
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shri Wagheshwar Gramvikas Pratishthan's Loknete Shri Dadapatil Pharate College of Pharmacy, A/p-Mandavgan Pharata, Tal-Shirur, Dist.-Pune, Pin Code: 412211.
Schiff bases and their transition metal complexes have long been recognized for their pharmacological relevance, particularly as antioxidant and anticancer agents. The imine functionality within Schiff bases provides versatile coordination sites for transition metals such as copper, cobalt, nickel, and ruthenium, which often enhances biological activity compared to free ligands. In recent years, molecular docking and related computational tools have become indispensable in guiding the design of Schiff base ligands, enabling prediction of binding affinity, target selectivity, and mechanistic insights before synthesis. Such in-silico approaches significantly reduce experimental trial-and-error, allowing rational optimization of lead scaffolds. Experimental studies consistently report that Schiff base metal complexes can exert anticancer effects through DNA intercalation, topoisomerase inhibition, and reactive oxygen species (ROS)-mediated apoptosis, while antioxidant effects are largely attributed to metal-assisted redox mechanisms. This review highlights advances in molecular docking-guided design of Schiff base complexes, with an emphasis on their synthesis, antioxidant potential, and anticancer activity. The integration of computational modeling with wet-lab validation represents a promising strategy to accelerate the discovery of novel Schiff base-based therapeutics. [1-5].
Schiff bases, first reported by Hugo Schiff in 1864, are a class of compounds characterized by the presence of an azomethine (–C=N–) functional group formed via the condensation of primary amines with carbonyl compounds [6]. Over the years, Schiff bases and their transition metal complexes have attracted immense attention in medicinal chemistry owing to their structural versatility, ease of synthesis, and wide spectrum of biological activities. Among these, antioxidant and anticancer activities have been most intensively investigated, with numerous reports demonstrating that metal coordination often enhances the pharmacological potential of Schiff bases compared to their parent ligands [7,8]. Transition metals such as copper, cobalt, nickel, and ruthenium readily coordinate with Schiff bases, producing complexes with unique geometries and electronic properties. These complexes have shown strong interactions with biological macromolecules, including DNA, RNA, and proteins, often leading to cytotoxic and pro-apoptotic effects in cancer cells [9,10]. Furthermore, the redox-active nature of these complexes contributes to their ability to scavenge free radicals, thereby exhibiting antioxidant properties relevant in preventing oxidative stress–related disorders [11]. In recent years, molecular docking and in-silico approaches have emerged as valuable tools in rational drug design. By predicting binding affinity, orientation, and possible target interactions, docking studies provide insights into the biological mechanisms of Schiff base complexes before laboratory synthesis. This computational guidance not only reduces experimental trial-and-error but also accelerates the optimization of lead compounds for enhanced biological activity [12,13]. Integrating molecular docking with synthetic and biological evaluation thus represents a powerful hybrid approach in modern pharmaceutical chemistry. This review aims to provide a comprehensive overview of recent advances in the molecular docking-guided design and synthesis of Schiff base metal complexes, with a focus on their antioxidant and anticancer properties. We summarize the role of computational modeling in identifying active scaffolds, highlight experimental findings on biological activities, and discuss future perspectives for the development of Schiff base–based therapeutics.
DISCUSSION:
1. Schiff Base Complexes and Antioxidant Activity
Schiff base ligands are widely recognized for their antioxidant activity because they can stabilize free radicals via hydrogen atom transfer (HAT) and single electron transfer (SET) pathways. The presence of phenolic –OH, methoxy, or other electron-donating groups in the ligand framework enhances radical scavenging ability [14]. Transition metal coordination often strengthens these effects. For instance, Cu(II) Schiff base complexes exhibit superior antioxidant properties compared to Ni(II) or Co(II) analogues due to their favorable redox potential and ability to stabilize intermediate radical species [15]. Zn(II) complexes, although redox-inactive, show high activity because of their efficient superoxide dismutase (SOD)-mimetic behavior [16]. Studies have reported Schiff base–metal complexes with strong DPPH radical scavenging, ABTS assay activity, and lipid peroxidation inhibition, suggesting their potential role as therapeutic antioxidants and as protective adjuvants in oxidative stress-mediated disorders [17,18].
2. Schiff Base Complexes as Anticancer Agents
Schiff base metal complexes display diverse anticancer mechanisms. Unlike cisplatin, which primarily crosslinks DNA, Schiff base complexes often act through DNA intercalation, oxidative DNA cleavage, topoisomerase inhibition, and mitochondrial dysfunction [19].

SAR studies suggest that planar aromatic ligands enhance DNA intercalation, while electron-withdrawing substituents increase electrophilic reactivity toward biomolecules [23]. These features make Schiff base complexes versatile multi-target anticancer agents.
3. Structure–Activity Relationships (SAR)
The biological performance of Schiff base complexes is highly dependent on their metal center, geometry, and substituents:
Such SAR insights are now being refined using molecular docking and molecular dynamics simulations, which provide atomistic details of DNA, protein, and enzyme interactions [27].
1. General Schiff Base Scaffold
Ar−CH=N−Ar′Ar-CH=N-Ar'Ar−CH=N−Ar′
Representative Structure: Salicylideneimine Schiff base
2. Cu(II)–Schiff Base Complexes
Representative structure: [Cu(L)?] (L = Schiff base ligand).
3. Ru(II)–Schiff Base Complexes
Representative structure: [Ru(L)?Cl?].
