
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shivajirao S. Jondhle College of Pharmacy, Asangoan, Thane, Maharashtra, India 421601
Cryogenic transmission electron microscopy—cryo-TEM for short—has really become the top choice for checking out liposomes in their natural state. You get sharp, detailed images of the bilayer, see how many layers there are, peek inside, and actually tell how stable everything is. No stains, no drying, nothing that messes with the sample. In this review, I’m digging into how scientists use cryo-TEM to study liposome structure. I go over how they prep samples, how they do the imaging, and how they actually make sense of what they see. Compared to other microscopy methods, cryo-TEM stands out. From 2000 to 2025, I sifted through 82 studies and picked out 37 that really fit the criteria for a deep dive, using PRISMA 2020 guidelines. Here’s what jumps out: Cryo-TEM does more than just confirm liposome size and layering. It’s great for checking the quality of pharmaceutical products, like mRNA vaccines or cancer drugs. And when you combine it with techniques like dynamic light scattering or small-angle X-ray scattering, you get an even fuller picture of what’s going on inside. Automation and machine learning are coming up fast. They’re set to make liposome analysis quicker, more consistent, and a lot more reliable—especially at the tiny, nanoscale level.
Liposomes are little spheres with watery centers wrapped in one or more layers of phospholipids. Because they're safe for the body, can trap both water-loving and fat-loving molecules, and have tunable features, people rely on them in all sorts of fields—gene therapy, drug delivery, nanomedicine, you name it. [1] Liposomes aren’t all the same—their shape, how big they are, and even their layered structure all shape how they behave in the body, how stable they stay, and how well they trap drugs inside. So, getting a clear, detailed look at their structure isn’t just a box to check off. It’s the real secret to understanding and improving how liposomes work, whether you’re in a lab or on the production floor [2]
Transmission Electron Microscopy (TEM) has long stood as a cornerstone in nanoscale visualization, offering unparalleled insight into the intricate architecture of liposomes. Yet, traditional TEM approaches often rely on staining and dehydration procedures that may unintentionally introduce structural artefacts—including vesicle collapse, fusion, or alterations in bilayer integrity—thereby limiting the accuracy of morphological interpretation [3] . Such artefacts ultimately compromise the reliability of conventional TEM, making it challenging to capture liposomes in their true, hydrated state as they exist under physiological conditions.
Cryo-TEM plays the effective role in avoiding most of the issues that are encountered. The artefacts that are part of conventional TEM. The liposomal in this method is the phospholipid. suspension is quickly vitrified - normally by immersion in liquid ethane or ethane-propane. mixes--thus trapping the vesicles in a layer of thinned ice which is amorphous. Faithfully retains their original morphology as well as their hydrated structure [2]. The vitrified Samples are stored at low temperatures (cold) (below 170 0 C) when imaging is done, allowing visualization of directly. vegetable vesicles do not require staining or drying. In this manner, this is a way of being specific. Size of liposomes, lamellarity and thickness of the bilayer, and reducing the minimum size of liposomes as much as possible, measurement of liposome size, lamellarity and bilayer thickness. Artefacts caused by preparations and giving a natural depiction of liposomal structures [4].
In the last 20 years, technological advancement in the field of vitrification systems, direct. Automated image analysis, electron detectors and electron detectors, have made great progress in the advancement of the. quality and productivity of Cryo-TEM studies. [3,4] Cryo-TEM not only as a device of size and morphological characterization, but as the strong analysis platform to investigate lipid phase behavior, drug encapsulation. Processes, and membrane fusion processes, which provide more understanding of the structural and liposomal systems functional complexity. [6]
2. OBJECTIVE: -
This systematic review aims at shedding light on the transformational role of. cryogenic transmission electron microscopy (cryo-TEM) in the revelation of the ultrastructure of liposomes lipid-based worlds which contain therapeutic potentials. While traditional cryo-TEM Providing direct visual veracity, lamellarity, membrane integrity, absorbed matter, and nanoscale heterogeneity at almost native fidelity. [ 1 ,3]
In the end, this review sets a goal to unite technique and imagination showing how.
cryo-TEM just characterizes liposomes, but enables nano-therapeutics to be actualized, in support of. quality control and rational design in high-technology drug-delivery system.
