
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Smt. Tarawati Institute of Biomedical & Allied Sciences Roorkee, Uttarakhand.
In current scenarios the dermatological problems have emerged to be one of the major concern across the globe. Eczema, or atopic dermatitis is the most common types of prevalent dermatological conditions. Topical corticosteroids are majorly used for the testing eczema hut due to many adverse reactions there is a need for exploring an adjuvant therapy for treating eczema.(1) The extract of plant Portulaca Oleracea of family Portulacae had shown the potential towards eczema and other dermatological diseases. One of the more advanced methods of topical drug delivery is topical gel formulation. The manufacture of gels using the extract of the Portulaca Oleracea plant, which belongs to the Portulacae family. In this study formulation of gel with aqueous extract of Portulaca Oleraca L was done. The gel was prepared at various combinations of aqueous extract (5% & 10%) from Portulaca oleracea leaves were tested with the polymers of Carbopol 940. The formulated gel was then evaluated on various parameters. Firstly, the aqueous extract of Portulaca Oleraca L was evaluated for its organoleptic properties, pH determination was done, melting point was assessed. After formulation of gel evaluation of different parameters such as UV spectroscopy, FTIR analysis, viscosity and spreadebility were assessed. After the formulation preparation it was noticed that aqueous extract was of light greenish yellow to green solid colour, odorless, slightly fussy in taste with pH 6.13. Its Spreadability was found as 19.79 for 5 % aqueous concentration and 22.5 for 10% aqueous extract of Portulaca Oleraca L. The gel formulated was found stable for thermal stress test and no separation of phase were reported. Since Portulaca Oleraca L has shown potential anti-inflammatory, anti-analgesic and anti-microbial activities, this plant can be a future boom towards treating different dermatological diseases. Previous research had shown the effectiveness of extract of Portulaca Oleraca L towards different dermatological diseases also many animal models had also shown beneficial effects towards dermatological diseases like atopic dermatitis. Preparation of gels with Portulaca Oleraca L has shown good stability. This study thus opens up the doors for further research on plant Portulaca Oleraca L to assess different pharmacological activity and different formulation can be developed for further research.
Over the past few decades, a variety of illnesses have been treated by giving patients drugs orally, sublingually, rectally, parenterally, etc. The topical drug delivery methodology is often employed when other methods of drug administration are ineffective, or it is mainly used to treat localised skin conditions such psoriasis, acne, and fungal infections. (1) Applying a medication-containing formulation to the skin to treat a cutaneous ailment is known as topical drug delivery. The goal of pharmaceutical research nowadays is to give patients the treatments they require. Since most newly created active pharmaceutical compounds are hydrophobic by nature, a novel drug delivery method utilizing microemulsions has a positive influence on the efficient administration of hydrophobic medications. (2) The human skin is a specially designed organ that allows life on Earth by controlling body temperature and moisture loss while blocking the entry of pathogens and hazardous materials. (3) There are several drawbacks to topical medications such ointments, creams, and lotions. They often cause discomfort to the patient following application because they are so sticky. Moreover, they must be rubbed in when applied because to their decreased spreading coefficient. All of these factors, which also highlight the stability problem within the main category of semisolid preparations, have led to a growth in the usage of translucent gels in pharmaceutical and cosmetic preparations. (4) Topical gels are semisolid compositions that are clear or translucent and have a high ratio of gelling agent to solvent. The dispersed phase's interlacing three-dimensional networks of particles or solvated macromolecules limit the mobility of the dispersing medium in a semi-rigid structure called a gel. (5) They have a localized effect with fewer adverse effects, are less greasy, can be removed from the skin with ease, are less intrusive, require lower doses than oral dosage forms, enhance drug bioavailability, reduce dosing frequency, and stabilize drug delivery patterns. (6) Gels have many benefits, but hydrophobic drug delivery is a serious drawback. An technique based on microemulsions is being utilised to get over this restriction and allow even a hydrophobic therapeutic component to take advantage of the special qualities of gels. After the creation of an oil-in-water microemulsion, a gelling agent is applied. For the delivery of medications that are hydrophobic or insoluble in water, emulsified gel is a dependable and excellent option. Gels based on microemulsions are essentially a