
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmaceutical Science, SAGE University Indore.
Herbal medicines have been widely acknowledged for their therapeutic value in treating numerous diseases. Metabolic disorders, which develop gradually but disrupt various bodily functions, pose a significant health challenge. Herbal remedies act synergistically on multiple biochemical pathways, providing a holistic and natural approach to managing such disorders. Among these, thyroid dysfunction is one of the most common endocrine disorders worldwide and remains a major public health concern. Although herbal treatments have been used for centuries in traditional healing practices, their acceptance in modern clinical settings is still limited due to insufficient scientific validation and standardized evidence of effectiveness. However, growing concerns about the adverse effects and high costs of synthetic drugs have renewed global interest in exploring herbal alternatives. This review aims to highlight the potential of herbal medicines in the treatment of thyroid and related metabolic disorders. It focuses on five specific herbs known for their ability to regulate thyroid hormone levels and enhance glandular function through antioxidant activity, modulation of hormone synthesis, and improvement of metabolic balance. Integrating traditional herbal wisdom with scientific research can pave the way for safer, more effective, and affordable therapies for thyroid-related diseases.
In the current era, considerable emphasis is being placed on expanding the frontiers of medicine through the study of phytoconstituents and secondary metabolites. In the recent past, particularly about a decade ago, the application of herbal drugs and phytomedicines was largely disregarded, as allopathic medicine-maintained dominance within the global healthcare system. During that time, Western medical practice relied extensively on synthetic pharmaceuticals, often underestimating the therapeutic significance and potential of naturally derived plant compounds.1 Over the past few years, herbal medicines have regained prominence in the treatment of several metabolic disorders, including diabetes and hormonal imbalances. The objective of the present review is to elucidate the therapeutic relevance of herbal drugs in the management of the gradually progressing endocrine disorder, thyroid dysfunction. In India, diabetes mellitus and thyroid disorders represent two of the most prevalent endemic metabolic diseases, with a remarkable rise in their incidence over the past few decades. Both conditions are closely associated with metabolic dysregulation primarily resulting from improper lifestyle habits and altered physiological homeostasis.2,3 The endocrine system constitutes an intricate network of glands that regulate a wide spectrum of vital physiological activities in the human body, ranging from cardiovascular rhythm to reproductive function. It encompasses ten principal glands, including the thyroid, adrenal, pituitary, and parathyroid glands, along with the hypothalamus, pancreatic islet cells, thymus, pineal gland, ovaries, and testes. Even minor alterations within this system can disturb its finely tuned hormonal equilibrium, giving rise to diverse endocrine pathologies. These disorders are typically classified according to several criteria: (i) abnormalities in hormone secretion, whether hypersecretion or hyposecretion; (ii) the formation of lesions or neoplasms within endocrine tissues that may or may not alter hormonal output; (iii) infectious processes impairing glandular activity; and (iv) the inability of one endocrine gland to adequately stimulate another. Under normal physiological circumstances, hormonal homeostasis is maintained through precise feedback control mechanisms; however, any disruption in these regulatory pathways can compromise endocrine balance, ultimately leading to disease manifestations contingent upon the specific gland involved. The thyroid gland is an essential component of the endocrine system, critically involved in the regulation of multiple physiological processes, including oxygen consumption, growth, development, and cellular metabolism. Anatomically positioned in the anterior region of the neck, the gland synthesizes and secretes the hormones thyroxine (T4) and triiodothyronine (T3), which are transported via the bloodstream to modulate basal metabolic rate, somatic growth, and overall physiological development. Thyroid disorders present with a wide spectrum of clinical manifestations, such as fatigue, depression, constipation, abnormal body weight changes, cold intolerance, bradycardia or tachycardia, tremors, diarrhoea, irritability, anxiety, and sleep disturbances. Thyroid dysfunction constitutes one of the most common endocrine disorders worldwide, with an estimated 42 million cases reported in India alone, underscoring its significant public health impact.4 The therapeutic management of thyroid disorders generally encompasses hormone replacement therapy, iodine supplementation, surgical procedures, and/or administration of antithyroid agents, tailored according to the specific disorder type and its clinical severity. Nevertheless, these conventional treatment strategies are frequently accompanied by adverse effects, such as myopathy, anorexia, alopecia, and additional systemic complications.5 Certain drugs can alter thyroid function or worsen hypo- and hyperthyroid symptoms, emphasizing the need for a structured patient management approach. Recently, alternative medicinal strategies, particularly herbal therapies, have gained attention for their efficacy and minimal side effects. This chapter reviews thyroid dysfunctions and explores the role and mechanisms of herbal medicines in managing these disorders effectively.
