
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
ITM SLS Baroda University.
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disorder characterized by persistent synovitis, progressive cartilage degradation, and bone erosion, resulting in irreversible joint damage and multisystem involvement. Its pathogenesis involves a complex interplay of genetic susceptibility, environmental triggers, and immune dysregulation, resulting in the production of autoantibodies such as rheumatoid factor and anti-citrullinated protein antibodies, along with elevated pro-inflammatory cytokines including TNF-? and IL-6. NSAIDs, glucocorticoids, and disease-modifying anti-rheumatic drugs (DMARDs) are some of the current treatments that help with symptoms and control the disease, but they have side effects and can be toxic in the long term. Phytotherapeutic agents like Curcuma longa, Boswellia serrata, and Withania somnifera have strong anti-inflammatory and immunomodulatory effects by changing the way cytokines signal and the way oxidative stress pathways work. More clinical validation is needed
The crippling autoimmune disease known as rheumatoid arthritis (RA) is characterised by persistent inflammation of the synovial joints, which causes pain, swelling, and stiff joint deformities. It can also harm extra-articular organs such as the heart, lung, digestive system, eye, skin, and nervous system. Rheumatoid arthritis (RA) is a chronic inflammatory, systemic, progressive autoimmune disease in which the body's immune system, which is primarily responsible for defending against foreign bacteria and viruses, unintentionally targets the joints, causing thickened synovium, the formation of pannus, and the destruction of bone and cartilage (1). If left untreated, it can lead to a number of complications, including rheumatoid vasculitis, Elty's syndrome, permanent joint damage that necessitates an arthroplasty, and splenectomy. The precise cause of this illness is still unknown to scientists. Age, gender, genetics, and environmental exposure—such as cigarette smoking, air pollution, and occupational exposure—are thought to contribute to the development of RA. Since there is no known cure for RA, the main objectives of treatment are to lessen pain and prevent or slow further damage. In the developed world, RA affects 0.5 to 1% of adults, with 5 to 50 new cases per 100,000 people annually. The immune system typically exhibits self-tolerance in autoimmune diseases and does not target the body's own constituents. An autoimmune disorder results from HL's self-tolerance breaking down. Articular cartilage and juxta-articular bone can be destroyed by chronic inflammation of the synovial membrane, which is part of the pathophysiology of RA. Arthritis is a very common health issue that causes functional disability related to activities of daily living (ADLs) and instrumental activities of daily living (IADLs), limits social and occupational participation, and makes it difficult for people to fulfil their roles in life. Rheumatoid arthritis, osteoarthritis, gout, tuberculosis, and other infections are among the more than 200 conditions that can result in arthritis (2). Ankylosing spondylitis, gout, osteoarthritis, psoriatic arthritis, rheumatoid arthritis, septic arthritis, calcium pyrophosphate deposition disease, crystal arthritis, bacterial infections, systemic lupus erythematosus (SLE), fibromyalgia, lupus, scleroderma, post-traumatic arthritis, hemochromatosis arthritis, enteropathy arthritis, giant cell arthritis, Bechet’s disease, relapsing polychondritis, gonococcal arthritis, Pseudo-gout, and Relapsing polychondritis are the most common types of arthritis (3)
