
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shivlingeshwar college of pharmacy, Almala. Tq. Ausa. Dist.Latur, Maharashtra, India. pin.413520.
Alcoholic liver disease (ALD) a range of liver conditions caused by prolonged excessive alcohol intake. These conditions can progress from fatty liver (steatosis) to alcoholic hepatitis, fibrosis, cirrhosis and ultimately liver cancer (hepatocellular carcinoma). ALD poses a considerable worldwide health challenge leading to significant illness, death and healthcare expenses. This review offers a thorough summary of present understanding regarding ALD, converting its occurrence, symptoms, methods of diagnosis, treatment options, and results. We draw upon resent advancement in treatment allocation, statistical methodologies, and personalied medicine, as highlighted in the latest literature. Despite progress in understanding the pathogenesis and management of Ald, challenges persist in early diagnosis, risk Stratification, and optimising individualised care. Integration of novel treatment frameworks, such as distributional targets and personalised prediction models, holds promise for enhancing therapeutic outcomes. The review concludes bye summarizing key findings and identifying avenues for future research and clinical practice improvement.
Alcoholic liver disease (ALD) refers to a range of liver damage resulting from sustained and excessive alcohol consumption it is primary contributor to liver related illness and health globally, with its worldwide impact increasing due to changes in the drinking habits and socioeconomic conditions. (ALD) Alcoholic liver disease progress through stages, starting with fatty liver (hepatic steatosis) and moving to inflammation (steatohepatitis), scarring (fibrosis), severe scarring (cirrhosis) and potentially liver cancer (hepatocellular carcinoma). The progression and symptoms of alcoholic liver disease (ALD) are affected by genetic, environmental, metabolic, add behavioural influences, which makes diagnosis and treatment more complex. Recent years have witnessed significant advances in the application of statistical decision theory, causal inference, and personalised treatment allocation to chronic diseases, including ALD. These developments, particularly in the domains of treatment recommendations with distributional targets, covariate-based policy optimization, and model averaging for heterogeneous data sources, have profound implications for both research and clinical practice this review integrates these methodological perspectives with the traditional clinical framework to provide a holistic examination of a ALD. [1][2]
SATAGES OF ALCOHOLIC LIVER DISEASE:[21]
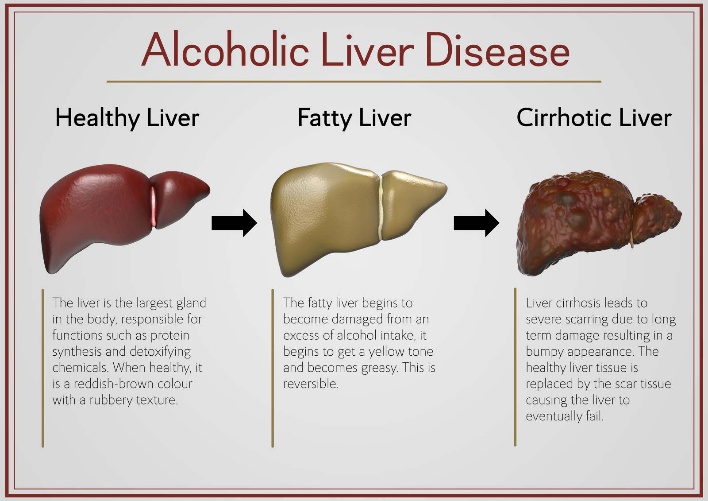
CLASSIFICATION:
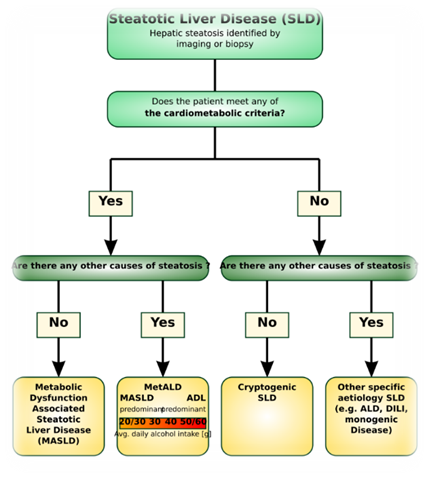
Hepatic steatosis or the build-up of fat in more than 5% of liver cells is the first of several disease types that fall under the broader category of steatotic liver disease (MASLD) describes the condition when at least one metabolic risk factor is present. The term metALD (metabolic dysfunction an alcohol related liver disease) applies when there is also a rise in alcohol use. This differs from alcohol related liver disease Ald where the steatotic liver disease is mainly due to alcohol intake. Therefore, MASLD is distinguished from other causes such as monogenic diseases, Drug induced liver injury (DILI), and cryptogenic SLD. Steatohepatitis (MASH) is a condition where steatosis occurs alongside inflammation and sometimes fibrosis. It can develop from SLD through various pathways and potential liver injuries. Complications such as cirrhosis hepatocellular carcinoma may arise from MASH.[6][7][8]
EPIDEMIOLOGY
global burden:
Ald is a major contributed to liver related death and accounts for significant proportion of cirrhosis and liver cancer cases globally. The incidence and prevalence of ALD vary by region, reflecting differences in alcohol consumption pattern, cultural practices, and healthcare infrastructure. In high income countries, ALD remains a leading indication for liver transplantation, and its burden is expected to increase in parallel with rising alcohol use in low- and middle-income nations.