Thyroid Disorders
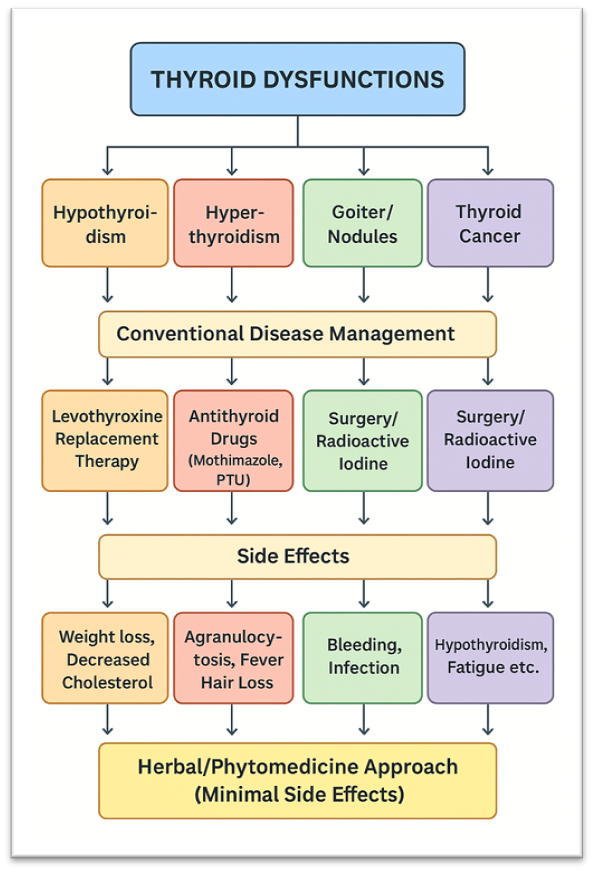
Hypothyroidism: Hypothyroidism, commonly referred to as underactive thyroid disease, is defined by inadequate secretion of thyroid hormones, triiodothyronine (T3) and thyroxine (T4), from the thyroid gland. These hormones are critical regulators of basal metabolism, modulating cardiovascular function and thermoregulation. Deficiency of T3 and T4 results in a generalized metabolic slowdown and can further impair thyroid hormone synthesis. Clinically, hypothyroidism presents with a spectrum of symptoms, including fatigue, depression, anxiety, dry skin, brittle hair, facial edema, periorbital puffiness, and heightened sensitivity to cold temperatures. The conventional management involves the daily administration of levothyroxine to normalize circulating thyroid hormone levels. Nonetheless, prolonged therapy may lead to alterations in lipid metabolism, including decreased cholesterol levels, as well as unintended weight reduction.
Hyperthyroidism: Hyperthyroidism, also referred to as overactive thyroid disease, is often used interchangeably with thyrotoxicosis, a condition resulting from excessive thyroid hormone levels. This disorder is less prevalent than hypothyroidism and frequently arises secondary to conditions such as Graves’ disease, toxic multinodular goiter, or toxic adenomas. Clinically, hyperthyroidism is characterized by a constellation of symptoms, including exophthalmia, irritability, fatigue, elevated basal metabolic rate (BMR), hyperactivity, dysphoria, tachycardia, unintentional weight loss, increased appetite, diarrhea, polyuria, and tremors.6 The management of hyperthyroidism is primarily symptomatic and involves antithyroid agents (such as methimazole and propylthiouracil), beta-adrenergic blockers (including propranolol, atenolol, and metoprolol) for rapid relief of symptoms, radioactive iodine therapy, and surgical intervention. Antithyroid medications inhibit the biosynthesis of thyroid hormones but may be associated with adverse effects, including agranulocytosis, fever, pharyngitis, and increased susceptibility to infections. Long-term administration of these drugs can achieve disease remission, although surgical intervention may subsequently be required. Beta-adrenergic blockers ameliorate hyperthyroid manifestations by counteracting the metabolic effects of excess thyroid hormones; however, they do not decrease circulating hormone concentrations and therefore do not address the underlying pathophysiology of hyperthyroidism, which can vary depending on the etiology.
Goiter: Goiter is defined as an abnormal, usually painless enlargement of the thyroid gland, which may result in respiratory and swallowing difficulties. This condition frequently arises due to dietary iodine deficiency; however, not all cases exhibit clinical manifestations such as throat tightness, anterior neck swelling, cough, hoarseness, or dysphagia. Goiter can be transient and resolve spontaneously without intervention, or it may signify an underlying chronic thyroid disorder necessitating medical evaluation. Thyroid enlargement is often associated with various thyroid pathologies, where increased activation of thyroid-stimulating hormone (TSH) receptors enhances thyroid hormone synthesis, leading to increased vascularization and hypertrophy of the gland.7 Goiter is not a distinct medical condition but often serves as an indicator of underlying disorders. It may result from a variety of causes, including autoimmune diseases, hypothyroidism, hyperthyroidism, Hashimoto’s thyroiditis, cyanide exposure, Graves’ disease, ingestion of goitrogenic substances, thyroid hormone resistance, adverse drug reactions, thyroid neoplasms, acromegaly, and thyroiditis, among others.
Thyroid nodules (benign) and cancer (malignant): Thyroid nodules, either benign or malignant, include colloid nodules, cysts, and nodular thyroiditis, affecting approximately 5% of the population. Most nodules are asymptomatic, with only ~1% being cancerous, though some may cause palpable neck masses, dyspnea, or dysphagia. Benign nodules are typically monitored through thyroid function tests, ultrasonography, and physical examination. While many remain stable and require no treatment, enlarging nodules may necessitate surgical removal, radioactive therapy, or thyroid hormone administration. The National Cancer Institute (USA) reports that approximately 56,000 new cases of thyroid cancer are diagnosed annually, with females demonstrating a threefold higher incidence than males. Thyroid malignancies are generally more aggressive in older populations, with a lifetime prevalence of approximately 1.2%. The disease is often asymptomatic but may occasionally present as a palpable cervical mass or, less frequently, with voice changes such as hoarseness. Although thyroid nodules are detected in roughly 75% of the population, the majority are benign. The risk of malignant transformation is comparatively higher in children and adolescents than in adults. By the age of 80, nearly 90% of individuals are estimated to develop at least one thyroid nodule.8,9 Thyroid cancer is divided into four main types: papillary or mixed papillary/follicular (≈85%), follicular or Hürthle cell (≈10%), medullary (≈3%), and anaplastic (≈1%). High-resolution ultrasonography is used to assess the nature of thyroid nodules, guiding partial or total thyroidectomy. Medullary cases require regional lymph node removal, while anaplastic cases may necessitate tracheostomy. Post-surgery, patients are managed with individualized radioactive iodine therapy and lifelong levothyroxine supplementation.10
