the main source of carbon dioxide in effervescent compositions. It is available as fine powder and free-flowing granules, among other forms. It tastes alkaline and has no smell. Sodium bicarbonate starts to decompose into carbon dioxide, sodium carbonate, and water when heated to roughly 50°C. After being briefly heated to 250 to 300°C, it totally transforms into anhydrous sodium carbonate. Time and temperature affect the conversion process; at 93°C, 90% conversion is accomplished in 75 minutes. 34 Although sodium bicarbonate is non-elastic and has low compressibility, techniques like spray drying help get over this limitation. Additionally, grades can now be compressed instantly [6].
Potassium bicarbonate and potassium carbonate
These salts are more expensive and have poorer solubility than sodium salts. They can, however, be employed as partial substitutes for sodium salts when the number of sodium ions in a formulation must be reduced [6,7].
Binders
Generally speaking, a binder is frequently required to provide effervescent pills the required hardness for handling. However, the use of binders in effervescent drugs should be enhanced since they absorb moisture and stop pills from dissolving. When choosing how much binder to employ in a formulation, it's critical to strike the right balance between granule strength and disintegration times. Water can serve as an effective binder in wet granulation when all of the ingredients are granulated together. A small amount of water properly distributed across the powder bed helps to promote agglomeration and slightly dissolve the raw materials. Alternatively, granulating solutions of solvents such as ethanol and isopropanol can be used to dissolve dry binders [7].
Lubricants
When making effervescent products, choosing the correct lubricant is crucial because each one has unique chemical and physical properties. Since effervescent pills are not particularly soluble in water, many lubricants have a tendency to impede their breakdown, which must occur rapidly. Effervescent pills must generate a clear, clean solution that is devoid of any residue or insoluble film on the water's surface. When selecting a lubricant, it is important to consider its water solubility and compatibility for the active ingredient's medicinal activity. Sodium benzoate, sodium acetate, L-leucine, and carbowax 4000 are common lubricants used in the production of effervescents. According to the literature, lubricant combinations have also been studied, such as calcium and potassium sorbets and micronized polyethylene glycol (PEG) with trisodium citrate or calcium ascorbate. Spray-dried L-leucine and PEG 6000 work well together [6,7].
Additives
To enhance the flavor and look of effervescent liquids, trace amounts of chemicals are added. To accommodate sensory requirements, water-soluble flavors—like those of lemon, orange, and fruit essences are commonly used. Typically, these flavors account for 0.5 to 3.0% of the dosage. Sweeteners such as sorbitol, sucrose, aspartame, stevia, and saccharin sodium are frequently mixed with tastes to provide the desired sweetness. The dosage can contain anything between 0.1 and 3.5% of coloring chemicals. These consist of artificial FD&C colors as well as natural coloring ingredients. The inclusion of surfactants or antifoaming agents can further improve the effervescent preparation's performance during use [6].
Excipients used in effervescent formulation is mentioned below [14]:
Acids
Carbonates/Bicarbonates
Lubricants
Binders
Sweeteners
Flavours
Drug-Excipients compatibility study [13]:
For drug-Excipient’s compatibility study following excipients were studied which are used in the experiments:
Table 2: Excipients and Categories
|
Sr.no. |
Excipients |
Category |
|
1. |
Citric acid |
acidifying agent |
|
2 |
Tartaric acid |
acidifying agent |
|
3. |
Fumaric acid |
Acidulant |
|
4. |
Ascorbic acid |
Antioxidant |
|
5. |
Sodium bicarbonate |
alkalizing agent |
|
6. |
Sodium carbonate |
alkalizing agent |
|
7. |
Polyvinylpyrollidone-30 |
binding agent |
|
8. |
Polyethylene glycol-6000 |
binding agent |
|
9. |
Mannitol |
binding agent |
|
10. |
Sodium citrate |
buffering agent |
|
11. |
Sodium lauryl sulphate |
Lubricant |
|
12. |
Sodium benzoate |
Lubricant |
|
13. |
Acesulfame potassium |
Sweetener |
According to the functional category these excipients were mixed in the different ratio. These mixtures were kept at 40°C + 75% RH, and 45°C.
