
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Kamalakshi Pandurangan College of Pharmacy, Ayyampalayam, Tiruvannamalai, Tamil Nadu, India
The aminopenicillin class of ?-lactam antibiotics includes the commonly used ampicillin. It has been widely used to treat respiratory tract infections, urinary tract infections, gastrointestinal infections, meningitis, and septicaemia because of its potent activity against a wide range of Gram-positive and Gram-negative bacteria. Ampicillin binds to penicillin-binding proteins (PBPs) to prevent the formation of bacterial cell walls. The introduction of organisms that produce ?-lactamases has restricted its effectiveness, despite its long-standing clinical utility. This article offers a thorough analysis of ampicillin's pharmacology, mode of action, pharmacokinetics, clinical uses, resistance mechanisms, side effects, and most recent research findings.
Antibiotics transformed the way infectious diseases were treated, greatly lowering morbidity and mortality rates across the globe. Penicillin continue to be one of the most significant types of antibacterial drugs among them. In order to increase antibacterial coverage, especially against Gram-negative pathogens, ampicillin, a semi-synthetic derivative of penicillin, was produced in the early 1960s [1]. In contrast to natural penicillin, ampicillin has an amino side chain that increases its ability to penetrate Gram-negative bacteria's outer membrane, broadening its antibacterial range.
Ampicillin's price, safety, and shown effectiveness have led to its widespread usage in both hospital and community settings. Neonatal sepsis, enterococcal infections, and meningitis brought on by Listeria monocytogenes are among the illnesses for which it is still essential [2]. But growing resistance has led to the cautious and sensible use of this antibiotic.

Fig.1 Historical development of penicillin antibiotics
2. Chemical Structure and Classification:
Ampicillin is a member of the β-lactam antibiotic subclass known as aminopenicillin. Its structure's β-lactam ring is necessary for its antibacterial effectiveness, and the amino side chain increases its hydrophilicity and ability to enter Gram-negative bacterial cells [3].
The presence of the amino group distinguishes ampicillin from natural penicillin and broadens its antimicrobial spectrum. Improved activity against pathogens like Escherichia coli and Haemophilus influenzae is made possible by the structural alteration of ampicillin in comparison to penicillin G. But because of its structure, β- lactamase enzymes can break it down [4].
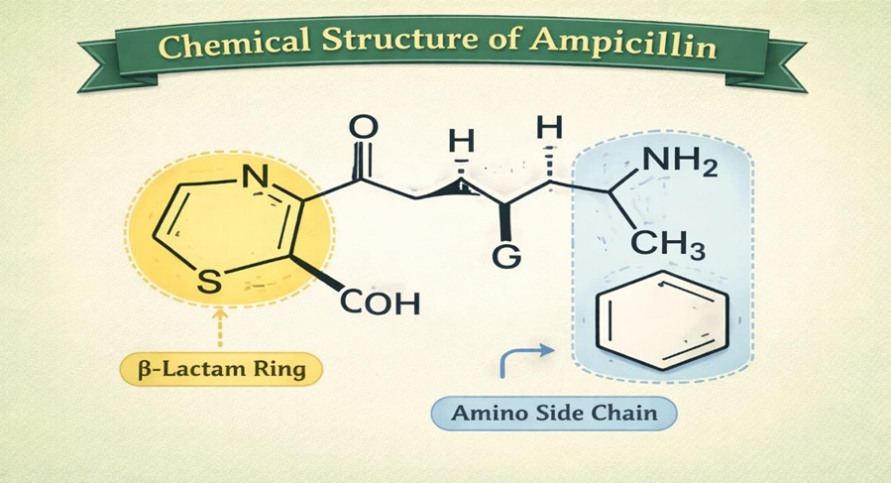
Fig.2 Chemical Structure of Ampicillin
3. Mechanism of Action:
Ampicillin works against bacteria by preventing the creation of their cell walls. It attaches itself to enzymes called penicillin-binding proteins (PBPs) that are involved in the last phases of peptidoglycan cross-linking in the bacterial cell wall [5]. This inhibition causes an osmotic imbalance and ultimately bacterial cell lysis by weakening the structure of the cell wall.