(4)
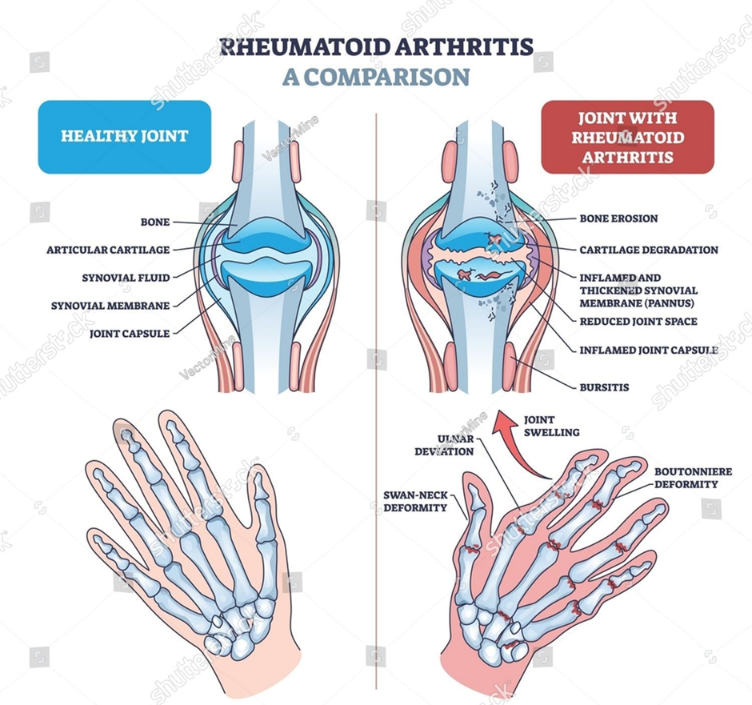
ETIOLOGY:-
The etiology of rheumatoid arthritis (RA), a chronic autoimmune disease, is multifactorial and involves a complex interplay of immune system dysregulation, environmental triggers, and genetic susceptibility. Genetic factors are important, especially differences in the HLA-DRB1 gene (shared epitope alleles), which make joint tissues more vulnerable to autoimmune reactions. In people who are genetically predisposed to the disease, environmental factors like cigarette smoking, infections, and exposure to specific pollutants can cause aberrant immune activation and the production of autoantibodies like anti-citrullinated protein antibodies (ACPAs) and rheumatoid factor (RF). These antibodies cause synovial hyperplasia, cartilage degradation, and bone erosion by starting a chronic inflammatory reaction in the synovial membrane of joints. It has also been proposed that changes in the gut microbiome and hormonal effects play a role in the development and progression of disease. Current research suggests that RA develops through the interaction of genetic predisposition with environmental and immunological mechanisms, which ultimately result in chronic joint inflammation and systemic manifestations, even though the precise cause is still unknown. (5,6,7).
PATHOPHYSIOLOGY:-
The chronic autoimmune disease known as rheumatoid arthritis (RA) is caused by a complex interplay between immune system dysregulation, environmental triggers, and genetic susceptibility. The human leukocyte antigen (HLA) system and other loci that affect immune signalling and lymphocyte activation, such as CD28, CD40, and PTPN22, are genetically strongly linked to the disease. Histone alterations and DNA methylation are examples of epigenetic modifications that further contribute to the development of disease. Through mechanisms like molecular mimicry, environmental factors like smoking, infections, and socioeconomic conditions act as triggers, causing the immune system to mistakenly attack self-antigens.
The development of autoantibodies, specifically rheumatoid factor (RF) and anti-citrullinated protein antibodies (ACPAs), which can be identified years before clinical symptoms manifest, is the defining feature of RA pathophysiology. These antibodies create immune complexes that trigger tissue damage by activating the complement system and inflammatory cells like macrophages and osteoclasts. Chronic inflammation of the synovial membrane caused by both innate and adaptive immune responses is the hallmark of the illness. Leukocyte infiltration and pro-inflammatory cytokines, particularly tumour necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and, to a lesser extent, interleukin-1 (IL-1), are involved in this inflammation.
