
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Vivekanandha Pharmacy College for Women, Sankagiri, Salem.
Pain is a physical discomfort ranging from mild to severe usually caused by injury, illness or nerve condition. Pain may arise from wide range of causes, categorized broadly into injuries, diseases and functional pain syndromes. Pain categorized into acute, chronic, neuropathic (pain caused by nerve damage) and nociceptive (pain caused by tissue damage). They are majorly managed by opioid analgesics and Non- Steroidal anti-inflammatory drugs. The opioid analgesic causes several risk factors such as addiction, dependence, respiratory depression and not suitable gestational period. The NSAID drugs induce gastrointestinal bleeding and cardiovascular risks. So, these risk factors are overcome by Suzetrigine as non- opioid and non- NSAID drug. It is the first new oral painkiller mainly used for short- term management of moderate to severe pain management, including post- operative pain. It targets NaV1. 8 sodium channels on peripheral nociceptive neurons and blocks sodium influx during action potential and prevents depolarization of pain- transmitting neurons then reduces transmission of nociceptive signals. NaV1.8 is found only in peripheral nerves and not expressed in the brain or spinal cord, so Suzetrigine does not cross into the CNS or interact with opioid receptor and avoiding risks associated with addiction. NSAID decrease prostaglandin, good for inflammatory pain and weak in neuropathic pain but Suzetrigine is highly effective in neuropathic pain.
Pain is an vexatious sensory and emotional experience induced by physical, chemical, psychological factors or often several combination factors [1]. The physical factors are wounds, burns, sprains, factures, muscle strain, pressure from swelling, tumour, tight bandages or any other surgical procedures [2]. Pain is also induced by the release of certain chemicals from the injury and inflammation site, that chemicals stimulate the pain receptors [3]. The chemical factors are bradykinin, prostaglandins, histamine, serotonin and substance P [3]. And also occurred by arthritis, diabetic neuropathy, post- herpetic neuralgia, carpal tunnel, spinal cord injury [4]. The psychological factors are depression, fear, tension. These are the factors made the pain sensation worsen [1]. Pain acts as a warning signal to feel something harmfully happen or make aware ourselves to protect from harm. The main functions of pain are to identify and locate the disease and disorder sites and also used to identify the injury and potential damage [2]. It can influence the mood, behaviour and mental health [5]. The effectiveness of pain can be vary depends upon the intensity, duration and location of the injury [2].
Types of Pain:
Pain can be classified into the following classes — based on duration, source, nature, or mechanism.
Table no: 1 Classification based on the duration
|
Type |
Description |
Example |
|
Acute Pain protective (helps avoid further damage) |
Sudden in onset, short-lived (seconds to weeks). It warns of injury and disappears when healing occurs. |
Post-surgical pain, fracture, burn
|
|
Chronic Pain non-protective (becomes a disease itself) |
Persists for months or years. It may continue even after the original Injury heals. Often dull and emotionally tiring. |
Arthritis, cancer pain, back pain |
[1,2,6,10] – Covers acute vs chronic pain definitions, protective vs non-protective pain.
Table no: 2 Classification based on the Site or Origin
|
Type |
Origin |
Nature of Pain |
Example |
|
Somatic Pain |
From skin, muscles, bones, joints |
Sharp, well-localized |
Cut, sprain
|
|
Visceral Pain |
From internal organs (heart, stomach, intestines) |
Dull, cramping, poorly localized |
Gastric ulcer, gallstones |
|
Neuropathic Pain |
From damage or disease of nerves |
Burning, shooting, tingling |
Diabetic neuropathy, shingles |
|
Psychogenic Pain |
No clear physical injury due to emotional or mental stress |
Varies (headache, body ache) |
Tension headache, stress pain
|
|
Referred Pain |
Felt at a location different from the actual site |
Deep, aching |
Heart attack felt in left arm
|
[2,3,7,12,13] – Covers somatic, visceral, neuropathic, psychogenic, referred pain.
