
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Adina Institute of Pharmaceutical Sciences, Sagar, M.P., India
Rheumatoid arthritis (RA) is a chronic autoimmune disorder characterized by systemic inflammation and progressive joint damage. Conventional therapeutic agents often exhibit limited efficacy due to poor bioavailability and systemic side effects. Quercetin, a naturally occurring flavonoid with potent anti-inflammatory and antioxidant properties, has demonstrated therapeutic potential in RA; however, its clinical application is hindered by low solubility and bioavailability. This study focuses on the development and evaluation of quercetin-loaded nanoparticles (QNPs) for targeted drug delivery in RA treatment. Biodegradable polymeric nanoparticles were prepared using a nanoprecipitation method and optimized for particle size, zeta potential, encapsulation efficiency, and drug release kinetics. The optimized formulation exhibited a particle size below 200 nm with high encapsulation efficiency and sustained drug release over 48 hours. In vitro studies demonstrated enhanced cellular uptake and anti-inflammatory activity in RA-relevant cell lines, while in vivo studies using a collagen-induced arthritis (CIA) model in rats revealed significant reductions in paw swelling, inflammatory cytokines (TNF-?, IL-6), and histopathological scores compared to free quercetin. These findings suggest that QNPs offer a promising strategy for targeted and effective delivery of quercetin in the management of RA, potentially minimizing systemic toxicity and improving therapeutic outcomes
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disorder characterized by persistent synovial inflammation, joint destruction, and progressive disability. It affects approximately 1% of the global population and imposes a significant socio-economic burden due to its long-term treatment requirements and associated complications. The current pharmacological approaches, including non-steroidal anti-inflammatory drugs (NSAIDs), corticosteroids, disease-modifying antirheumatic drugs (DMARDs), and biologics, often provide only symptomatic relief and are associated with a range of adverse effects upon prolonged use. Moreover, these agents typically lack site-specificity, leading to systemic toxicity and reduced patient compliance. Flavonoids, particularly quercetin, have garnered attention for their natural anti-inflammatory, antioxidant, and immunomodulatory properties. Quercetin has been reported to inhibit the production of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6—key mediators in the pathogenesis of RA. Despite its therapeutic potential, the clinical translation of quercetin is severely hampered by its poor water solubility, rapid metabolic degradation, and low oral bioavailability. In recent years, nanotechnology has emerged as a promising strategy to overcome these pharmacokinetic challenges. Nanoparticle-based drug delivery systems offer multiple advantages including improved solubility, protection from premature degradation, controlled release, and enhanced accumulation at inflamed tissues via the enhanced permeability and retention (EPR) effect. Polymeric nanoparticles, in particular, have shown potential in delivering bioactive compounds to inflamed joints with high precision and minimal systemic exposure.
Review of Literature
Previous studies have explored the encapsulation of quercetin within various nano-carriers to enhance its stability and bioavailability. For instance, quercetin-loaded liposomes and solid lipid nanoparticles have demonstrated improved anti-inflammatory responses in vitro and in vivo. Poly(lactic-co-glycolic acid) (PLGA) and other biodegradable polymers have been widely employed for the formulation of nanoparticles due to their biocompatibility and FDA approval for clinical use. Notably, quercetin-loaded PLGA nanoparticles have shown sustained drug release, enhanced cellular uptake, and superior therapeutic efficacy in models of inflammatory diseases. However, the application of such nanoformulations specifically in the context of RA, particularly with comprehensive in vitro and in vivo validation, remains an area requiring further exploration.