MECHANISM OF EFFERVESCENCE
A tablet that dissolves or disperses in water prior to ingestion is known as an effervescent tablet. Uncoated effervescent tablets contain carbonates or bicarbonates (such as sodium, potassium, alkali metal carbonate, and hydrogen carbonate) and acid or acid salts (such as citric, tartaric, and malic acid) that react rapidly with water to produce carbon dioxide. The release of CO2 gas improves taste masking and API dissolution [5]. The reaction between Citric acid and Sodium bicarbonate & Tartaric acid and Sodium bicarbonate, which results in liberation of carbon dioxide shown as follows,
C6H8O7.H2O+3NaHCO3 (aq) → Na3C6H5O7 + 4H2O + 3CO2 (g) ↑
Citric acid + Sodium bicarbonate → Sodium citrate + Water + Carbon dioxide
C4H6O6 + 2 NaHCO3→ Na2C4H4O6 + 2H2O + 2CO2 (g) ↑
Tartaric acid + Sodium bicarbonate → Sodium tartrate + Water + Carbon dioxide
Importance of CO? generation in drug delivery and patient compliance in effervescent tablet
The effervescent reaction, which generates carbon dioxide (CO?), plays a critical role in enhancing the drug delivery process:
Table 3: Role of CO? in Drug Delivery [17]
|
Aspect |
Details |
|
Improved Disintegration |
The CO2 gas produced during effervescence aids in breaking apart the tablet matrix, ensuring rapid disintegration. |
|
Enhanced Dissolution |
CO2 bubbles help to mix and disperse the drug evenly in the solution, improving its dissolution rate. |
|
Increased Absorption |
Faster dissolution leads to rapid drug absorption, which is particularly beneficial for APIs requiring immediate action. |
|
pH Modification |
The reaction can temporarily alter the pH of the solution, creating an environment favorable for certain APIs' solubility and stability. |
|
Stability of APIs |
Effervescent systems can protect sensitive APIs by ensuring they are dissolved in a buffered solution prior to administration. |
Table 4: Importance of CO? in Enhancing Patient Compliance [12,16]
|
Aspects |
Details |
|
Ease of Administration |
Effervescent tablets produce a palatable, fizzy drink, making it easier for patients who have difficulty swallowing pills. |
|
Taste Masking |
The effervescence masks unpleasant tastes and odors of the API, improving the sensory experience. |
|
Convenience for All Ages |
Suitable for children, elderly patients, and individuals with dysphagia, ensuring broader accessibility. |
|
Fun and Appeal |
The fizzing effect creates a pleasant and engaging experience, encouraging consistent use, especially in pediatric patients. |
|
Minimized Gastric Irritation |
Effervescent formulations dissolve completely before ingestion, reducing the risk of localized irritation in the gastrointestinal tract. |
Therapeutic Advantages Linked to CO? Generation
Rapid Onset of Action: Because effervescence facilitates rapid disintegration and absorption, the dosage form is suitable for conditions that require fast treatment (e.g., pain, fever, or acidity) [4].
Targeted Delivery: The CO? reaction can enhance medicine delivery to specific gastrointestinal areas by altering release characteristics [4].
FORMULATION DEVELOPMENT OF EFFERVESCENT TABLETS
Preformulation studies
Preformulation is a branch of pharmaceutical research that evaluates the physicochemical qualities of a drug ingredient using biopharmaceutical principles. Pre-formulation studies aim to identify the best specimen shape, assess the material's physical characteristics, and comprehend its consistency in different scenarios—all of which support the best possible drug distribution mechanism. Physiochemical properties that could affect the formation of an effective dosage form are the main focus of pre-formulation research. Lastly, the formulation design may be supported by these characteristics. The medication's time to market would be shortened, drug development costs would be decreased, and issues in the later phases of drug manufacturing would be lessened. It contains the details required to communicate the main idea of the treatment and to give a framework for mixing drugs with prescription excipients in dose forms [25].