hybrid of the two. (7) When determining health priorities, skin conditions are occasionally viewed as minor players in the global sickness league in comparison to conditions like HIV/AIDS, community-acquired pneumonias, and tuberculosis that result in significant mortality. Nonetheless, skin conditions are typically among the most prevalent illnesses seen in tropical primary care settings, and in certain areas where transmissible infections like onchocerciasis or tinea imbricata are endemic, they even take centre stage in patient presentations. For example, skin illnesses were linked to 20,000 death rates in Sub-Saharan Africa in 2001, according to the World Health Organization's 2001 study (WHO 2005) on the worldwide burden of disease. This burden was equivalent to the death rates linked to rheumatic heart disease, meningitis, hepatitis B, and obstructed labour in the same region. (8)
It is noncontagious inflammation of the epidermis and dermis with characteristic clinical features (itch, erythema, papule, seropapule, vesicle, scale, squame, crust or lichenification that emerge simultaneously or evolve from one another) and distinct histological picture (spongiosis, acanthosis, parakeratosis, lymphocytic and granulocytic infiltrates. (9) The debate on the differences between the terms “eczema” and “dermatitis” has been ongoing for many decades, with no definite conclusion. Therefore, in the present article these terms will be considered as synonyms. The clinical spectrum of dermatitis/eczema diseases includes an array of diseases that sometimes are depicted as mutual opposites; however, their clinical features and Patho mechanisms overlap to an extent making any clearcut differentiation virtually impossible. (10) It is a non-contagious inflammation of the dermis and epidermis with distinct histological features (spongiosis, acanthosis, parakeratosis, lymphocytic and granulocytic infiltrates) and characteristic clinical features (itch, erythema, papule, seropapule, vesicle, scale, squame, crust, or lichenification that emerge simultaneously or evolve from one another). (11) The issue of whether "dermatitis" and "eczema" are interchangeable has been debated for many years without reaching a consensus. As a result, these terms will be used interchangeably throughout this text. The clinical spectrum of dermatitis/eczema illnesses encompasses a wide range of conditions that are occasionally portrayed as mutually exclusive; yet, there is much overlap between their clinical characteristics and path mechanisms, making definitive classification nearly impossible.
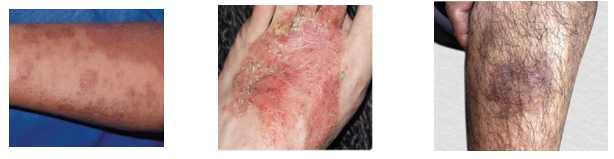
Fig 1: Atopic dermatitis (Eczema) in different part of body
It is a long-term inflammatory skin condition that usually starts in the first few months of life, has flare-ups and remissions throughout its duration, and is characterized by a particular distribution and form of skin lesions. Furthermore, one of the main symptoms of this illness is pruritus, which leads to insomnia. (17)
The common name for Portulaca Oleracea, "moss rose," appropriately represents the plant's major appealing traits, according to research from the Missouri Botanical Garden: Blooms that resemble a bed of mossy leaves, with ruffled petals up to 1" in diameter, and prostrate to gradually ascending stems. Typically, this pulp reaches a height of 6 to 8 inches and a spread of at least 12 inches. Flowers can be red, rose, orange, yellow, white, or pastel tones of any of these colours. They can also be single, semi-double, or double in shape. From June till the first frost, they are in flower. Flowers do not bloom during cloudy or damp days. Medium-green, cylindrical leaves form thick, cylindrical clusters up to one inch long along the reddish stalks. (18)

Fig 2: - Portulaca Oleraca L Plant
Plant Profile
Family: Portulacaceae
Type: Annual
Zone: Two to Eleven
Height: Between 0.25 and 0.75 feet
Spread: Between 0.50 and 1 foot
Sun: Direct sunshine
Water: Medium-to-dry
Low upkeep required
Recommendations for Use: Ground Cover, Naturalise, Annual
Blossom: Decorative
Suitability: Drought-prone, arid soil
Common Names
English: Portulaca Oleraca L
Assamese: Tit –bhekuri, Hati – bhekuri
Malayalam: Kandakarichunda
The herbal extract of plant Portulaca Oleracea L. has shown great anti-inflammatory activity and is found effective in atopic dermatitis and its topical preparation of gel can be effective and economical. It is accomplished by the degranulation of mast cells as well as the production of TNF, IL-1, and IL-6, among other pro-inflammatory cytokines.(19) When macrophages are activated, proinflammatory cytokines and chemokines are generated. Ramesh et al. (2014) looked at the antibacterial activity of P. Oleracea L. against a variety of microbes, including Aspergilus niger, Bacillus subtilis, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans.(20) That’s why the prime objective of this study is to formulate a topical gel preparation and evaluate the stability with herbal extract of plant Portulaca Oleraca L. (20)
MATERIALS AND METHODOLOGY:
List of chemicals used in present work?