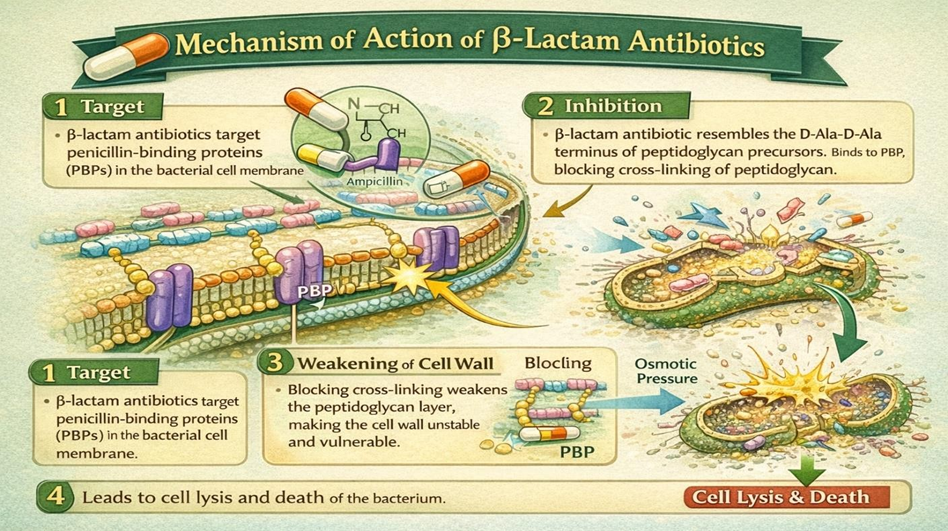
Fig.3 Mechanism of action of β-lactam antibiotics on bacterial cell wall synthesis
Ampicillin works best against germs that divide quickly since it is a bactericidal agent. Since its mode of action is time-dependent, treatment efficacy depends on keeping the medication concentration above the minimum inhibitory concentration (MIC) [6].
4. Pharmacokinetics and Pharmacodynamics:
Ampicillin can be given intravenously, intramuscularly, or orally. When taken with food, oral absorption may be diminished [7].
Oral absorption: 40–50%
Distribution: During meningeal inflammation, it is widely dispersed throughout bodily fluids and tissues, including bile and cerebrospinal fluid.
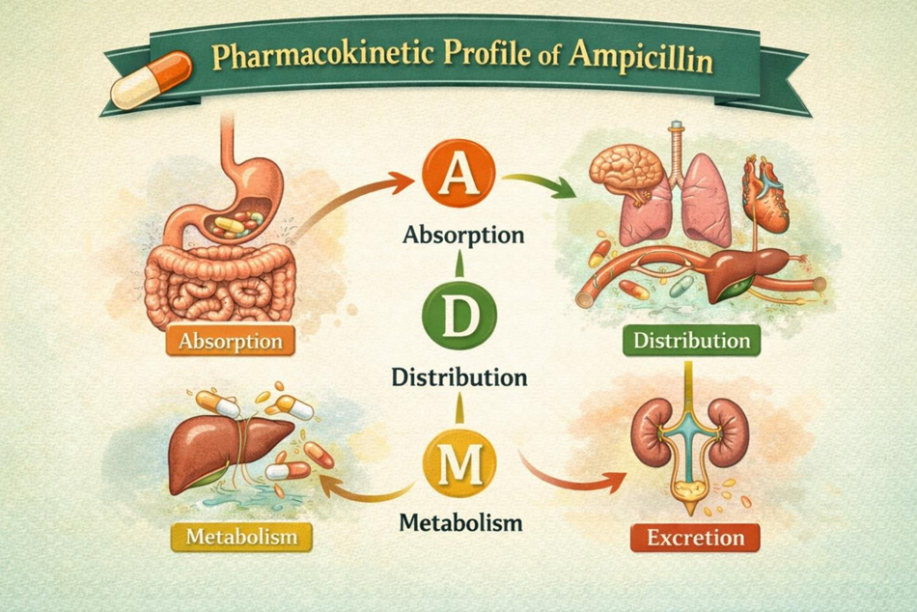
Fig.4 Pharmacokinetic profile of Ampicillin
Elimination: Mostly eliminated unaltered by the kidneys
Half-life: 1 to 1.5 hours
Hepatic metabolism is minimal.