Hashimoto’s thyroiditis: Hashimoto’s thyroiditis, or chronic lymphocytic autoimmune thyroiditis, is the leading cause of hypothyroidism in the United States. It is an autoimmune disorder in which anti-thyroid antibodies target and damage thyroid tissue, resulting in inflammation and progressive hypothyroidism. Clinical manifestations include fatigue, weight gain, joint stiffness, constipation, cold intolerance, facial and periorbital edema, hair thinning, menstrual irregularities, bradycardia, and cognitive impairment. The disease predominantly affects females aged 30–50 and is more common in individuals with other autoimmune conditions such as type 1 diabetes, rheumatoid arthritis, lupus, Addison’s disease, vitamin B12 deficiency, or gluten sensitivity. While not all hypothyroidism cases are caused by Hashimoto’s thyroiditis, levothyroxine therapy effectively restores normal thyroid function in affected patients.11 In summary, thyroid dysfunctions are prevalent conditions that significantly impact human health, and current therapeutic options are often limited by various adverse effects and constraints. These challenges underscore the need for alternative treatment strategies. Herbal medicines, or phytomedicines, offer promising therapeutic potential due to their efficacy and minimal side effects. Consequently, greater emphasis should be placed on the translational research and clinical application of herbal formulations, bridging bench-side findings to bedside therapies for the management of thyroid disorders.
Fig.1. A representative diagram depicting various thyroid disorders, standard treatment methods with their associated side effects, and a comparison with herbal medicine-based approaches.
Role of Herbs in Thyroid
Medicinal plants have been employed since ancient times for their pharmacological properties. Bioactive phytochemicals present in these plants exert physiological effects on the human body, and herbal formulations are typically associated with a lower incidence of adverse effects. Additionally, numerous culinary herbs and spices serve as sources of bioactive compounds with therapeutic potential.12 In recent years, there has been significant interest in medicinal plants, particularly those utilized in traditional systems such as Ayurveda, Siddha, Unani, Modern Arñchi, Homeopathy, and Naturopathy. Phytopharmaceuticals derived from plants are considered relatively safe and demonstrate notable efficacy in managing various diseases. Moreover, traditional folk medicine continues to play a significant role in human health and in shaping interactions with the environment.
Mechanism of Action
The standard treatment for hypothyroidism predominantly involves levothyroxine replacement therapy. Nonetheless, long-term administration of this therapy has been linked to neurocognitive deficits, diminished quality of life, and suboptimal patient adherence. Phytotherapeutic interventions and their bioactive compounds have emerged as viable alternatives for hypothyroidism management. In this regard, Costus pictus (family Costaceae), commonly referred to as the insulin plant due to its antidiabetic activity, has shown potential efficacy in hypothyroidism. Extracts of this plant have been reported to normalize thyroid hormone levels and enhance overall thyroid function.13 For example, seaweeds are frequently utilized in the management of thyroid disorders owing to their high iodine content. Fucus vesiculosus (bladderwrack, a brown seaweed) has demonstrated thyroid-stimulating activity; however, there is a lack of established evidence regarding its safety, therapeutic efficacy, and clinical dosing parameters.14 Fucus vesiculosus and sea kelp (Ascophyllum nodosum) are recognized as rich natural sources of iodine; however, excessive consumption may lead to hyperthyroidism.
The administration of withania (1.4 g/kg body weight) in combination with Bauhinia purpurea (2.5 mg/kg body weight) markedly improved thyroid function and alleviated diabetes-induced hypothyroidism.15 Triphladya Guggulu exerts direct effects on the thyroid gland independent of the pituitary–TSH axis. Commiphora mukul (Guggul) exhibits thyroid-stimulating activity and aids in reducing LDL levels in hypothyroid conditions. Administration of Guggulu (200 mg/kg/day) for 30 days enhanced thyroid function and demonstrated antioxidant and antiperoxidative effects by upregulating the activity of superoxide dismutase (SOD) and catalase (CAT), thereby contributing to the amelioration of hypothyroidism.16
Medicinal Herbs with Therapeutic Potential for Thyroid Disorders