Objective: Preformulation Testing aims to create relevant knowledge for formulation by designing appropriate, safe, and bioavailable dosage kinds [25].
Scope: The application of pre-formulation criteria increases the odds of producing an acceptable, safe, effective, and stable product [5].
Preformulation encompasses at least following tests:
Determination of melting point: The melting point of Drug was determined by using Thermocal melting point apparatus [6].
Bulk Density
A powder's bulk density is calculated by dividing its mass by its bulk volume. A powder's bulk density is greatly influenced by its conformation, content, and particle size distribution. The loose bulk density (LBD) and tapped bulk density (TBD) were also computed. After being shaken to break up any agglomerations that had formed, a carefully weighted powder (bulk) from each formula was put in a 25 ml measuring cylinder. At 2-second intervals following the determination of the starting volume, the cylinder was let to collapse under its own weight from a height of 2.5 cm onto a hard surface. The recording began when the volume rise was no longer perceptible [3].
Tapped Density
The tap density instrument was used with the same measuring cylinder after the bulk volume measurement. The tap density unit was changed by a drop of 300 taps per minute, which was effective for 500 taps. The volume was recorded as (Va) and then tapped 750 times to record (Vb). If the difference between Va and Vb is less than 2%, Vb is regarded as the final quantity tapped. The density that is tapped is calculated using the following formula [12].
Tapped density = Weight of powder / Tapped volume
Percentage Compressibility
The compressibility index of Carr was used to determine the granules' index of compressibility. It is evident in the way the powders are graded for their flow characteristics using Carr's Index.
Table 5: Grading of the powders for their flow properties according to Carr’s Index [25]
|
Consolidation Index (%) |
Flow |
|
5-15 |
Excellent |
|
12-16 |
Good |
|
18-21 |
Fair to passable |
|
23-35 |
Poor |
|
33-38 |
Very poor |
|
> 40 |
Very Very poor |
Drug–Excipients IR compatibility study
The medicine and excipients were kept at 40°C and 75% relative humidity for the duration of the drug compatibility study. IR tests were conducted every seven days for physical observation and final excipients. IR tests were performed on the pure drug to determine its composition and excipients. In the present investigation, the Prestige 21 Shimadzu FTIR Spectrometer was utilized to analyze the potassium bromide pellet [25].
Formulation strategies
Wet Granulation
Wet granulation is the most often used granulation method. This procedure involves wetting the material with a binder solution, followed by drying to produce dry granules [14].
The wet granulation method involves the following steps:
Wet Granulation through fluid bed Process
Effervescent tablets can also be made by fluid bed granulation. A binder solution is sprayed on the material once it has been added. The granules are then dried by blowing hot air from the bottom. A substantial amount of granulation fluid is needed for thought, which lengthens processing times and raises expenses [13,14].
Wet Granulation through fusion method
This process involves mixing an acid and a base (alkali), which is heated to release water. There is no need to introduce more external water as a granulating agent because the water generated during crystallization will function as a granulating agent. This technique has the potential to cause a chain reaction that generates too much moisture, so it must be stopped. Rarely, if effervescent composition is made using this procedure, a lower carbon dioxide content is seen. One drawback of this approach is the reported decrease in the mechanical strength of the product (tablets) [11,14]. When a medication or excipient is sensitive to heat or moisture, dry granulation is an essential pharmaceutical procedure used in the manufacture of tablets and capsules. Dry granulation uses mechanical force to condense powders into dense masses before milling them into granules, in contrast to wet granulation, which includes the addition of liquid binders and drying stages. This method is crucial for creating high-dose medications, compounds that are sensitive to moisture, and powders that are difficult to compress because it improves the flowability, compressibility, and homogeneity of powder mixes [3].