Table 2: List of Chemicals Used
|
S.no |
Chemical |
Manufacture/supplier |
|
1 |
extract of Portulaca Oleraca L ? |
Herbal Garden, Roorkee, Haridwar |
|
2 |
Ethyl Oleate |
Central drug House, New Delhi |
|
3 |
Lauromacrogel 300 |
Central drug House, New Delhi |
|
4 |
Sorbittan monooleate (span 80) |
Central drug House, New Delhi |
|
5 |
Propylene glycol |
Central drug House, New Delhi |
|
6 |
Carbopol – 940 |
Central drug House, New Delhi |
|
7 |
Menthol 5% |
Central drug House, New Delhi |
|
8 |
Propylene glycol monocaprylate (Capryol 90) |
Central drug House, New Delhi |
List of equipment used in present work
Table 3: List of equipment used
|
S.no |
Equipment |
Supplier |
|
1 |
Digital balance |
Sansui Japan |
|
2 |
FTIR spectroscopy |
Perkin elmer spectrum twoTM |
|
3 |
UV- vis spectrophotometer |
Cary win UV, Agilent Technologies |
|
4 |
Sonicator |
Sonar, india |
|
5 |
Dissolution apparatus |
electrolab |
Preparations of Extract:
Pre-Formulation Studies:
Organoleptic features of aqueous extract of Portulaca Oleracea L were differentiated according to their odour, colour, and description.
The purity of the medicine is crucial in the creation of dosage forms. Any drug's effectiveness is based on how pure it is. The purity of medications may be verified using a variety of techniques. The UV spectroscopy is the most effective technique since it is affordable and yields accurate, repeatable findings. The maximum concentration of a medication can be found via UV spectroscopy. The drug's maximum dosage cannot be changed under any circumstances. When the compound's maximum value changes, it means the compound's original purity has been compromised. In our study we determined the λ max of Aqueous extract of Portulaca Oleraca L by using UV spectroscopy. UV spectra were obtained from a solution of Aqueous extract of Portulaca Oleraca L dissolved in methanol. Between 200 and 400 centimeters, the sample was scanned.
The Fourier Transform-Infrared (FT-IR) spectroscopy is widely used in the pharmaceutical business. It is commonly used to detect substances that are known and unknown. Additionally, it can be used to assess drug interactions. Various excipients are mixed with the active component to give the finished product the desired shape and appearance. Once the active ingredients and excipient have been mixed, a drug's interaction with its excipient may occasionally cause incompatibility. Medication incompatibility may have an impact on the formulation's potency. There could also be detrimental effects on the body. For the pharmaceutical companies, ascertaining whether the medication and excipient are compatible is therefore of utmost importance. We added 10 mg of KBr with the medication and excipients and mashed them up in a mortar to see if the drug and polymer used to form the f Aqueous extract of Portulaca Oleraca L gel would be compatible. An FTIR Spectrophotometer was used to measure a small sample of the triturated material that was placed on the sample holder and measured between 4000 and 400 cm-1. By comparing the spectra to the peaks found in the FTIR analysis of the pure drug sample, interactions between the drug and the excipient were examined.
It is necessary to understand the drug's solubility before preparing any dosage form. A specific solvent is required to dissolve the solid dosage form, which causes pharmacological effects in the body. Additionally, a drug's solubility affects how well it is absorbed when it is present in solid dose form. Due to the medication's reduced availability to receptors, a medicine that is sparingly soluble in a solvent will have a minimal therapeutic effect. Therefore, the solubility of a medication is crucial to its therapeutic benefits. A little amount of the sample was weighed out and put in a test tube, and then a known volume of the solvent was added to the tube to determine the sample's solubility. After each addition, the system is shaken violently and checked visually for any particles of undissolved solute.
The purity of a medicine can be determined by its melting point. Every medicine has a unique melting point under specific circumstances. If the drug's melting point changed, it implied that the drug's therapeutic response would also have changed. The melting point of active ingredients is altered when they are combined with foreign substances or adulterants. Drugs' melting points are also altered when they come into touch with moisture. The best way to determine a drug's purity is hence to look at its melting point. If a medicine is pure, the body will receive the full benefit of the desired therapeutic impact. Melting point equipment was used to measure the melting point of aqueous extract of Portulaca Oleracea L . A little quantity of medication was inserted in a capillary tube, sealed on one end, and then placed in a melting point device, and the melting point temperature was recorded.