Fig.2. Aegle marmelos L. Correa
It has been used since ancient times in the preparation of various Ayurvedic medicines.18

Fig. 3. Aloe barbadensis
It is well recognized for its diverse therapeutic properties such as protection against UV and gamma radiation-induced skin damage, anti-inflammatory activity, immune-modulatory effects, as well as moisturizing and anti-aging benefits.20

Fig.4. Avena sativa L.

Fig.5. Bacopa monnieri L. Pennell

Fig.6. Fucus vesiculosus
Such as exhibiting anti-inflammatory properties, supporting thyroid function, promoting skin health, and aiding in the regulation of menstrual cycle patterns.24

Fig.7. Commiphora wightii
It has been traditionally employed in the management of various ailments including inflammation, gout, rheumatism, obesity, and lipid metabolism disorders. The resin comprises a complex mixture of phytoconstituents, including volatile oils with terpenoidal components such as monoterpenoids, sesquiterpenoids, diterpenoids, and triterpenoids; along with steroids, flavonoids, guggultetrols, lignans, sugars, and amino acids, all contributing to its diverse pharmacological activities.26

Fig.8. Linum usitatissimum L.
Flaxseed is an abundant source of dietary fiber, constituting approximately 40% of its composition, with soluble fiber making up 25% and insoluble fiber 75%. The soluble fraction, which includes gums, pectin, and β-glucan, plays a significant role in lowering blood glucose levels and absorbing cholesterol and triglycerides, thereby contributing to the prevention of cardiovascular diseases and diabetes.28

Fig.9. Pistia stratiotes L.