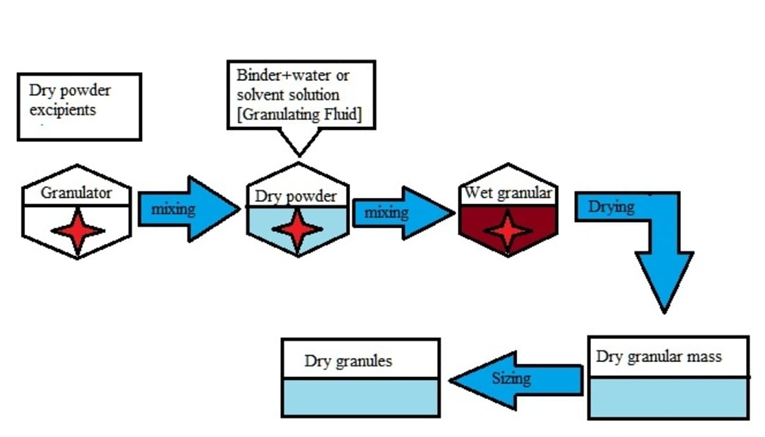
Figure 1: Wet granulation process for effervescent tablets
Dry Granulation
This type of granulation involves compacting a powder mixture without the use of heat or solvents. It is believed to be the least often used granulation method. The process involves compressing the powder combination to create a solid object first, and then grinding the compact to create granules. Slugging and compressing the powder with pressure rolls are two popular methods for dry granulation; the latter is usually done with a machine similar to the chilosonator [14,25]. The dry granulation method crushes the powder combination without the use of heat or a solvent. Among the granulation procedures, it is the least preferred. Compressing a material into a shape and then milling the compact to create a granule are the two basic operations. There are two ways to make dry granulation. The most popular method, slugging, produces the granules by recompressing the powder and then grinding the tablet or slug that results. The alternative method is to recompress the powder using pressure rolls and a device like a chilosonator [25].
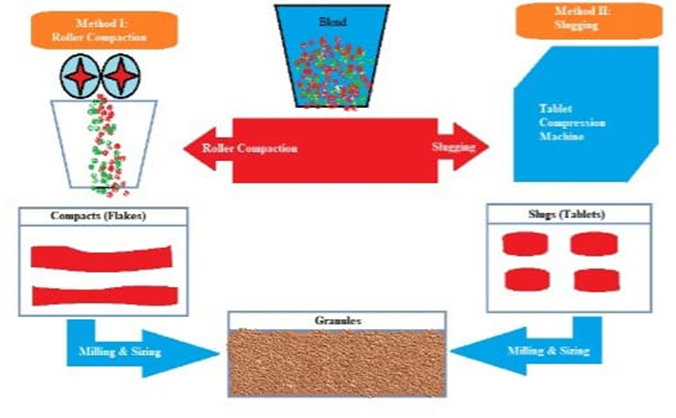
Figure 2: Dry granulation process for effervescent tablets
Roller compaction
The chilosonator uses pressure rolls to condense powder. Unlike tablet machines, the chilosonator operates constantly and generates a compact mass. The powder is fed between the rollers via a hopper with a spiral auger for easy entry into the compaction zone. Granules, similar to slugs, are formed after filtering or crushing the accumulated particles.50 The chilosonator can be used for granulating inorganic materials, compacting excipients, and creating formulations for both immediate and prolonged release [10,14].