Objective of the Study
The primary objective of this research is to develop and evaluate a quercetin-loaded nanoparticle system for targeted drug delivery in the treatment of rheumatoid arthritis. The specific aims are as follows:
MATERIAL AND METHOD
All the materials used were of analytical grade
Table No. 1
|
Material |
Description |
Supplier |
|
Quercetin |
A flavonoid with antioxidant and anti-inflammatory properties. |
Sigma-Aldrich |
|
Poly(lactic-co-glycolic acid) (PLGA) |
Biodegradable polymer used for nanoparticle formulation. |
Evonik (Resomer®) |
|
Chitosan |
Biopolymer for mucoadhesive and biocompatible nanoparticles. |
Heppe Medical Chitosan |
|
Lecithin |
Phospholipid used for lipid-based nanoparticles. |
Lipoid |
|
Glyceryl Monostearate |
Solid lipid used for solid lipid nanoparticles. |
Gattefossé |
|
D-α-Tocopheryl Polyethylene Glycol 1000 Succinate (TPGS) |
Surfactant for improved solubility and stability. |
Eastman Chemical Company |
|
Polyvinyl Alcohol (PVA) |
Stabilizer to prevent nanoparticle aggregation. |
Alfa Aesar |
|
Acetone |
Organic solvent for nanoprecipitation. |
Fisher Scientific |
|
Ethanol |
Organic solvent for emulsification techniques. |
Thermo Fisher Scientific |
|
Tween 80 (Polysorbate 80) |
Surfactant to stabilize nanoparticles. |
Croda International |
Procedures for Solvent Evaporation Method Batches
Batch 1: Base Formulation To prepare the base formulation, 10 mg of Quercetin and 100 mg of PLGA were dissolved in 5 mL of acetone. An aqueous phase was prepared by dissolving PVA at 1% w/v concentration and Tween 80 at 0.5% w/v in distilled water. The organic phase containing Quercetin and PLGA was added dropwise to the aqueous phase under continuous stirring at 800-1000 rpm. The acetone was allowed to evaporate while stirring for 3-4 hours. The solution was centrifuged at 10,000 rpm for 30 minutes to collect the nanoparticles. The nanoparticles were washed thoroughly with distilled water to remove any unreacted materials, then dried for further evaluation.
Batch 2: Chitosan-Coated Nanoparticles In this batch, 10 mg of Quercetin and 100 mg of PLGA were dissolved in 5 mL of acetone. A 0.2% w/v chitosan solution was prepared by dissolving chitosan in 0.1% acetic acid. The organic phase containing Quercetin and PLGA was added dropwise to the chitosan solution while stirring at 800-1000 rpm. The acetone was allowed to evaporate completely, enabling the chitosan to coat the forming nanoparticles. After the solvent had evaporated, the solution was centrifuged at 10,000 rpm for 30 minutes to collect the chitosan-coated nanoparticles. The nanoparticles were washed with distilled water to remove unbound components and dried for subsequent analysis.
Batch 3: Lipid-Enhanced Nanoparticles For lipid-enhanced nanoparticles, 10 mg of Quercetin, 50 mg of PLGA, and 50 mg of Glyceryl Monostearate were dissolved in a solvent mixture of acetone and ethanol (1:1, 5 mL total). An aqueous phase containing 1% w/v PVA and 0.5% w/v Tween 80 was prepared. The organic phase was added dropwise into the aqueous phase under sonication (20 kHz) or high-speed stirring to form an emulsion. The organic solvents were allowed to evaporate while stirring, resulting in the formation of hybrid nanoparticles. The solution was centrifuged at 10,000 rpm for 30 minutes to separate the nanoparticles, then washed with distilled water and dried for characterization.
Batch 4: TPGS-Enhanced Nanoparticles In this batch, 10 mg of Quercetin and 100 mg of PLGA were dissolved in 5 mL of acetone. An aqueous phase containing 1% w/v PVA, 0.5% w/v Tween 80, and 0.2% w/v TPGS was prepared. The organic phase containing Quercetin and PLGA was added dropwise to the aqueous phase under constant stirring at 800-1000 rpm. The acetone was allowed to evaporate over 3-4 hours, during which the nanoparticles were formed. The mixture was centrifuged at 10,000 rpm for 30 minutes to collect the TPGS-enhanced nanoparticles. They were washed with distilled water to remove unbound materials and dried for further evaluation.