Fig.10. Withania somnifera L
The principal bioactive constituent of Withania somnifera is Withaferin, which pharmacologically enhances thyroid function, promotes antiperoxidative activity, and stimulates the production of T4 hormone.31

Fig. 11. Zingiber officinale (Rpsc.)

Fig.12. Bugleweed (Lycopus virginicus)

Fig.13. Lemon balm (Melissa Officinalis)

Fig.14. Motherwort (Leonurus cardiac)

Fig. 15. Gromwell (Lithospermum Ruderale)

Fig.16. Rose Marry (Rosmarinus officinalis)

Fig.17. Sage (Salvia officinalis)

Fig.18. Gotu Kola ( Centella asiatica )

Fig. 19. Coleus or forskohlii ( Plectranthus barbatus)
Dosage
The dosage of phytotherapeutic formulations can exhibit significant inter-individual variability, complicating the establishment of uniform dosing guidelines. Administration often relies on conventional volumetric measures such as tablespoons, teaspoons, hand palm, little finger, cups, or glasses. For decoctions and infusions, a standard glass cup is typically employed, and honey (approximately one tablespoon) may be incorporated as a palatability enhancer, whereas sucrose or other sugars should be excluded. Certain preparations are recommended to be formulated 1–2 hours prior to administration, while others may require preparation and storage for up to one week to achieve optimal efficacy. Clinically, a typical regimen involves 1–3 teaspoons per dose, administered 1–3 times daily, depending on the specific phytoconstituents, formulation type, and patient-specific pharmacodynamic responses.39
CONCLUSION
Hypothyroidism is linked to multiple cardiovascular and metabolic comorbidities, including hypertension, dyslipidemia, and systemic dysfunction, reflecting the critical regulatory role of thyroid hormones in physiological and pathophysiological processes. Standard treatment predominantly involves levothyroxine replacement therapy, which, while effective in restoring hormone levels, may contribute to neurocognitive deficits, diminished quality of life, and suboptimal long-term adherence. Phytotherapeutic interventions have demonstrated efficacy in modulating thyroid function. Selected medicinal herbs can stimulate thyroid hormone biosynthesis, decrease serum TSH concentrations, augment T4 production, and enhance peripheral T4-to-T3 conversion, thereby improving endocrine homeostasis. Additional pharmacological benefits include anti-inflammatory effects, glycemic regulation, modulation of lipid profiles, maintenance of dermal health, and normalization of menstrual cyclicity. Specific bioactive constituents, such as sesquiterpenoids in Zingiber officinale (ginger), act as antigens on thyroid follicular cells, while withaferin A in Withania somnifera (Ashwagandha) enhances thyroid activity, exhibits antiperoxidative properties, and stimulates T4 synthesis. Dosage regimens of herbal preparations are individualized according to disease severity, patient-specific parameters, and clinical context. Administration frequently relies on standardized volumetric measures, including tablespoons, teaspoons, hand palm, little finger, cups, or glasses, as guided by qualified professionals. Commiphora mukul (Guggul) demonstrates thyroid-stimulating activity and contributes to the reduction of LDL cholesterol in hypothyroid subjects.
Several marketed herbal formulations combine multiple phytoconstituents to enhance therapeutic efficacy:
In summary, medicinal herbs offer a natural, efficacious, and adjunctive approach to thyroid health management, either as monotherapy or in combination with conventional pharmacotherapy, underscoring the importance of evidence-based guidance, standardized dosing, and professional supervision.
REFERENCES


Swapnil R. Dudhakohar*, Dr. Khushboo Arora, Herbal Remedies in Traditional Medicine for Thyroid Disorders, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 10, 2734-2749 https://doi.org/10.5281/zenodo.17442806
 10.5281/zenodo.17442806
10.5281/zenodo.17442806