MANUFACTURING OF EFFERVESCENT DOSAGE FORMS
Effervescent products need to be produced under particular circumstances to guarantee product stability even after packaging. Avoiding moisture absorption from the surrounding air is crucial because the majority of the basic materials used in the production of effervescents are very hygroscopic. This is required to prevent the effervescent reaction from starting before use and to ensure a consistent shelf life. The entire manufacturing process, including the dispensing and sifting of raw materials, granulation, mixing of additional components (if necessary), lubricant addition, compression, and packing, can be efficiently finished within a closed and integrated system. Using intermediate bulk containers and a specialized blender for materials handling helps minimize product transfers and exposure. When using open handling and pneumatic transfer systems, it is highly recommended to control the airflow in the manufacturing area to maintain the lowest possible moisture content. The recommended operating parameters for the whole manufacturing facility are to maintain a relative humidity (RH) of less than 20% and an even, consistent temperature of 21°C. It is enough to keep the granules or pills at a controlled room temperature of 25°C with a relative humidity of 25% to prevent them from sticking to the equipment and from absorbing moisture from the environment. 45, 46 [6].
Challenges in Manufacturing Effervescent Tablets: Moisture Sensitivity and Handling
Effervescent tablets are extremely sensitive to moisture, which can prematurely initiate the acid-base interaction, jeopardizing the product's purity, efficacy, and shelf life. The following are the issues linked with moisture sensitivity and handling during manufacturing, as well as solutions to solve them [10,11].
Table 6: Challenges Due to Moisture Sensitivity [14,19]
|
Challenge |
Impact |
|
Premature Effervescence Reaction |
Moisture during manufacturing might cause inadvertent CO? release, limiting product effectiveness. |
|
Degradation of Ingredients |
APIs and excipients can deteriorate in the presence of moisture, reducing stability and potency. |
|
Caking and Lump Formation |
Moisture absorption can cause powder blends to clump, reducing flow ability and homogeneity. |
|
Reduced Compressibility |
The physical characteristics of powders are changed by excessive moisture, which results in poor tablet formation. |
|
Packaging Complications |
Poor moisture management can make protective packaging less effective and raise the danger of spoiling. |
Table 7: Challenges in Handling Moisture-Sensitive Materials [6,13]
|
Challenge |
Impact |
|
Hygroscopic Ingredients |
Some excipients (such as sodium bicarbonate and citric acid) absorb moisture quickly, which increases their reactivity. |
|
Static Electricity Accumulation |
Dry handling circumstances can cause static buildup, which results in segregation and irregular mixing. |
|
Equipment Contamination |
Residual moisture on equipment might cause localized effervescent reactions during processing. |
|
Environmental Variability |
Variations in ambient humidity levels during production can result in uneven batch quality. |
Table 8: Strategies to Address Moisture Sensitivity and Handling Challenges [6,16]
|
Area |
Solution |
|
Material Handling |
|
|
Manufacturing Environment |
|
|
Processing Equipment |
|
|
Granulation Techniques |
|
|
Packaging |
|
|
Quality Assurance |
|
4. Advanced Solutions:
CoatingTechniques: Apply moisture-resistant coatings on effervescent granules to improve stability [11].
ModifiedBlends: Use hygroscopic inhibitors or additives that protect sensitive components.
Real-TimeMonitoring: Implement inline moisture sensors for continuous environmental monitoring [6].
Packaging considerations to ensure product stability:
EVALUATION OF EFFERVESCENT TABLETS
Post-compression parameters:
Weight variation:
To ascertain whether several tablet batches were consistent, weight variation was calculated. Each of the twenty tablets was weighed separately, and the average weight was determined by comparing the weights of the pills. The tablets pass the test if there are no more than two deviations from the percentage restriction and no tablet deviates by more than twice the percentage limit [2].
Tablet thickness:
For tablet size consistency, tablet thickness and diameter were crucial. Vernier Calipers were used to measure diameter and thickness [3,4].
Tablet hardness:
The hardness of tablets determines how resistant they are to breaking or shipping during handling, storage, and transportation conditions prior to use. The Monsanto Hardness Tester was used to measure the hardness of the tablets in each formulation. The hardness was measured in kilograms per centimeter. The force needed to shatter a tablet under diametric compression is known as tablet crushing strength or hardness. For uncoated tablets, a hardness of roughly 3-5 kg/cm2 is deemed adequate, and the force is expressed in kilograms [4].