Table 4: Showing the concentration of ingredients used in formulation
|
Sr. No. |
Ingredients |
Control |
PO1 |
PO 2 |
|
1. |
Carbopol 940 |
1gm |
1gm |
1gm |
|
2. |
Methyl paraben |
0.4 ml |
0.2ml |
0.2ml |
|
3. |
Propylene glycol 400 |
5ml |
5ml |
5ml |
|
4. |
Triethanolamine (Q.S) |
1.2ml |
1.2ml |
1.2ml |
|
5. |
Distilled Water |
Required volume |
Required volume |
Required volume |
|
6. |
P.O extract (5%) |
- |
5.0g |
- |
|
7. |
P.O extract (10%) |
- |
- |
10.0g |
Evaluation Parameters
B) Evaluation parameters of gel
Colour- The colour of the formulations were checked against black and white background.
Odour- The odour of the gels was checked by mixing a little amount of gel in water and by taking smell of it.
Consistency- Consistency of the formulations were checked by applying the gel on to the skin.
Homogeneity- All the formulations were tested for occurrence of any aggregate by visual inspection after the gels have been set in the container.
Greasiness- Greasiness were checked by applying the formulations on to the skin.
Phase separation- Phase separation was observed by visual inspection.
Brookfield viscometer is used to determine the viscosity of gel formulations at 10rpm for 3 minutes with spindle 6.
Spreadability is determined by apparatus suggested by Mutimer et al (1956) which is suitably modified in the laboratory and used for the study. It consists of a wooden block, which is provided by a pulley at one end. By this method, spreadability is measured on the basis of ‘Slip’ and ‘Drag’ characteristics of gels. A ground glass slide is fixed on this block. An excess of Gel (about 2 gm) under study is placed on this ground slide. The Gel was sandwiched between this slide and another glass slide having the dimension of fixed ground slide and provided with the hook. A 1 Kg weight was placed on the top of the two slides for 5 minutes to expel air and to provide a uniform film of the Gel between the slides. Excess of the Gel was scrapped off from the edges. The top plate was subjected to pull of 80 gms. With the help of string attached to the hook and the time (in seconds) required by the top slide to cover a distance of 7.5 cm be noted. A shorter interval indicates better Spreadability. Spreadability was calculated by using the formula.
S = M.L /T
Where,
S = Spreadability,
M = Weight tied to upper slide,
L = Length of glass slides
T = Time taken to separate the slides completely from each other.
RESULT: -
Evaluation of Plant Extract:
The aqueous extract of plant Portulaca Oleraca L was subjected to following observations.
Organoleptic Properties: -
Drug's organoleptic qualities, including colour, flavour, aroma, crystallinity, and pH, were examined. The results were reported in a table.
Table 5: Showing Organoleptic properties of Portulaca Oleraca L
|
Parameters |
Result |
|
Colour |
Light greenish yellow to green solid |
|
Odour |
Odourless |
|
Taste |
Tasteless fussy taste very little sour in taste |
|
pH |
6.13 |
Melting point-
Melting point of drug was determined by capillary method was found to be 162?. The observed value was identical to the reported value i.e. 162?. The observed melting point confirmed the crude powder extract of Portulaca Oleraca l.
Table 6: Depicting the melting point of crude extract of Portulaca Oleraca L
|
Drug |
Observed |
Reference |
|
Portulaca Oleraca L |
162±2? |
161-167? |
|
Portulaca Oleraca L |
162±2? |
|
|
Portulaca Oleraca L |
162±2? |
Calibration curve of Portulaca Oleraca L: -
With the use of a UV-Visible Spectrophotometer, the wavelength of maximum absorbance, or λmax, for plant extract of Portulaca Oleraca L in methanol was identified. A prepared solution that had a concentration of 10 µg/ml was scanned between 200 - 400 nm. The 300nm maximum was noted. For subsequent investigation of absorption for concentrations ranging from 2 to 20 g/ml, observed absorption maxima, or max 300 nm, were employed. The concentration range for the linear plot was 2 to 20 g/ml, and the correlation coefficient (r2) value was shown to be 0.9892.