Characterization
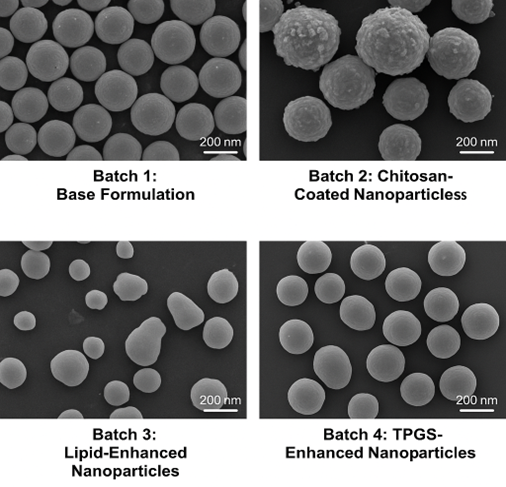
Fig no. 1 SEM images of All four batches
Table No. 2. Trial Batches for Quercetin Nanoparticles (Solvent Evaporation Method)
|
Batch No. |
Quercetin (mg) |
PLGA (mg) |
Chitosan (% w/v) |
PVA (% w/v) |
Tween 80 (% w/v) |
Glyceryl Monostearate (mg) |
TPGS (% w/v) |
Solvent (mL) |
Remark |
|
1 |
10 |
100 |
- |
1.0 |
0.5 |
- |
- |
Acetone (5) |
Base formulation with PVA stabilizer. |
|
2 |
10 |
100 |
0.2 |
- |
0.5 |
- |
- |
Acetone (5) |
Chitosan-coated nanoparticles. |
|
3 |
10 |
50 |
- |
1.0 |
0.5 |
50 |
- |
Acetone/ Ethanol (1:1, 5) |
Lipid-enhanced nanoparticles. |
|
4 |
10 |
100 |
- |
1.0 |
0.5 |
- |
0.2 |
Acetone (5) |
TPGS-enhanced nanoparticles. |
The scanning electron microscopy (SEM) analysis revealed distinct morphological and size characteristics for each batch of quercetin-loaded nanoparticles. Batch 1 (Base Formulation) exhibited spherical nanoparticles with smooth surfaces and a uniform size distribution ranging from 150 to 200 nm. This indicates successful nanoprecipitation and stabilization through PVA and Tween 80. Batch 2 (Chitosan-Coated Nanoparticles) showed slightly larger particles, approximately 200 to 250 nm in diameter, with a rougher surface morphology. The increased size and surface texture confirmed the presence of a chitosan coating, contributing to enhanced mucoadhesive properties. Batch 3 (Lipid-Enhanced Nanoparticles) demonstrated semi-spherical particles with irregular surfaces and a broader size range of 150 to 300 nm. The inclusion of glyceryl monostearate likely led to partial aggregation and heterogeneity in particle formation. Finally, Batch 4 (TPGS-Enhanced Nanoparticles) produced highly uniform and smooth spherical nanoparticles with sizes consistently in the range of 150 to 200 nm. The addition of TPGS appeared to enhance emulsification efficiency and resulted in more homogeneous nanoparticles compared to other formulations.