Friability:
The tablet's friability was assessed using the Roche friabilator. In order to expose a tablet to the combined effects of shock and abrasion, this device rotates a plastic chamber at 25 rpm, plummeting the tablet six inches high with each rotation. The friabilator rotated a tablet sample 100 times after it had been previously weighed. The tablets were wiped with a soft cotton towel and then weighed again. The USP limit is between 0.5 and 1% [5].
Friability (%) = Initial weight (W1) - Final weight (W2)/Initial weight (W1) x 100
Measurement of effervescence time:
200 ml of filtered water at 20 °C ± 1 °C is placed in a beaker with one tablet. The effervescence period ends when a clear solution free of particles is achieved. It is necessary to give the average of each formulation's three measurements [6].
Determination of effervescent solution pH:
of solution is measured using a pH meter with one tablet in 200 milliliters of filtered water at 20 ± 1 °C as soon as the dissolve time is over. For every formulation, repeat the experiment three times [7].
Measurement of CO2 content:
Following the dissolution of one effervescent tablet in 100 milliliters of a 1N sulfuric acid solution, weight changes were recorded. The weight difference found is shown as the amount of CO2 (mg) per tablet. reports the averages of the three determinations [7].
Evaluation of the water content:
10 tablets of each formulation are dried in a desiccators containing of activated silica gel for 4 hours. Water content of 0.5% or less is acceptable [6].
Uniformity of content:
A random selection of ten tablets was made. Following its transfer into a 50 mL volumetric flask, each tablet was diluted with 50 mL of phosphate buffer (pH 6.8) and allowed to dissolve. One milliliter of this solution was diluted to one hundred milliliters using phosphate buffer (pH 6.8). UV spectroscopy at 246 nm was used to measure the amount of medicine in each pill [12].
Determination of the equilibrium moisture content:
Three desiccators are made using saturated salt solutions of potassium nitrate (to produce 90% relative humidity at 18°C), sodium chloride (71% relative humidity at 18°C), and sodium nitrite (60% relative humidity at 18°C). Each formulation's three tablets are put in a desiccator. The autotitrator device and the Karl Fischer method are then used to measure the equilibrium moisture content on the first day and seven days later [14].
In-vitro disintegration time:
Disintegration is the process by which a tablet breaks apart into small pieces. Using disintegration test equipment, the in-vitro disintegration time of a tablet was determined in compliance with I.P. Situations I.P. and prerequisites for I.P. Put one tablet into each of the six tubes in the basket. Attach a disk to each tube and use the phosphate buffer (pH-6.8) that is kept as the immersion solvent at 37 °± 2 °C to run the system. The phosphate buffer (pH-6.8) kept at 37 °C ± 2 °C can be used to raise and decrease the assembly at a pace of about 30 cycles per minute. The estimated time in seconds required for the tablet to fully dissolve was noted once there was no more palpable mass in the device. At 15–250C and 250C ± 10C (IP) in water, the usual disintegration time limit is 3 minutes [25].
Dissolution Studies:
The rate at which atorvastatin was released from mouth-dissolving tablets was measured using the USP Dissolution Research Apparatus II (Paddle type). 900 milliliters of phosphate buffer pH 6.8, kept at 37±0.50C, served as the dissolving media. Throughout the report, the paddle speed was kept at 50 rpm. Every five minutes, five milliliters of the extracted samples were diluted to ten milliliters. After that, five milliliters of fresh dissolving medium were replanted at the same temperature. Utilizing phosphate buffer pH 6.8 as the null, the materials were spectrophotometrically analyzed at 246 nm. To estimate the amount of drug released and the percentage of total drug released at various time intervals, the raw dissolving data was analyzed [25].