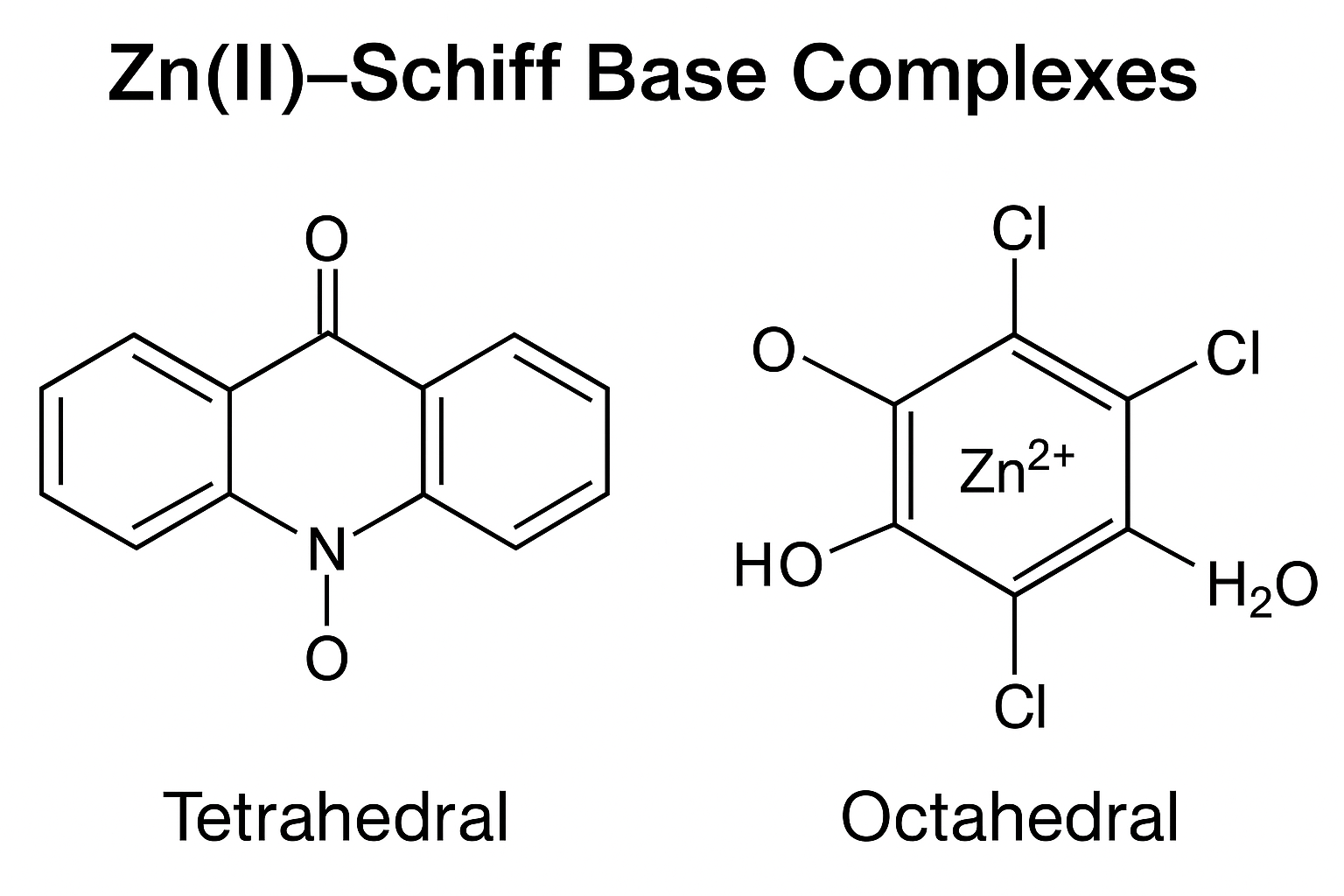
4. Zn(II)–Schiff Base Complexes
Representative structure: [Zn(L)?] or [Zn(L)(H?O)?].
4. Toxicity, Safety, and Delivery Systems
Despite strong in vitro activity, Schiff base complexes often face challenges of toxicity, solubility, and bioavailability. For instance, certain Cu(II) complexes have demonstrated dose-dependent hepatotoxicity in animal models [28].
To overcome these issues, researchers are exploring advanced delivery systems:
These strategies highlight a translational pathway from bench to bedside.
5. Clinical Relevance and Future Perspectives
Current chemotherapy relies heavily on platinum drugs such as cisplatin, which, despite effectiveness, suffer from drug resistance, nephrotoxicity, and neurotoxicity [31]. Schiff base complexes, particularly those of Ru(II) and Cu(II), present promising alternatives, with multiple modes of action and lower resistance potential [32].
Future directions include:
Thus, Schiff base complexes occupy an exciting niche in medicinal inorganic chemistry, bridging fundamental coordination chemistry with modern drug discovery.
CONCLUSION
Schiff base metal complexes represent a versatile class of compounds with remarkable antioxidant and anticancer potential. Their biological activity is governed by structural features such as the type of metal ion, ligand substituents, and planarity, which influence their binding affinity toward DNA, proteins, and enzymes. Molecular docking has emerged as a valuable predictive tool, correlating theoretical binding interactions with experimental antioxidant and cytotoxic outcomes. Together, these findings demonstrate that Schiff base complexes are not only strong radical scavengers but also promising anticancer agents capable of inducing apoptosis through DNA binding, topoisomerase inhibition, and ROS generation.
REFERENCES



Sneha J. Dighe*, Dr. Hemant V. Kamble, Sugriv R. Ghodake, Molecular Docking-Guided Design and Synthesis of Schiff Base Metal Complexes as Antioxidant and Anticancer Agents – A Review, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 8, 2786-2791 https://doi.org/10.5281/zenodo.16949332
 10.5281/zenodo.16949332
10.5281/zenodo.16949332