RISK FACTORS:
the development and progression of ALD are influenced by multiple interacting factors:
Trends in social determinants:
socioeconomic status, mental health disorders, and access to healthcare also shape the epidemiology of ALD. Patterns of alcohol marketing, policy interventions, and public health campaigns significantly impact population level outcomes.
CLINICAL MANIFESTATION:
spectrum of disease:
ALD manifest across a continuum, with the severity and reversibility of each stage verying markedly:
Alcoholic fatty liver steatosis: characterised by hepatic lipid accumulation, often asymptomatic and reversible with abstinence.
Alcoholic hepatitis: presents with acute on chronic liver injury, jaundice, Coagulopathy, n systemic inflammatory response; mortality is high in severe cases.
Alcoholic fibrosis and cirrhosis: progressive scarring and architectural distortion of the liver, leading to portal hypertension, hepatic synthetic dysfunction, and risk of decompensation.
Hepatocellular carcinoma (HCC): ALD is a recognised risk factor for HCC, particularly in the context of cirrhosis
SYMPTOMS AND SIGNS:
clinical presentation ranges from asymptomatic elevation of liver enzymes fulminant hepatic failure. Common features include:
extrahepatic manifestations:
ALD is associated with multisystem complications, including pancreatitis, cardiomyopathy, and neuropsychiatric disorders.
Comorbidities:
Type 2 diabetes, insulin resistance, and metabolic syndrome which is characterized by at least three of sugar, high serum triglycerides, and low serum high density lipoprotein are strongly linked to or cause the condition. Hormonal disorders (panhypopituitarism, hypothyroidism, hypogonadism, polycystic ovary syndrome), persistently elevated transaminases, aging, and hypoxia from obstructive sleep apnea are also linked to it some of these conditions predict the course of the disease.
Most normal weight individuals with MASLD (lean MASLD) are sedentary, have elevated liver lipid levels and cardiovascular disease risk, and have impaired insulin sensitivity. These are the results of increased hepatic de novo lipogenesis, decreased fat storage capacity, and decreased mitochondrial function in fat. MASLD patients had a higher risk of severe COVID-19 infection, according to a recent systematic review, but there was no difference in mortality between MASLD and non-MASLD patients. [9][10][11][12]
PATHOPHYSIOLOGY:
Fatty changes:
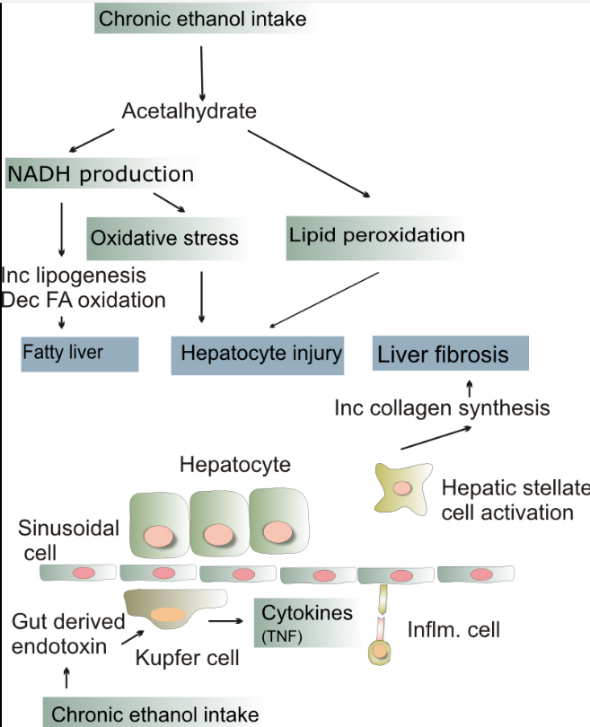
we still don't fully understand how ALD works. The liver detoxifies 80% of alcohol. Prolonged alcohol use causes oxidative stress Kama lipid peroxidation, acetaldehyde toxicity and the release of pro inflammatory cytokines (TNF-alpha, interleukin 6, and interleukin 8). These elements lead to liver cell inflammation, apoptosis, and ultimately fibrosis. It is still unknown why this only happens in a small number of people. furthermore, the liver has a remarkable capacity for regeneration; it can continue to function normally even when 75% of its hepatocytes are dead. [14] The buildup of fatty acids in liver cells is known as steatosis or fatty change. Under a microscope these appear as fatty globules. After a few days of heavy drinking alcoholism can start to develop large fatty globules (macro-vesicular steatosis) throughout the liver. Alcohol dehydrogenase (ADH) breaks down alcohol into acetaldehyde, which is subsequently broken down into acetic acid by aldehyde dehydrogenase (ALDH) which is ultimately oxidized into carbon dioxide (CO2) and water (H2O). The NADH/NAD + ratio rises as a result of this process. Fatty acid synthesis is induced by higher NADH concentrations, whereas fatty acid oxidation is inhibited by lower NAD concentrations. The liver cells are then prompted by the increased fatty acid levels to convert it to glycerol in order to create triglycerides. These triglycerides accumulate, resulting in fatty liver.[15]