APPLICATIONS OF EFFERVESCENT TABLETS:
Effervescent tablets have broad applications in pharmaceuticals due to their unique formulation benefits, offering enhanced drug delivery and improved patient compliance. Below are their key therapeutic uses and emerging roles in personalized medicine and complex drug delivery systems [6]:
Table 9: Therapeutic Uses in Various Conditions [8,9]
|
Therapeutic Area |
Examples of APIs |
Application s and Benefits |
|
Pain Relief |
Aspirin, paracetemol, Ibuprofen |
|
|
Vitamin Supplementation |
Vitamin C, Vitamin B Complex |
|
|
Electrolyte Replacement |
Sodium, Potassium, Magnesium Salts |
|
|
Antacids |
Calcium Carbonate, Magnesium Hydroxide |
Effective for fast relief from acidity and heartburn. |
|
Respiratory Conditions |
Acetylcysteine, Bromhexine |
Facilitates mucus clearance and improved solubility for respiratory therapy. |
Effervescent tablets can be customized to cater to individual patient needs, making them a valuable tool in personalized medicine:
Table 10: Use in Personalized Medicine [16,17]
|
Feature |
Description |
|
Dose Flexibility |
Effervescent formulations can be tailored to deliver specific API doses, improving therapeutic precision. |
|
Combination Therapy |
Multiple APIs or supplements can be incorporated into a single effervescent tablet to simplify complex regimens. |
|
Patient-Specific Profiles |
Suitable for pediatric, geriatric, and dysphagic patients due to easy administration and pleasant taste. |
|
3D Printing Technology |
Enables precise customization of effervescent tablets based on patient-specific pharmacokinetics. |
Emerging Applications:
CHALLENGESAND FUTURE TRENDS IN EFFERVESCENT TABLETS:
Effervescent tablets have a number of difficulties, especially with relation to manufacturing complexity and stability. have a number of difficulties, especially with relation to manufacturing complexity and stability. Ongoing developments in excipients, manufacturing techniques, and formulation methods, however, are propelling improvements in their application for new APIs and combination treatments [14].
Table 11: Challenges [6,14:
|
Challenge |
Details |
|
Moisture Sensitivity |
|
|
Shelf-Life Limitations |
|
|
Complex Manufacturing Process |
Requires precise control of environmental conditions and particle size to ensure consistent quality. |
|
API Selection Limitations |
Not all APIs are suitable for effervescent formulations, particularly those sensitive to pH changes or heat. |
|
Cost Considerations |
Higher production costs compared to conventional tablets due to specialized equipment and packaging. |
1. Moisture-Resistant Excipients:
2. Multifunctional Excipients:
3. Taste-Masking Polymers:
1. Advanced Granulation Techniques:
2. 3D Printing in Effervescent Tablets:
3. Eco-Friendly Manufacturing:
CONCLUSION
A common dose type that has a number of benefits over alternative drug delivery techniques is effervescent tablets. Better bioavailability and a quicker beginning of effect are the results of their rapid absorption by the body and dissolution in water. Additionally, they taste better and are more convenient for people who have trouble swallowing, both of which improve patient compliance. Additionally, effervescent tablets have good stability, are easy to travel and store, and can lessen gastrointestinal distress. They can enable accurate dosing, enhance absorption, and stop first-pass metabolism. In conclusion, a promising path for the creation of pharmaceutical formulations is offered by the effervescent drug delivery method. It is a useful and adaptable choice in contemporary drug delivery because of its capacity to improve drug solubility, bioavailability, solve taste issues, and provides controlled and targeted drug release. Further breakthroughs in this field could result from ongoing research and development, broadening the range of therapy alternatives and eventually improving patient outcomes.
REFERENCES






Sidhesh Nahata, Sajanraj Kankariya, Rohan Gugaliya, Krishna Kurne, Jay Pardeshi, Dr. Ganesh Basarkar, Effervescent Tablets: Advances in Formulation, Manufacturing Challenges, and Future Prospects in Drug Delivery, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 7, 1687-1705. https://doi.org/10.5281/zenodo.15869807
 10.5281/zenodo.15869807
10.5281/zenodo.15869807