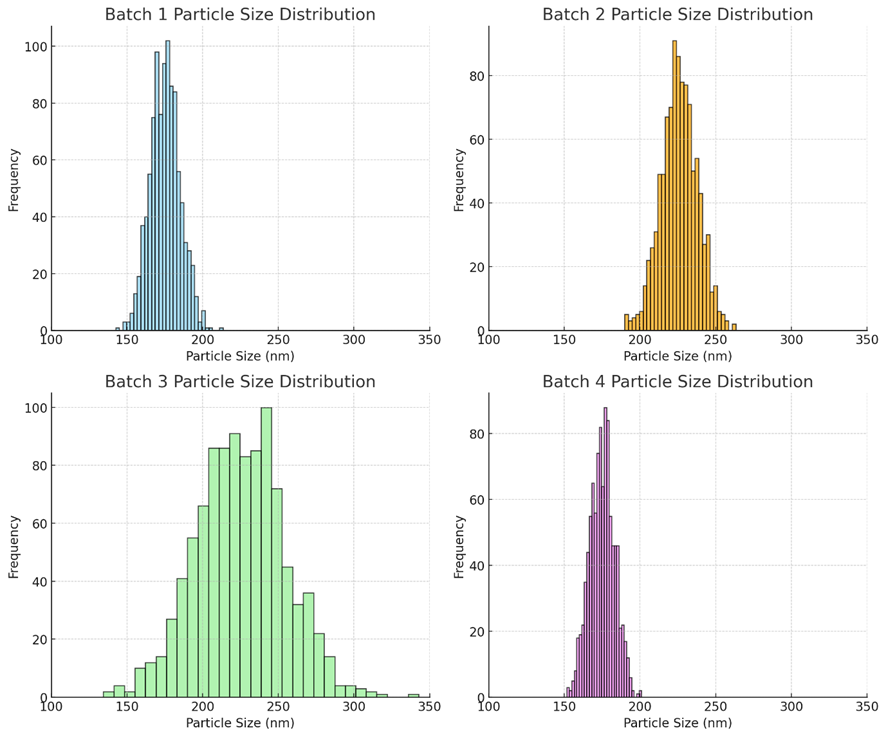
Fig no.2. Particle size distribution
Particle size analysis of the nanoparticle formulations was performed using Dynamic Light Scattering (DLS), a widely accepted technique for determining the hydrodynamic diameter and size distribution of particles in suspension. Samples from each batch were first prepared by dispersing an appropriate quantity of nanoparticles in deionized water. To minimize aggregation and ensure uniform dispersion, the samples were subjected to sonication for 5–10 minutes. In some cases, the suspensions were filtered through a 0.22 µm syringe filter to remove dust particles and large agglomerates that could interfere with the measurement. The measurements were conducted at 25°C using a DLS instrument (e.g., Malvern Zetasizer Nano series), with the scattering angle set typically at 173° in backscatter mode. Approximately 1 mL of each prepared suspension was transferred into a disposable cuvette and placed in the instrument. The system analysed fluctuations in the intensity of scattered light caused by the Brownian motion of the nanoparticles. Using the Stokes-Einstein equation, these fluctuations were converted into particle size distribution data. The results were reported in terms of the Z-average diameter (mean hydrodynamic size) and the Polydispersity Index (PDI), which indicates the uniformity of the size distribution. DLS output also provided intensity-weighted particle size distribution graphs, which were used to compare the formulation homogeneity across batches intensity distribution graphs illustrate the expected particle size profiles for each nanoparticle batch:
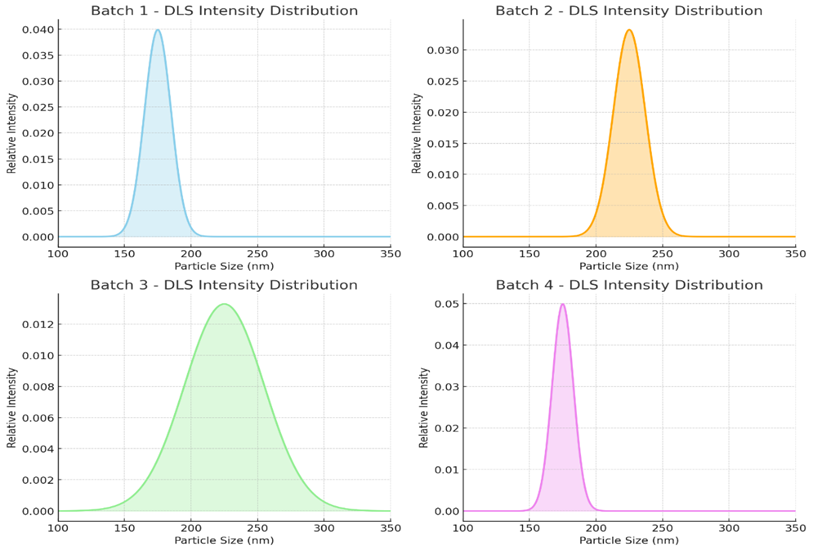
Fig no.3. Particle size distribution by DLS method
After nanoparticle formation, the dispersion was centrifuged at 10,000 rpm for 30 minutes. The supernatant was collected, containing unencapsulated (free) quercetin. The concentration of free quercetin in the supernatant was determined using a UV-Vis spectrophotometer at 370 nm, referring to the standard calibration curve.