Table 7: Absorbance of Portulaca Oleraca L at different concentrations
|
Sr. No |
Concentration (µg/ml) |
Absorbance |
|
1 |
2 |
0.054 |
|
2 |
4 |
0.208 |
|
3 |
6 |
0.336 |
|
4 |
8 |
0.51 |
|
5 |
10 |
0.65 |
|
6 |
12 |
0.72 |
|
7 |
14 |
0.89 |
|
8 |
16 |
0.98 |
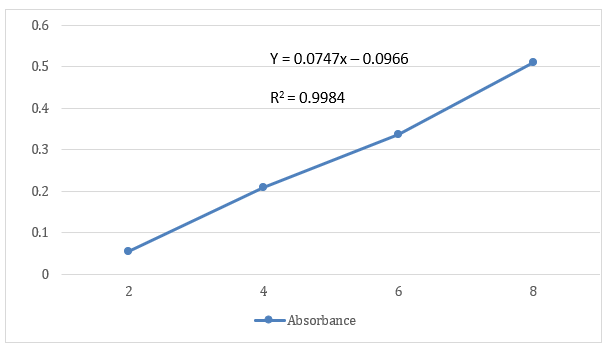
Fig 3: Calibration Curve of Aqueous extract of Portulaca Oleraca L.
The existence of many functional groups of minor and major phytoconstituents was shown by FT-IR analysis of aqua-methanol extracts of P. oleracea. This was achieved by producing spectrum profiles, with absorption bands ranging from 1019.17 to 38.24.44 cm-1 for both species. The broad peaks between 3360.20 and 3362.20 cm-1 represent the free hydroxyl (−OH: Ar−OH stretching) group of alcohols, water, organic acids, and phenols, as well as the amine and amide (N−H: stretched) group of proteins and water, respectively; the smaller peaks between 2979.65 and 2834.99 cm-1 represent alkanes, alkenes, and alkynes (C−C, C=C, and C≡C: symmetric and asymmetric) of fatty acids and lipids; the broad peak array between 1651.10 and 1104.41 cm-1 represents carbonyl (C−O) group of esters and ketones (−C=O), carboxylic acids (−COOH), aldehydes (−CHO), and aryl and ether hydrocarbons (C−O−C), di and tri-substituted aromatics due to C=C stretching, and lower strong peaks between 1019.17 and 1019.90 cm-1 signify aliphatic amines (C−N), phosphoryl groups (P=O), nitro compounds (−NO), and nucleic acids.
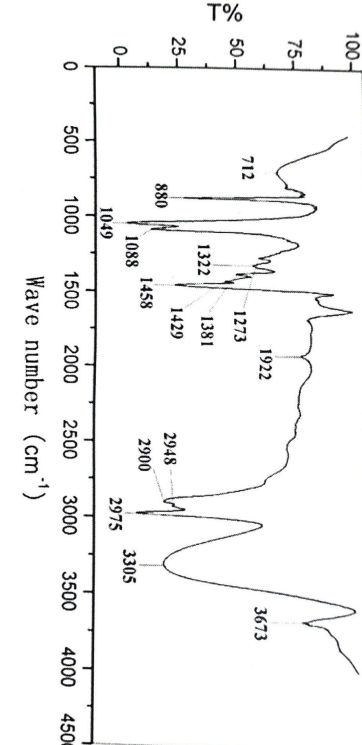
Fig 4: Depicting the FTIR of Aqueous extract of Portulaca Oleraca L
Table 8: Depicting the FTIR vibrations of aqueous extract of Portulaca Oleraca L
|
Functional Group |
Vibrational Mode |
PO 1 |
PO 2 |
|
Alcohols, phenols, organic acids, primary and secondary amides and amines |
−OH, N−H |
3362.20 |
3360.08 |
|
Saturated and unsaturated hydrocarbons |
C−C, C=C, C≡C |
2834.81-2979.65 |
2834.99-2980.13 |
|
α, β-unsaturated ketones, aldehydes, carboxylic acid, nitro compounds, aliphatic amines, esters, phosphoryls and nucleic acids |
C−O, −COOH, −C=O, C−N, P=O, −NO |
1104.41-1651.10 |
1104.36-1740.18 |
|
Aromatics, ethers and aryl hydrocarbons |
n(C−O−C), C=C |
1090.90 |
1019.17 |
Pre formulation Study
Gel formulations were created using the water titration method. Gels must meet the definition's requirements for clarity and transparency. All of the formulations showed excellent gel formation as they were all transparent, clear, and free of precipitation.