Alcoholic hepatitis:
Hepatocyte inflammation is a hallmark of alcoholic hepatitis. Alcoholic hepatitis affects 10% to 35% of heavy drinkers (NIAAA, 1993). Although the amount of alcohol consumed has no direct correlation with the development of hepatitis, some individuals appear to be more susceptible to this reaction than others. This condition is known as alcoholic steato-necrosis, and it seems that the inflammation makes liver fibrosis more likely. By causing apoptosis and severe hepatotoxicity, inflammatory cytokines (TNF-alpha, IL-6, and IL-8) are believed to be crucial in the development and maintenance of liver damage and cytotoxic hepatomegaly.
Increased intestinal permeability brought on by liver disease is one potential explanation for the elevated TNF-a activity. This makes it easier for the endotoxin produced in the gut to enter the portal circulation. After the endotoxin is phagocytosed by the liver's Kupffer cells, TNF-a is released. TNF-a then activates caspases to initiate apoptotic pathways, which leads to cell death.[16][17]
Cirrhosis:
Cirrhosis is a late stage of serious liver disease that causes inflammation (swelling), fibrosis (cellular hardening), and damaged membranes that stop the body from getting rid of chemicals. This leads to scarring and necrosis (cell death. Heavy drinkers have a 10% to 20% chance of getting cirrhosis of the liver (NIAAA, 1993). Acetaldehyde may be responsible for alcohol-induced fibrosis by stimulating collagen deposition by hepatic stellate cells. The production of oxidants derived from NADPH oxidase and/or cytochrome P-450 2E1 and the formation of acetaldehyde-protein adducts damage the cell membrane. Symptoms include jaundice (yellowing), liver enlargement, and pain and tenderness from the structural changes in damaged liver architecture. Without total abstinence from alcohol use, cirrhosis will eventually lead to liver failure. Late complications of cirrhosis or liver failure include portal hypertension (high blood pressure in the portal vein due to the increased flow resistance through the damaged liver), coagulation disorders (due to impaired production of coagulation factors, ascites (heavy abdominal swelling due to a buildup of fluids in the tissues) and other complications, including hepatic encephalopathy and the hepatorenal syndrome. Cirrhosis can also result from other causes than hazardous alcohol use, such as viral hepatitis and heavy exposure to toxins other than alcohol. The late stages of cirrhosis may look similar medically, regardless of cause. This phenomenon is termed the "final common pathway" for the disease. Fatty change and alcoholic hepatitis with abstinence can be reversible. The later stages of fibrosis and cirrhosis tend to be irreversible but can usually be contained with abstinence for long periods of time. [18][19]
DIAGNOSIS:
Clinical Assessment:
Diagnosis of ALD relies on a combination of clinical history, physical examination, and laboratory evaluation. Key elements include:
Laboratory Investigations:
Typical laboratory findings in ALD include:
Imaging and Histology:
Prognostic Scoring:
Several scoring systems have been developed to predict outcomes in ALD, including the Maddrey discriminant function, Model for End-Stage Liver Disease (MELD), and Glasgow Alcoholic Hepatitis Score. These tools assist in risk stratification and treatment decision-making.