By encouraging the expression of receptor activator of nuclear factor kappa-B ligand (RANKL), which stimulates osteoclast formation and bone resorption, synovial fibroblasts become activated as the inflammatory process advances and contribute to joint destruction. Concurrently, chondrocytes are affected by cytokines, which causes the release of matrix metalloproteinases that break down cartilage. Joint deterioration, deformity, and loss of function follow from this. Furthermore, although the precise mechanisms are still unknown, changes in the microbiome have been noted in RA patients and may contribute to the development and course of the disease. In general, the pathophysiology of RA involves an ongoing cycle of inflammation and autoimmunity that, if left untreated, eventually results in irreversible joint destruction. (8)
TYPES OF RHEUMATOID ARTHRITIS:-
Seronegative Patients who exhibit clinical symptoms consistent with rheumatoid arthritis but do not exhibit detectable RF or anti-CCP antibodies in laboratory tests are referred to as having seronegative RA. In contrast to seropositive RA, the disease course is frequently milder and less damaging. Clinical symptoms, imaging results, and inflammatory markers like ESR and CRP are the primary factors used in diagnosis. As antibodies start to show up over time, some seronegative patients may eventually develop seropositive RA. Because conditions like psoriatic arthritis or spondyloarthritis can initially mimic seronegative RA, differential diagnosis is crucial. (9,10,11)
MECHANISM OF RHEMATOID ARTHRITIS:-
SYMPTOMS OF RHEUMATOID ARTHRITIS:-
|
Category |
Symptom |
Description |
Citation |
|
Articular |
Joint pain (Arthralgia) |
Persistent pain in multiple joints, commonly small joints (hands, wrists, feet) |
[13] |
|
|
Joint swelling |
Synovial inflammation leading to soft tissue swelling |
[13] |
|
|
Morning stiffness |
Stiffness lasting >30–60 minutes, especially in early morning |
[14] |
|
|
Reduced range of motion |
Limited joint mobility due to inflammation and pain |
[14] |
|
|
Symmetrical joint involvement |
Bilateral involvement of MCP, PIP, wrist, knee joints |
[14] |
|
Systemic |
Fatigue |
Most common systemic complaint due to chronic inflammation |
[15] |
|
|
Low-grade fever |
Mild fever associated with inflammatory activity |
[15] |
|
|
Malaise |
General feeling of discomfort and illness |
[15] |
|
Extra-articular |
Rheumatoid nodules |
Firm subcutaneous nodules on extensor surfaces |
[16] |
|
|
Dry eyes (Keratoconjunctivitis sicca) |
Autoimmune involvement of lacrimal glands |
[16] |
|
|
Dry mouth (Xerostomia) |
Salivary gland involvement |
[16] |
|
|
Pulmonary involvement |
Interstitial lung disease, shortness of breath |
[16] |
|
|
Neurological symptoms |
Nerve compression causing numbness/tingling |
[16] |
|
Early/Preclinical |
Arthralgia (without swelling) |
Early symptom before clinical RA develops |
[17] |
|
|
Palindromic rheumatism |
Intermittent episodes of joint inflammation |
[17] |
|
General Clinical Feature |
Chronic progressive inflammatory disorder |
RA is a systemic autoimmune disease affecting synovial joints with progressive damage |
[18] |

(19)
DIAGNOSIS:-
COMPLICATION OF RHEUMATOID ARTHRITIS:-
|
SERIAL N.O. |
SYSTEM/CATEGORY |
COMPLICATION |
DESCRIPTION |
|
|
|
ARTICULAR |
Joint destruction & deformity |
Chronic synovial inflammation → cartilage erosion, bone destruction, deformities (e.g., ulnar deviation) |
|
|
Functional disability
|
||||
|
Progressive loss of joint mobility and function |
||||
|
|
SKELETAL |
Osteoporosis |
Reduced bone density due to inflammation and corticosteroid use → ↑ fracture risk |
|
|
|
HEMATOLOGIC |