Globule size determination
Gels are translucent, thermodynamically stable systems with particles smaller than 270 nm. The estimation of globule size is a crucial evaluation metric since the therapeutic efficiency of a gels sizes ranging from 45.39–271.1 nm. The mean particle diameter was lowest for formulation ME1, which had the largest proportion of surfactant and co-surfactant at a fixed amount of oil, and highest for formulation F6, which had the highest proportion of surfactant and fixed amount of oil.
Freeze thawing test
If any formulations are identified, the test will examine their stability and phase separation.
Table 9: Showing result of Freeze Thawing
|
Formulation Code |
Freeze Thawing |
|
PO 1 |
Stable and no separation |
|
PO 2 |
Stable and no separation |
Determination of zeta potential:
The kind and makeup of the carrier utilised in the formulation affects the zeta potential value. F1 zeta potential was discovered to be -30.5 mV, indicating strong stability.
Determination of Viscosity
The viscosity of PO 1, PO 2 gel formulations was determined using spindle number 6 at room temperature and 20, 30, 50, 60, and 100 rpm. With the data acquired, the viscosity curve was plotted using the shear rate (rpm) on the x-axis and the viscosity on the y-axis.
Table 10: Depicting the viscosity of the prepared gel formulation
|
RPM |
Viscosity |
|
|
PO 1 |
PO 2 |
|
|
20 |
82±0.01 |
84 ±0.01 |
|
30 |
91 ± 0.07 |
87 ±0.01 |
|
50 |
84±0.02 |
89 ±0.02 |
|
60 |
81 ±0.03 |
79 ±0.03 |
|
100 |
95 ±0.02 |
96 ±0.01 |
Preparation of Gel formulation:
The gel formulations of Portulaca Oleraca L (PO 1 & PO 2) that contained carbopol looked to be clear, smooth, and transparent. The pH and drug content of these gel formulations incorporating drugs were also assessed.
Thermal stress test
The test was done as primary stability studies. Packaged products were under thermal stress in aluminum-coated tubes. The samples were placed at oven at 30°C ± 2°C and relative humidity of 60% ± 5% for 1 months. Gel formulations were evaluated at the times of 24 h, 1 week, 1 month.
Spreadebility Test
Table 11: Depicting the result of spreadebility test of prepared formulation
|
Formulation |
Spreadebility Viscosity |
|
PO 1 |
19.79 |
|
PO 2 |
22.5 |
DISCUSSION
The FTIR research's peaks in the physical combination demonstrated the compatibility of the drug with the gelling agents. The globe size study's results showed that as the oil phase ratio rose, a decrease in the surfactant/co-surfactant ratio caused an increase in particle size. A drop in the oil ratio and an increase in the surfactant/co-surfactant ratio lead to a decrease in mean particle size. The dilution test findings showed that gels retained their clarity even after being diluted with water, indicating their good miscibility with water, which was used as the continuous phase. After determining that the F1, F3, and F4 formulations were all stable liquids by centrifugation testing, the pH, medication concentration, and zeta potential were evaluated. The freeze-thawing experiments provided additional evidence that all three formulations were stable and that there was no phase separation. The high zeta potential of -30.5 mV in Formulation F1, which indicates great stability, led to its selection as the optimal formulation. The produced gel's pH was within the range that is safe for skin. At room temperature, the viscosity of the PO 1 and PO 2 gel formulations was measured using spindle number 92. At 20, 30, 50, 60, and 100 rpm, the results showed that the formulations were highly stable, with a range of 84 to 96. The samples were kept in an oven for one months at 30°C ± 2°C and 60% ± 5% relative humidity to test their thermal stability. After being tested for 24 hours, a week and a month gel composition were ultimately determined to be extremely stable. According to the viscosity investigation, all three formulations showed pseudoplastic non-Newtonian flow, and the viscosity values decreased as the shear rates increased. This might be as a result of the small particles in the gel that spontaneously generated, enabling a faster release of the drug. therefore, increasing the permeability of the solution. Portulaca oleracea L's gel formulation is said to improve skin absorption.
REFERENCES


Prachi Rajput*, Sunita Rani, From Weed to Wonder Herb: Purslane (Portulaca oleracea L) in Traditional and Modern Medicine, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 6, 3641-3654. https://doi.org/10.5281/zenodo.15722763
 10.5281/zenodo.15722763
10.5281/zenodo.15722763