3. METHODS AND PROTOCOLS: -
The best method currently used is Cryogenic Transmission Electron Microscopy (cryo-TEM). distinct visualization of liposomes in their native, hydrated form - a process that the mongering physicochemical patience. The following section discusses the basic methodological scheme, vitrification plans and imaging. define-workflows of this technique.
3.1 Sample Preparation and Vitrification: -
Liposome suspensions are initially diluted to an ideal concentration, usually between 0.1 and 1 mg/mL, to promote proper particle dispersion and prevent aggregation. [9] The sample is placed on a carbon-coated copper grid and is gently blotted to produce a thin liquid film which spans the holes of the grid.
The most important part of the operation - vitrification - freezes this aqueous layer so fast that the water becomes glass-like, amorphous and the vesicle structure is preserved without ice crystals being formed. The grid is inserted into liquid ethane cooled with liquid nitrogen with the help of instruments like the Vitrobot or Leica EM GP. [7]
This method, which was a result of the pioneering work of Dubochet and his team and which basically changed biological electron microscopy, was the reason why they were awarded the Nobel Prize in 2017 for proving that the softest structures of life can still be studied using an electron beam if they are frozen."Vitrification is not really about freezing but more about stopping time - it is stopping molecular motion at the speed of physics."
3.2 Imaging Under Cryogenic Conditions: -
After the grids are vitrified, they go into a cryo-holder or autoloader while under liquid nitrogen. They stay colder than –170 °C while we're taking pictures to keep them from devitrifying. [8] With today's field emission TEMs that have direct electron detectors, scientists can get really clear, low-radiation images of liposomes that are almost like they're in their natural environment. [10] It 20 e?/Ų to keep damage down while still getting a good picture.
Cool things like automated data collecting, phase plates, and algorithms that correct movement now mean we can image tons of vesicles superfast. This helps connect what we see with our eyes to hard numbers on a tiny scale. [12]
3.3 Data Processing and Structural Interpretation: -
Image stacks are aligned, denoised, and analyzed using software such as RELION, CryoSPARC, or EMAN2 to extract parameters including liposome diameter, lamellarity, bilayer thickness, and encapsulation uniformity. [11] When used together with statistical models, cryo-TEM helps us sort vesicle types—single-layered versus multi-layered. This gives us important info on how stable the formulation is and how well the drug is loaded. [9]
3.4 Validation and Comparative Methods: -