Anemia of chronic disease |
Decreased RBC production due to chronic inflammation |
|
|
Felty’s syndrome |
Triad: RA + splenomegaly + neutropenia |
|||
|
|
CRDIOVASCULAR |
Atherosclerosis |
Chronic inflammation accelerates plaque formation → ↑ MI and stroke risk |
|
|
Pericarditis / Myocarditis |
||||
|
Inflammatory involvement of cardiac tissues |
||||
|
|
PULMONARY |
Pleural effusion Pulmonary |
Fluid accumulation in pleural cavity |
|
|
hypertension Interstitial lung disease (ILD |
Increased pulmonary arterial pressure |
|||
|
|
NEUROLOGICAL |
Peripheral neuropathy |
Nerve compression or vasculitis-related nerve damage |
|
|
Cervical myelopathy |
Atlantoaxial subluxation leading to spinal cord compression |
|||
|
|
OCULAR |
Sjögren’s syndrome |
Dry eyes due to autoimmune gland destruction |
|
|
Scleritis / Episcleritis |
||||
|
Painful inflammatory eye disorders |
||||
|
|
CUTANEOUS |
Rheumatoid nodules |
Subcutaneous nodules, commonly over pressure points |
|
|
Vasculitis |
||||
|
Inflammation of blood vessels → ulcers, ischemia |
||||
|
|
RENAL |
Amyloidosis |
Protein deposition in kidneys → renal dysfunction |
|
|
|
INFECTIOUS |
Increased infection risk |
Due to immunosuppression (disease + therapy)C |
|
|
|
PSYCHOLOGICAL |
Depression / anxiety |
Chronic pain and disability affecting mental health |
|
|
|
METABOLIC/SYSTEMIC |
Cachexia |
Muscle wasting due to chronic inflammation. (21-23) |
|
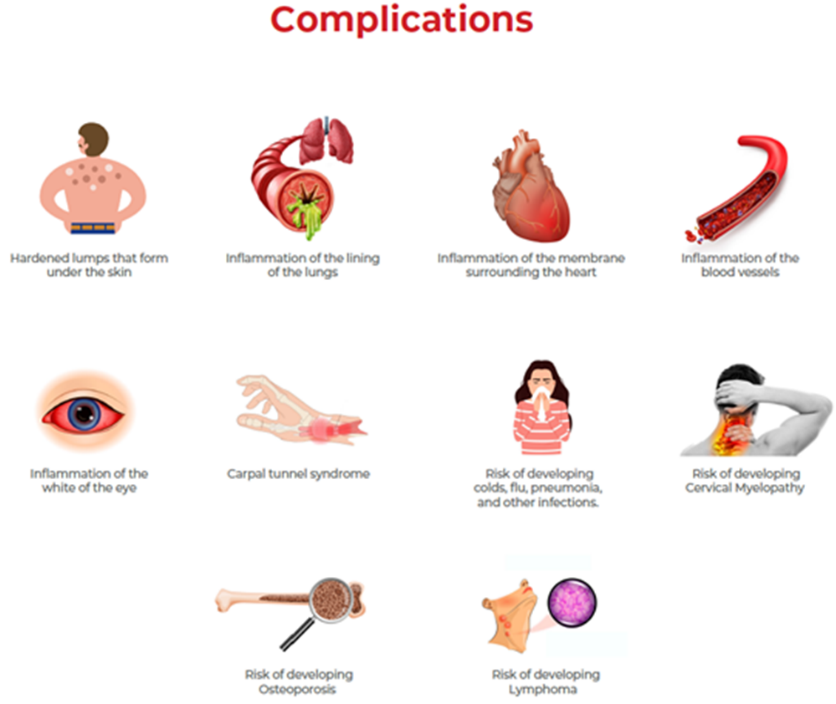
(24)
CONVENTIONAL DRUGS OF RHEUMATOID ARTHRITIS WITH LIMITATIONS:-
|
Category |
Drug/Class |
Mechanism of Action |
Key Effects in RA |
Major ADRs / Limitations |
Refs |
|
NSAIDs |
Non-selective (Ibuprofen, Diclofenac, Naproxen) |
COX-1 & COX-2 inhibition → ↓ Prostaglandins |
Symptomatic relief only |
GI ulcer, bleeding, renal & CV risk |
[25-27] |
|
|
COX-2 inhibitors (Celecoxib, Etoricoxib) |
Selective COX-2 inhibition |
Reduced GI toxicity vs NSAIDs |
↑ Cardiovascular risk |
[26-28] |
|
Glucocorticoids |
Prednisolone |
↓ Cytokines (IL-1, TNF-α), immunosuppression |
Rapid relief, slows joint damage |
Osteoporosis, diabetes, infection, adrenal suppression |
[29-31] |
|
Conventional DMARDs |
Methotrexate |
↑ Adenosine, ↓ T-cell activation |
First-line, slows progression |
Hepatotoxicity, myelosuppression, GI toxicity |
[32-34] |
|
|
Hydroxychloroquine |
Inhibits lysosomal activity & cytokines |
Mild RA, combination therapy |
Retinopathy, GI upset |
[35-36] |
|
|
Sulfasalazine |
↓ NF-κB, TNF-α |
Anti-inflammatory |
GI upset, hypersensitivity |
[37] |
|
|
Leflunomide |
Inhibits pyrimidine synthesis |
↓ Lymphocyte proliferation |
Hepatotoxicity, teratogenicity |
[38] |
|
|
Azathioprine |
Inhibits purine synthesis |
Immunosuppressive |
Bone marrow suppression, hepatotoxicity |
[39] |
|
|
Cyclosporine |
Calcineurin inhibitor → ↓ IL-2 |
Reduces immune activation |
Nephrotoxicity, hypertension |
[40] |
|
Biologic DMARDs |
TNF inhibitors (Etanercept, Infliximab, Adalimumab) |
Block TNF-α |
Strong disease control |
Serious infections (TB), malignancy risk |
[41-43] |
|
|
Abatacept |
Inhibits T-cell activation (CD80/86) |
Reduces immune response |
Infection risk |
[44] |
|
|
Tocilizumab |
IL-6 receptor blocker |
Effective in resistant RA |
Infection, liver enzyme elevation |
[45] |
|
|
Rituximab |
Anti-CD20 → B-cell depletion |
Severe RA |
Infusion reactions, infection |
[46] |
|
Targeted Synthetic DMARD |
Tofacitinib |