Advances in Statistical Diagnosis:
Recent methodological contributions, such as distributional targets and empirical likelihood approaches; offer robust, nonparametric alternatives to traditional model-based inference, improving accuracy in heterogeneous and sequentially collected clinical data (Kock et al.,
2022; Zou et al., 2018). These methods facilitate individualized risk estimation and more nuanced treatment allocation.
TREATMENT:
General Principles:
The cornerstone of ALD management is complete abstinence from alcohol, which can halt or even reverse the early stages of liver damage. Comprehensive care includes nutritional support, management of complications, and psychosocial interventions.
Pharmacological Therapies:
Management of Complications:
Standard protocols for cirrhosis complications apply, including diuretics for ascites, beta-blockers for portal hypertension, and lactulose for hepatic encephalopathy.
Liver Transplantation:
Liver transplantation is the definitive treatment for end-stage ALD. Selection criteria typically require a period of abstinence (usually six months), though recent debates challenge the rigidity of this "six-month rule," especially in life-threatening cases.[20]
Psychosocial Interventions:
Integrated approaches combining pharmacotherapy, counselling, and support groups are essential for sustained abstinence.
Personalized Treatment Allocation:
Recent theoretical advances emphasize the need for individualized treatment recommendations based on patient-specific characteristics and preferences. Kock et al. (2022) highlight the use of distributional characteristics such as inequality, welfare, and poverty measures—in guiding policy recommendations, recognizing that optimal treatment may involve mixtures or sequential allocation strategies rather than a uniform approach. This framework allows for the incorporation of constraints (e.g., resource limitations, patient heterogeneity and tailors interventions to maximize population-level and individual outcomes (Kock et al., 2022).
Similarly, the integration of covariate information into sequential treatment allocation policies, as developed by Kock et al. (2020), enables dynamic, data-driven decision-making that adapts to patient profiles and evolving evidence. These methods have been shown to achieve near minimax optimality in expected regret, ensuring that only a minimal proportion of patients receive suboptimal treatments relative to their individualized risk profile (Kock et al., 2020).
Model Averaging and Distributed Data:
In the context of multi-center and real-world data, model averaging approaches-such as the tree-based ensemble proposed by Tan et al. (2022) facilitate accurate estimation of conditional average treatment effects (CATE) without requiring centralization of patient-level data. This is especially relevant for ALD, where patient populations and treatment responses may vary across institutions and regions. By leveraging site-specific models and adaptively combining them, these frameworks improve estimation accuracy and inform personalized care, even in the presence of heterogeneity and privacy constraints (Tan et al., 2022).
RESULT
Clinical Outcomes:
The prognosis of ALD varies by disease stage, patient characteristics, and response to intervention. Key outcome metrics include:
Impact of Personalized and Distributional Approaches:
Application of advanced statistical frameworks has yielded several notable results:
Simulation and Empirical Evidence:
Monte Carlo simulation studies and real-data applications confirm the superiority of these novel methodological approaches over conventional methods in terms of bias, coverage, and efficiency. For example, the density-based empirical likelihood ratio test demonstrates higher power and smaller average sample number than classical nonparametric tests in health-related treatment effect evaluations (Zou et al., 2018).
DISCUSSION
Challenges in ALD Management:
Despite advances in understanding and therapy, several challenges persist in ALD care:
Advancements in Decision Science and Personalized Medicine:
Incorporation of statistical decision theory and personalized medicine principles into ALD research and practice addresses some of these challenges by:
Limitations and Future Directions:
While promising, these approaches also face limitations:
Future research should focus on integrating these methods into electronic health records, developing user-friendly decision support tools, and conducting prospective studies to evaluate their impact on patient outcomes.
CONCLUSION
Alcoholic liver disease remains a formidable public health challenge, characterized by a complex interplay of biological, behavioral, and social determinants. Advances in statistical methodology, treatment allocation theory, and personalized medicine offer new avenues for optimizing care and improving outcomes. By synthesizing clinical knowledge with cutting-edge decision science, this review underscores the potential of individualized, data-driven approaches to transform ALD management. Continued interdisciplinary collaboration and methodological innovation are essential to realize these benefits and address the persistent burden of ALD worldwide.
REFERENCES



Sayyed Mehraj J., Durgam Gautam Banayya, Giri Ashok B., Alcoholic Liver Disease: A Comprehensive Review, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 4260--4269. https://doi.org/10.5281/zenodo.18790089
 10.5281/zenodo.18790089
10.5281/zenodo.18790089