For patients with renal impairment, dose modifications are required to avoid toxicity and buildup. Ampicillin's bactericidal action is pharmacodynamically dependent on time above MIC rather than peak concentration [8].
5. Clinical Indications:
Ampicillin is indicated for the treatment of various bacterial infections, including:
It is frequently used in pediatric and obstetric care due to its established safety profile.
6. Spectrum of Activity:
Ampicillin demonstrates activity against a broad range of microorganisms.
Gram-positive bacteria
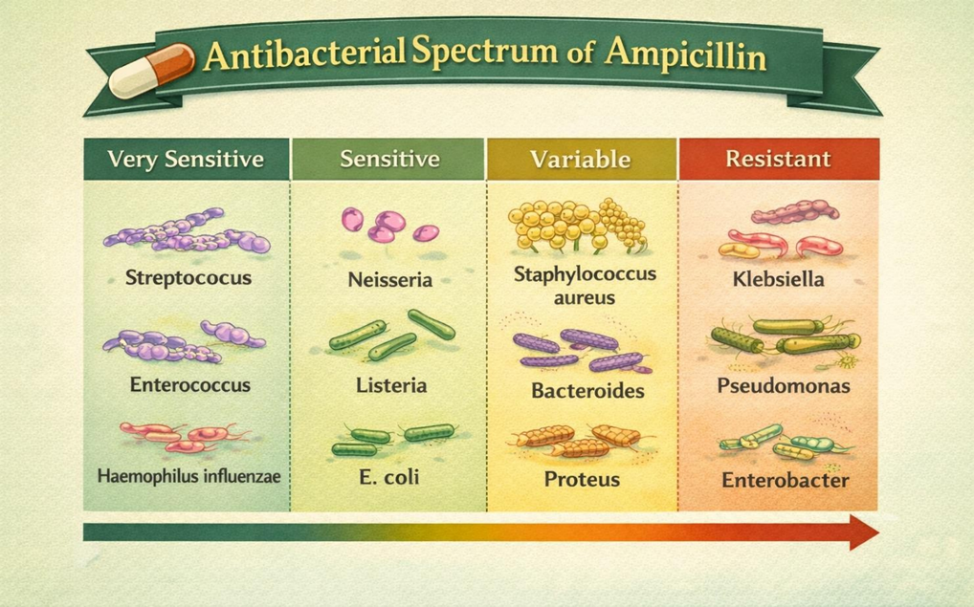
Fig.5 Antibacterial Spectrum of Ampicillin
Gram-negative bacteria
However, ampicillin is ineffective against penicillinase-producing Staphylococcus aureus and Pseudomonas aeruginosa [11].
7. Resistance Mechanisms:
A number of factors have contributed to the rise in ampicillin resistance. The formation of β-lactamase enzymes, which hydrolyse the β-lactam ring and make the antibiotic inert, is the most frequent method [12]. Changes in PBPs, reduced membrane permeability, and active efflux pumps are additional resistance mechanisms.