JAK inhibitor |
Oral alternative |
Infection, thrombosis risk |
[47] |
MANAGEMENT OF RHEUMATOID ARTHRITIS:-
MEDICINAL PLANTS WITH ANTI-ARTHRITIC ACTIVITY:-
|
S. No. |
Plant |
Family |
Active Constituents |
Mechanism of Anti-RA Action |
Ref. |
|
1 |
Aloe barbadensis |
Liliaceae |
Aloe-emodin |
↓ PGE2, NO, IL-6, TNF-α, NF-κB inhibition |
[49-51] |
|
2 |
Abrus precatorius |
Fabaceae |
Abrusosides |
Anti-inflammatory |
[52,53] |
|
3 |
Alstonia scholaris |
Apocynaceae |
Alkaloids |
↓ COX, LOX, NO, leukocyte migration |
[54,55] |
|
4 |
Andrographis paniculata |
Acanthaceae |
Andrographolide |
NF-κB inhibition |
[56-59] |
|
5 |
Arctium lappa |
Asteraceae |
Arctigenin |
↓ NO, iNOS, cytokines |
[60,61] |
|
6 |
Artemisia capillaris |
Asteraceae |
Scoparone |
↓ NO, PGE2, NF-κB |
[62,63] |
|
7 |
Berberis lyceum |
Berberidaceae |
Alkaloids |
↓ prostaglandins |
[64,65] |
|
8 |
Boswellia serrata |
Burseraceae |
Boswellic acids |
↓ NF-κB, COX-2, LOX |
[66-68] |
|
9 |
Cannabis sativa |
Cannabaceae |
Cannabidiol |
↓ TNF-α, IL-6 |
[69,70] |
|
10 |
Curcuma longa |
Zingiberaceae |
Curcumin |
↓ NF-κB, LOX |
[71-75] |
|
11 |
Glycyrrhiza glabra |
Leguminosae |
Glycyrrhizin |
↓ IL-6, TNF-α |
[76,77] |
|
12 |
Moringa oleifera |
Moringaceae |
Polyphenols |
↓ COX-2, cytokines |
[78,79] |
|
13 |
Nigella sativa |
Ranunculaceae |
Thymoquinone |
↓ IL-1β, TNF-α |
[80-85] |
|
14 |
Piper nigrum |
Piperaceae |
Piperine |
↓ prostaglandins |
[86] |
|
15 |
Rheum palmatum |
Polygonaceae |
Emodin |
↓ NF-κB, cytokines |
[87,88] |
|
16 |
Terminalia chebula |
Combretaceae |
Tannins |
↓ TNF-α, IL-6 |
[89] |
|
17 |
Withania somnifera |
Solanaceae |
Withanolides |
NF-κB inhibition |
[90] |
|
18 |
Zingiber officinale |
Zingiberaceae |
Gingerol |
↓ PGE, NO |
[91,92] |
|
19 |
Vitis vinifera |
Vitaceae |
Resveratrol |
↓ Th17, IL-17 |
[93-95] |
MECHANISM OF ANTI-RHEUMATOID ARTHRITIS ACTIVITY:-
LIMITATIONS OF CONVENTIONAL ANTI-RHEUMATOID ARTHRITIS:-
RECENT ADVANCEMENT AND FUTURE PROSPECTS OF ANTI-RHEUMATOID ARTHRITIS THERAPY:-
Targeted, disease-modifying strategies have replaced symptomatic relief in the treatment of rheumatoid arthritis (RA) due to recent developments. The first-line treatment is still methotrexate, but by focusing on particular immune pathways, biologic DMARDs like TNF-α inhibitors, interleukin inhibitors, rituximab, and abatacept have greatly improved disease control. Targeted synthetic DMARDs act on intracellular signaling pathways and are administered orally, especially JAK inhibitors such as tofacitinib and baricitinib. While the treat-to-target strategy emphasizes early diagnosis and ongoing monitoring to achieve remission, the introduction of biosimilars has improved treatment accessibility. Personalized medicine, pharmacogenetics, epigenetic treatments, and stem cell-based strategies for accurate and long-term management are some of the future prospects. However, there are still a lot of obstacles to overcome, including high costs, safety issues, and inconsistent patient response. (96-98)
REFERENCES






Patel Prince Jignesh Kumar, Patel Mit Kajal kumar, Chauhan Parth kumar Shailesh bhai, Anil Govardhan bhai Dewasi, Aasiya Khan, Jaswandi Mehetre, Herbal Therapeutics in Rheumatoid Arthritis: A Comprehensive Review, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 1252-1267, https://doi.org/10.5281/zenodo.19468568
 10.5281/zenodo.19468568
10.5281/zenodo.19468568