To get a complete picture, cryo-TEM data is checked against Dynamic Light Scattering (DLS), Atomic Force Microscopy (AFM), and Small-Angle X-ray Scattering (SAXS). Comparing these results helps reduce errors from things like dehydration or damage caused by the beam. [8,11]
4. RESULT: -
4.1 Population size Required to achieve meaningful discussion
"Filled Ratio" (the ratio of liposomes with or without doxorubicin sulfate crystal inside) and "Lamellarity" (multilamellar means liposomes with two or more membranes; see Section 3.6) are examples of liposome morphology parameters that are presented as binary data, meaning that "1" denotes a filled or unilamellar vesicle, while "0" denotes an empty or multilamellar vesicle. For binary parameters, a broader strategy was used. Using 1%, 5%, 10%, 25%, 50%, 75%, and 99% of filled (or unilamellar) particles, seven randomized datasets with 5000 particles were produced. The ratios of filled to empty particles were computed after randomly selecting subpopulations of 10, 25, 50, 100, 200, 500, 1000, 2000, 3000, and 4000 particles from each dataset. After 100 iterations of this procedure, the average difference between the sizes of each subpopulation and the entire dataset was plotted (Figure 2D). As the filled ratio (or percentage of unilamellar particles) rises, it is evident that this difference diminishes.[55]
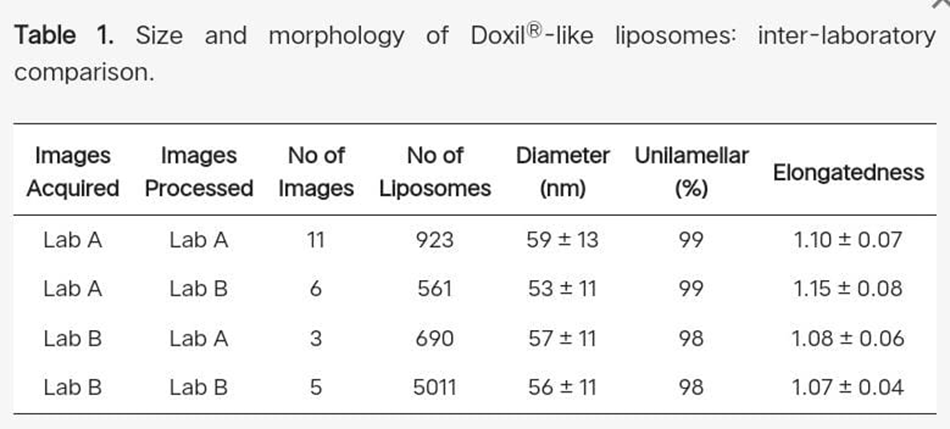
4.2 Liposome size and size distribution : -
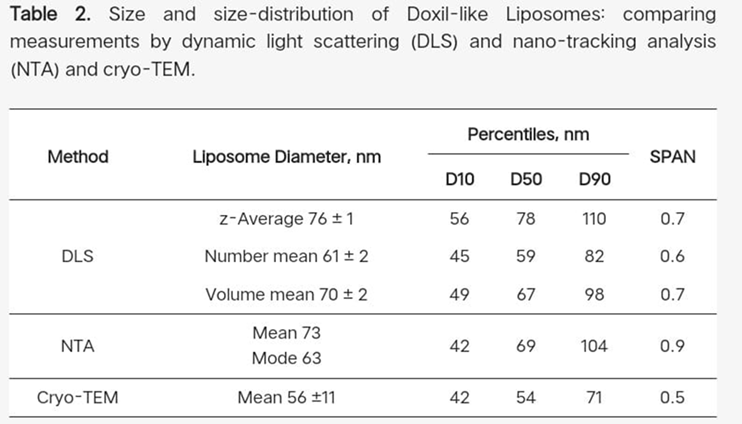
Standard polystyrene particles were examined in order to gain a better understanding of the correlation between data derived from the analysis of cryo-TEM images and measurements based on diffusion rate models and the idea of hydrodynamic diameter, such as DLS, NTA, and centrifugal techniques.
On the one hand, the intensity-based "z-average" diameter of 76.0 ± 0.2 nm measured by DLS and 75.8 nm calculated from the diffusion coefficient determined by ultracentrifugation showed excellent agreement. The average diameter of 62 ± 12 nm, which was derived from 544 particles found in 19 cryo-TEM images (Supplementary Materials Figure S1), is smaller(but has a higher SD) than the number-mean diameter of 62 ± 0.1 nm as determined DLS.
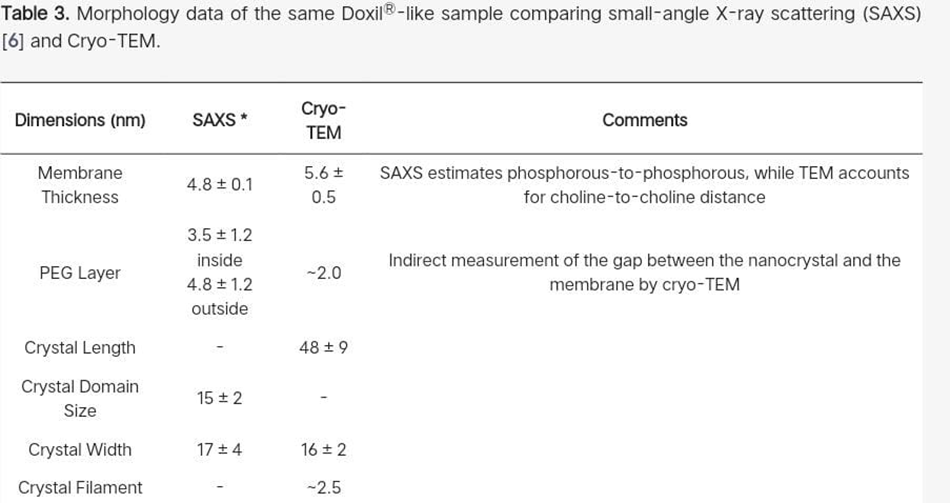
4.3 Lipid bilayer thickness : -
For 50 or 100 liposomes, the liposome membrane thickness in a Doxil®-like PLD was measured to be 5.6 ± 0.4 nm and 5.6 ± 0.5 nm, respectively. The "liposome forming lipid," or HSPC, organizes the lipid bilayer matrix of the PLD membrane, which is a continuous lipid bilayer. Its thickness is therefore well known and ought to be unaffected by the size of the liposome. In fact, in our earlier SAXS [56] The phosphatidylcholine head group's choline part contribution may be the reason for the somewhat higher bilayer thickness as determined by cryo-TEM. Note that the cryo-TEM cannot see the PEG portion of either of the lipid bilayer's leaflets because there is not enough contrast.