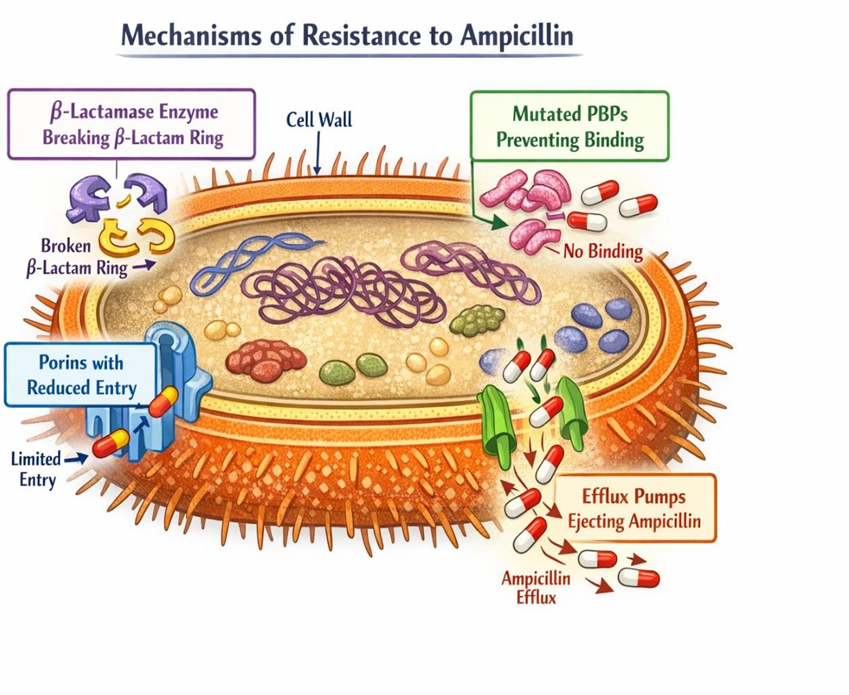
Fig.6 Mechanisms of Resistance to Ampicillin
Ampicillin is frequently used in conjunction with β-lactamase inhibitors, including sulbactam, which shield the antibiotic from enzymatic breakdown, to combat resistance [13].
8. Adverse Effects and Safety Profile:
In general, ampicillin is well tolerated. Gastrointestinal problems as nausea, vomiting, and diarrhoea are frequent side effects [14]. Skin rashes are common, especially in patients with viral diseases such as infectious mononucleosis. Anaphylaxis, hypersensitivity reactions, pseudomembranous colitis, and haematological abnormalities such thrombocytopenia and anaemia are examples of serious adverse events [15].
9. Drug Interactions:
Numerous medications may interact with ampicillin. Probenecid raises the plasma concentration of ampicillin by reducing its renal excretion [16].
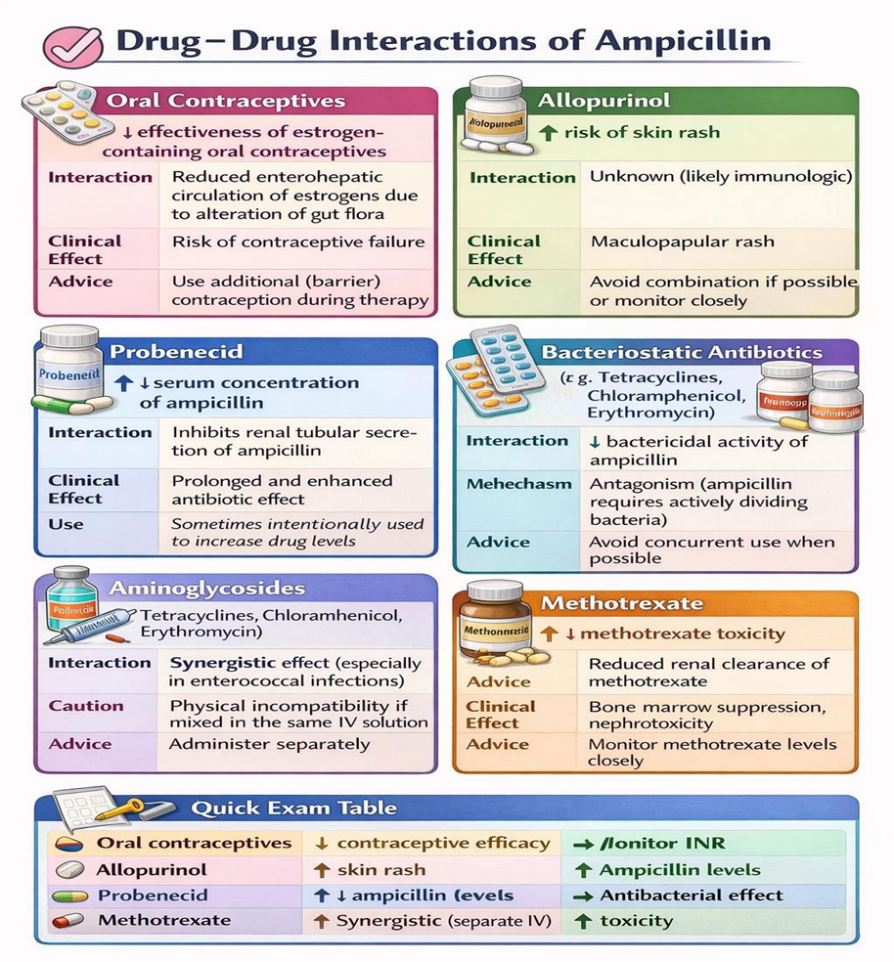
Fig.7 Drug Interactions of Ampicillin
Skin rashes are more likely to occur when allopurinol is used concurrently. By changing gut flora, ampicillin may potentially lessen the efficacy of oral contraceptives [17]. Ampicillin may interact with oral contraceptives, reducing their efficacy, and with other antibiotics, particularly bacteriostatic agents, potentially reducing bactericidal activity [17][18].
10. Special Considerations:
Pregnancy: Ampicillin is categorised as Pregnancy Category B, meaning that neither appropriate controlled research with pregnant women nor animal studies have shown any risk to the foetus [18]. Because of its well-established safety profile, it is frequently chosen for treating bacterial infections in expecting mothers and is generally regarded as safe for usage during pregnancy. To prevent any possible difficulties for the mother or newborn, careful monitoring is recommended, especially during the third trimester.