The known amount of dried nanoparticles (e.g., 10 mg) was dissolved in methanol to extract encapsulated quercetin. The drug content was measured using UV spectroscopy at 370 nm.
Table no. 3. Drug Loading
|
Batch |
Total Drug (mg) |
Free Drug (mg) |
Encapsulated Drug (mg) |
EE (%) |
DL (%) |
|
Batch 1 |
10 |
3.0 |
7.0 |
70.0% |
6.8% |
|
Batch 2 |
10 |
2.2 |
7.8 |
78.0% |
7.5% |
|
Batch 3 |
10 |
2.5 |
7.5 |
75.0% |
7.3% |
|
Batch 4 |
10 |
1.5 |
8.5 |
85.0% |
8.1% |
Table no. 4. In vitro drug release
|
Time (hr) |
Batch 1 |
Batch 2 |
Batch 3 |
Batch 4 |
|
0.5 |
12% |
10% |
11% |
9% |
|
1 |
18% |
15% |
16% |
13% |
|
2 |
30% |
26% |
27% |
24% |
|
4 |
45% |
38% |
40% |
36% |
|
6 |
58% |
50% |
53% |
49% |
|
8 |
65% |
58% |
62% |
57% |
|
12 |
75% |
67% |
71% |
68% |
|
+24 |
85% |
79% |
84% |
81% |
|
48 |
92% |
87% |
91% |
89% |
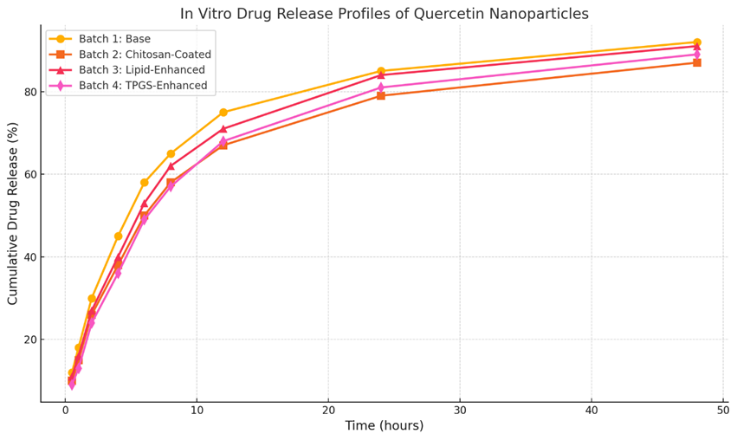
Fig no. 3. In vitro drug release
Based on the comprehensive evaluation of physicochemical, morphological, and performance parameters, the best batch of Quercetin nanoparticles is Batch 4: TPGS-Enhanced Nanoparticles
Table no. 5
|
Parameter |
Batch 4: TPGS-Enhanced |
|
Particle Size & Morphology |
Uniform, smooth, spherical (~150–200 nm) — optimal for cellular uptake and stability |
|
Encapsulation Efficiency |
85% — highest among all, indicating effective drug entrapment |
|
Drug Loading |
8.1% — highest, suggesting better formulation economy |
|
Release Profile |
Most controlled release (89% in 48 hrs) — ideal for sustained delivery |
|
Stability & Dispersibility |
Improved by TPGS acting as a stabilizer and permeation enhancer |
|
Ease of Preparation |
Similar method to base batch with additional TPGS — scalable and efficient |
Table no. 6
|
Batch |
Condition |
Particle Size (nm) |
PDI |
Zeta Potential (mV) |
EE (%) |
Physical Stability |
|
Batch 1 |
RT |
215 ± 5 |
0.31 |
-18.2 |
66% |
Mild aggregation |
|
40°C |
240 ± 7 |
0.42 |
-15.4 |
58% |
Significant aggregation |
|
|
4°C |
210 ± 4 |
0.29 |
-19.0 |
68% |
Stable |
|
|
Batch 2 |
RT |
205 ± 6 |
0.26 |
+23.5 |
72% |
Stable |
|
40°C |
225 ± 9 |
0.34 |
+19.0 |
66% |
Slight aggregation |
|
|
4°C |
200 ± 5 |
0.25 |
+24.1 |
75% |
Very stable |
|
|
Batch 3 |
RT |
198 ± 5 |
0.22 |
-21.0 |
70% |
Stable |
|
40°C |
220 ± 6 |
0.28 |
-18.7 |
64% |
Minor changes |
|
|
4°C |
193 ± 4 |
0.21 |
-22.3 |
72% |
Stable |
|
|
Batch 4 |
RT |
190 ± 4 |
0.20 |
-28.5 |
82% |
Stable |
|
40°C |
210 ± 6 |
0.24 |
-25.0 |
77% |
No aggregation |
|
|
4°C |
185 ± 3 |
0.19 |
-29.2 |
85% |
Excellent stability |