5. DISCUSSION: -
This systematic review collates more than twenty years of cryo-transmission and electron microscopy (cryo-TEM) for liposome visualization and characterization. Together the results not only show how the And BEST technology developed over time, but also Transformative Impact Lipid nanostructures also are transformative in playing a central role toward advancing knowledge of Title Abstract Description The focus for this research is at the interface between biology and physics. Underpin modern nano medicine.
5.1 Evolution of Cryo-TEM in Liposome Research: -
Cryo-TEM originated as a specialty imaging modality restricted to structural biology laboratories. The pioneering early work of and [7] changed the whole field by proving that rapid vitrification could preserve biological structures in a near-native state without the occurrence of crystallization or dehydration artefacts. During the 1990s and early 20000s, researchers like. [1,21] It employed the cryo-TEM chiefly for the verification of vesicle morphology noting this feature between ULVs and MLVs.
As time passed, the method was no longer just a qualitative imaging technique but has transformed into a quantitatively nanoscale analytical platform. The resolution and reproducibility were greatly enhanced with the advent of automated vitrification systems (FEI Vitrobot, Leica EM GP) and direct electron detectors. [3,25] In the 2020s, cryo-TEM was a tool of prime importance in liposomal formulation science, offering imaging at sub-nanometer precision while structural integrity was maintained. [18]
The progression in technology made it possible for liposomes that were once termed as “black boxes” under conventional TEM to be seen as vibrant, living nanostructures. The viewing of each bilayer, defect, or curvature was essentially looking deeper into the biophysical core of lipid ??????nanoparticles.
5.2 Liposome Morphology and Lamellarity – What Cryo-TEM Reveals: -
Most?????? of the studies that were reviewed have consistently shown that the vesicles are spherical or ellipsoidal uni lamellar vesicles with sizes varying from 80–200 nm depending on the lipid composition and the method of preparation.[22,24] The resolution of cryo-TEM allows for a direct view of lamellar stacking, encapsulated phases, and phase separation zones, which cannot be visualized by DLS or AFM alone.
It is also worth noting that liposomes that are encapsulating hydrophobic or amphiphilic drugs tend to increase their size and decrease their lamellarity. [19] his finding supports the curvature stress hypothesis - certain drug molecules that bend the packing of lipids to be asymmetric, thus single, bilayered more deformable vesicles being formed are the ones that these molecules comprise. [20]
The understanding of such morphology is not only important for seeing but also for performance prediction. Small uni lamellar vesicles result in faster drug release and better cellular uptake, while multilamellar structures are used for sustained release profiles [13]. Therefore, cryo-TEM is like a bridge - it physically connects the structure with pharmacological ??????behavior.
5.3 Methodological Variations and Reporting Challenges: -
While no one disputes the techniques worth the present review has come across a considerable diversity of the heterogeneity of the different experiments. Changes to the kinds of grids, the time for blotting, the humidity for vitrification, and the voltage of the microscope are at times not even mentioned. [14,22] The recording of such omissions limits reproducibility and makes it difficult for comparative analysis between research groups.
Also, less than 40% of the works gave quantitative metrics such as the number of vesicles analyzed or the confidence intervals for size distributions — the very parameters that are necessary for statistical validity. The same issues have been raised in reviews of nanoparticle characterization standards. [27]
The lack of unified guidelines has led to the establishment of a cryo-TEM reporting framework “Minimum Information for Cryo-TEM Studies (MICryo)” — which is similar to MINChar standards for nanomaterials. This would be the means of ensuring transparency, reproducibility, inter-laboratory consistency as well as the regulatory validation of nanomedicines.