Renal Impairment: Patients with impaired renal function are at risk for drug accumulation, which can result in toxicity, including neurotoxic consequences such convulsions in severe situations, because ampicillin is mainly eliminated unaltered by the kidneys [8][10]. It is advised to modify the dosage in accordance with creatinine clearance. To maintain therapeutic levels, ampicillin should ideally be given to haemodialysis patients following dialysis sessions.
Paediatrics: Ampicillin is often used to treat meningitis and neonatal sepsis brought on by Group B Streptococcus or Listeria monocytogenes [10]. The medication is usually well tolerated, and paediatric dosage is weight-based. Neonates' renal function and level of hydration are particularly important since their developing kidneys can change how well drugs are absorbed.
Geriatrics: Older adults often have reduced renal function, even if serum creatinine appears normal due to decreased muscle mass. Monitoring renal function in geriatric patients is essential when prescribing ampicillin to avoid drug accumulation and toxicity [8][18]. Additionally, elderly patients may be more susceptible to gastrointestinal side effects, such as diarrhoea or pseudomembranous colitis, requiring careful observation.
11. Advantages:
12. Limitations:
CONCLUSION
Ampicillin's broad-spectrum efficacy, safety, and affordability make it a valuable antibacterial agent. It is an essential component of therapeutic treatment due to its ability to treat both gram-positive and some gram-negative illnesses. To maintain its clinical relevance, however, the emergence of resistant bacterial strains calls for prudent use, rigorous adherence to antibiotic stewardship, and continuous research into combination medicines, innovative formulations, and alternative delivery modalities [21]. Ampicillin can continue to be a key component of efficient infection control with judicious administration.
REFERENCES






R. V Sivaprakash, S Sathish, K Udayanithi, N Vadivarasan, D Rajalingam, N Ganasekar, Ampicillin: A Contemporary Exploration of Its Therapeutic Role, Pharmacology, and Resistance Challenges, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 3594-3601. https://doi.org/10.5281/zenodo.18726379
 10.5281/zenodo.18726379
10.5281/zenodo.18726379