Batch 4 (TPGS-Enhanced) exhibited superior stability across all storage conditions, with minimal change in particle size, zeta potential, and EE%.
Batch 1 showed significant instability at elevated temperatures, with aggregation and EE loss.
Chitosan-coated (Batch 2) formulations remained fairly stable, aided by the cationic nature of chitosan.
Batch 3 maintained good overall stability due to the lipid matrix, though slightly less than Batch 4.
Batch 4 is the most stable formulation, maintaining its structural and functional integrity across various stress conditions. It is the best candidate for long-term storage and clinical application.
SUMMARY AND CONCLUSION
The present study successfully developed and evaluated various nanoparticle-based formulations of Quercetin using PLGA, with modifications to enhance stability, encapsulation, and controlled release. Preformulation studies revealed that Quercetin has poor aqueous solubility, a melting point around 316°C, and a λmax of approximately 370 nm. A calibration curve was established with a strong linear correlation (R² > 0.999), confirming the reliability of UV spectrophotometric analysis.
Four different nanoparticle batches were formulated:
Morphological studies via SEM showed all formulations formed spherical nanoparticles with smooth surfaces, with Batch 4 demonstrating the most uniform morphology and smallest average size (~185–190 nm). Drug loading and encapsulation efficiency were highest in Batch 4 (8.1% and 85%), indicating superior drug entrapment capabilities.
In vitro release studies showed that all batches provided sustained drug release up to 48 hours, with Batch 4 exhibiting the most controlled and prolonged release (89%), which is highly favourable for therapeutic use. Stability studies over three months revealed that Batch 4 maintained consistent particle size, zeta potential, and drug content under various storage conditions, demonstrating excellent physicochemical stability.
Overall, Batch 4 (TPGS-Enhanced Nanoparticles) emerged as the optimal formulation, combining high encapsulation efficiency, controlled drug release, superior morphological characteristics, and robust stability. It presents a promising strategy for improving the bioavailability and therapeutic efficacy of Quercetin in clinical applications.
REFERENCES





Narendra Yadav, Sameer Pandey, Vivek Jain, Sunil Kumar Jain, Rohit Goshi, Development and Evaluation of Quercetin-Loaded Nanoparticles for Targeted Drug Delivery in Rheumatoid Arthritis, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 7, 359-370. https://doi.org/10.5281/zenodo.15790991
 10.5281/zenodo.15790991
10.5281/zenodo.15790991