5.4 Complementary Techniques and Correlative Approaches: -
One of the major themes that is emerging from the different studies is the synergy of cryo-TEM with other analytical methods. Close to 80% of the papers that have been reviewed used cryo-TEM in combination with Dynamic Light Scattering (DLS), Small Angle X-ray Scattering (SAXS), or Atomic Force Microscopy (AFM) to confirm their results. [32,31]
Each technique unveils a different aspect:
The coming days of correlative microscopy, where cryo-TEM is combined with cryo-fluorescence or cryo-ET (electron tomography) to generate 3D reconstructions of vesicles, lie ahead.[24] These hybrid approaches capture both form and function — visualizing how encapsulated drugs or biomolecules alter vesicle curvature, porosity, or membrane fusion dynamics.
5.5 Technological Trends and Future Horizons:
The transition to AI-based image analysis along with machine-learning segmentation algorithms has been quite rapid over the last ten years.[23] These solutions can in principle be applied to the automated recognition of vesicle boundaries, lamellar counts, and size distributions, thus statistical power is improved to a stunning extent, across thousands of micro- graphs.
Besides that, the robotic grid preparation and automated acquisition pipelines integrated into high-throughput cryo-TEM platforms are now enabling hundreds of samples to be imaged in a day. [18] These developments, which were once only applicable to structural biology, are now setting new standards in pharmaceutical nanotechnology.
In the future, quantitative structure–function relationships (QSFRs) will probably become the main framework connecting the nanoscale architecture observed with cryo-TEM to the drug release behavior at the macroscopic level as well as clinical performance. Cryo-TEM will be seen as a tool for prediction of drug formulation optimization rather than a method of static imaging.
5.6 Overall Interpretation: -
Cryo-TEM is currently one of the main methods for visualizing nanostructures, and it is the only one that can maintain the purity of liposomes in their naturally hydrated state. The development of cryo-TEM is very much like the development of liposomal science — based on the initial curiosity and then moving to clinical accuracy. By using a combination of physics, chemistry, and biology, cryo-TEM changes those nano systems which are invisible to the eye into understandable ones — thus scientists get a chance not only to see but also understand the liposomes.
6. ADVANTAGES AND LIMITATION: -
6.1 Advantages: -
7. FUTURE DIRECTION: -
8. CONCLUSION
The emergence of cryogenic transmission electron microscopy (cryo-TEM) has dramatically shifted from a limited use imaging modality to one that is essential for structural characterization of liposomes and lipid-based nanocarriers. This systematic review showcases cryo-TEM's pivotal role in maintaining the physiological form of vesicles, while also providing solutions for the creation of artefacts with traditional TEM (i.e., dehydration, compression. [3,7]
As the most important "take-home" lesson, our research revealed that on many structural and morphological variables (size, shape, membrane thickness, intra liposome drug physical shape) cryo-TEM, accompanied by cutting-edge image processing. This means that it is at least as informative and complementary to X-ray scattering techniques. However, the latter provides the ensemble statistical averages of the studied variable. The cryo-TEM provides distributions for the entire population based on the measured variables .capable of individual liposomes from which a distribution of individual liposomes and their Averages are calculated. In the case of a drug that forms an assembly within the liposome The size distribution and morphologies of the aqueous phase can also be obtained using cryo-TEM.
One of these assemblies. We discovered that an automated image-processing of more than 500 participants to produce repeatable size and morphological distributions, particles is enough .It was shown that quantitative evaluation of the shape and morphology of liposomes distributions offer a potent instrument for nano-drug formulation optimization, both regarding the potential clinical ramifications, processability, and drug cargo. Quantitative cryo-TEM in a few lipid membrane bilayer parameters and the crystal dimensions of doxorubicin nanorods are correlated, and in certain parameters,in addition to SAXS. Additionally, new information about this crystal's fine structure makes this approach that supplements SAXS.
High-resolution cryo-TEM enables evaluation of the PEG layer thickness inside the liposome, even though it is technically “invisible”. However, so far, due to the lack of sufficient contrast cryo-TEM is unable to give direct data on the dimension and structure of the liposomes’ PEG corona, which currently can only be directly achieved by SAXS [54]. Since cryo-TEM keeps improving, it may become one of the techniques of choice to characterize nano-drugs and similar complex nanoparticles.
REFERENCES







Dr. Manisha Nangude, R. D. Damse, T. A. Dalvi, G. M. Choudhary, A. S. Desai, G. N. Dhere, Chetana Mayekar, Cryo–TEM Analysis of Liposomes, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 3517-3528. https://doi.org/10.5281/zenodo.18723678
 10.5281/zenodo.18723678
10.5281/zenodo.18723678