3.Based on Mechanism (How Pain Occurs)
Table no: 3 Classification based on the Mechanis
|
Type |
Description |
Example |
|
Nociceptive Pain |
Due to activation of pain receptors (nociceptors) by injury or inflammation |
Cut, burn, fracture |
|
Inflammatory Pain |
Caused by release of chemical mediators during inflammation |
Arthritis, infection |
|
Neuropathic Pain |
Caused by nerve injury or irritation |
Trigeminal neuralgia, diabetic neuropathy |
|
Functional Pain |
No clear tissue or nerve damage; abnormal pain processing in brain |
Fibromyalgia, irritable bowel syndrome |
[3,4,8,9,13] – Explains nociceptive, inflammatory, neuropathic, functional mechanisms.
Table no: 4 Classification based on the sensation [2,3,4]
|
Type |
Description |
Example |
|
Sharp pain |
Sudden and localized |
Pinprick, knife cut |
|
Dull pain |
Deep, aching, poorly localized |
Muscle ache |
|
Burning pain |
Tingling, nerve-related |
Neuropathy |
|
Colicky pain |
Comes and goes in waves |
Kidney stones, intestinal colic |
|
Throbbing pain |
Rhythmic with heartbeat |
Migraine, abscess |
|
Cramping pain |
Due to muscle spasm |
Menstrual cramps |
[2,3,4,12,13] – Describes sharp, dull, burning, throbbing, colicky, cramping.
Table no: 5 Classification based on etiology
|
Type |
Description |
Example |
|
Cancer Pain |
Due to tumour growth pressing on tissues |
Bone metastasis |
|
Phantom Pain |
Felt in an amputated limb |
After limb amputation |
|
Breakthrough Pain |
Sudden pain in a patient already on pain medication |
Cancer therapy |
|
Incident Pain
|
Triggered by movement or activity
|
Pain during physiotherapy
|
[5,14,15,16] – Cancer pain, phantom pain, breakthrough pain, incident pain.
Table no: 6 Classification based the pathway
|
Type |
Pathway Involved |
Example |
|
Peripheral Pain |
Originates in peripheral tissues |
Burn, injury |
|
Central Pain |
Originates in central nervous system (brain/spinal cord) |
Post-stroke pain |
[3,4,8,13] – Central vs peripheral pain pathways.
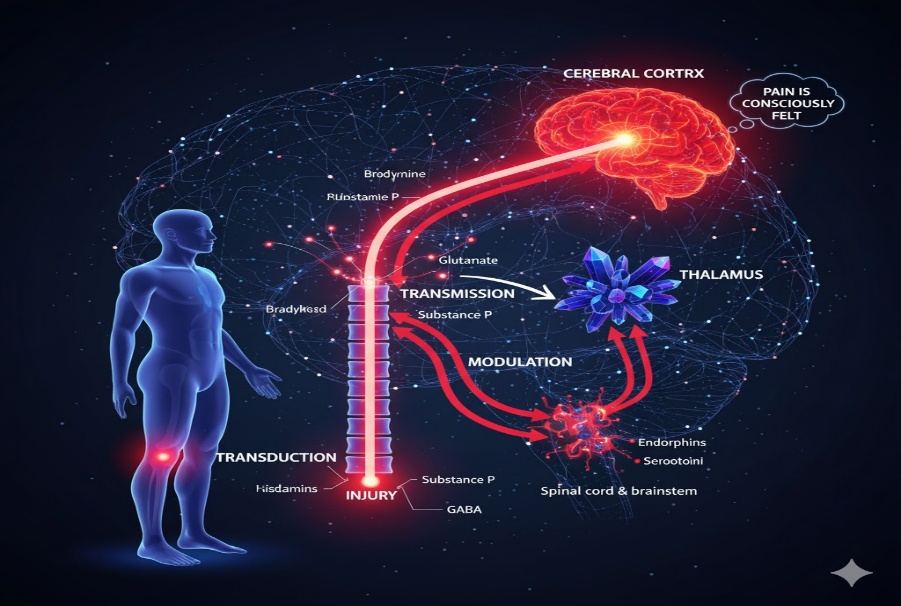
Pain Generation Pathway:
Pain generation is one of the complex physiological process, that can arise when noxious stimuli activate the specialised sensory receptors called nociceptors [17,18,19]. The pain generation pathway involves mainly four steps, there are

1.Transduction:
Transduction is the process that can converts harmfull stimulus into electrical nerve stimulus, through chemical mediators such as bradykinin, prostoglandins, serotonin, histamine and substance Preleased from the tissue injury [18,19,24]. These type of chemical mediators stimulate pain receptors present in the skin, muscles, joints and viscera [18,19]. The nociceptive receptors present converts the arising chemical signals into an action potential (electrical impulses) [24]
2.Transmission:
Transmission is the process transmits the electrical signals from the site of injury to the brain. In this process it consists three pathways, there are first order- neurons, second order- neurons, third- order neurons. In first order- neurons [20,24], Impulses transmit from nociceptors to the dorsal horn of the spinal cord. Second order- neurons, caries the signal from the spinal cord to the thalamus through spinothalamic tract [20]. Third- order neurons, relies the signal from the thalamus to the cerebral cortex, where the pain is recognized [17,20].
3.Modulation:
Modulation is the process, the central nervous system either amplified or reduced the pain signals, occurs at the spinal cord through descending neural pathways such as periaqueductal gray (PAG), rostral ventromedial medulla (RVM) and locus coeruleus [21,23]. These pathway release the neurotransmitters such as endporphins, serotonin, norepinephrine and enkephalins block the excitatory chemicals likes substance P and glutamate, inhibit the pain transmission [21,22]. Modulation process is also called central sensitization, increased excitability and reduced pain inhibitors by release of excitatory chemicals [21,24].
4. Perception:
Pain perception occurs when nociceptive impulses reach higher brain regions and are interpreted as a conscious sensory experience [17,23]. These impulses are first integrated in the thalamus and subsequently analyzed in the somatosensory cortex to define the intensity and site of pain, while the limbic system mediates the associated emotional response [22,23]. Consequently, the experience of pain depends on coordinated processing within multiple central nervous system structures rather the solely on peripheral stimulation [17,22,23].
Various Possibilities of Pain Management:
Pain Management methods can be depend on the types, cause and severity of pain. The pain management is usaully a combination of various class of drugs and other physio methods, there are [6,25]
Non- Pharmacological methods:
Pharmacological methods:
Interventional Methods:
Indication: Suzetrigine is indicated for the treatment of pain.
Suzetrigine (formerly VX-548 in development phases, branded as Journavx) is a non-opioid, non- NSAID first-in-class analgesic approved in January 2025 by the U.S. FDA to moderate-to-severe acute pain [26,27,28]. It is regarded as the breakthrough in pain management as it is the first new pharmacologic class in decades [26,27].
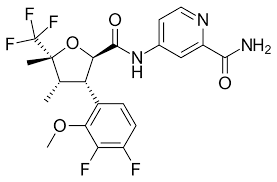
Suzetrigine is referred to in the IUPAC name as 4-[[(2R,3S,4S,5R)-3-(3, 4-difluoro-2-methoxyphenyl)-4,5-dimethyl-5-(trifluoromethyl)oxolane- 2 -carbonyl] amino]pyridine-2-carboxamide. It is also called the VRT-1737548 code name. The chemical formula of Suzetrigine is C21H20F5N3O4 [26, 29, 33]. Pain is a highly frequent cause of visit to the doctor and typically treated using drugs such as non-steroidal anti-inflammatory drugs and opioid analgesics. The long-term use of these medicines may cause severe conditions such as stomach and kidney damages where NSAIDs are used, respiratory complications, decreased effectiveness with time, and an addiction to opioids. Development of less addictive pain medication that does not rely on opioids is thus an extremely critical objective in therapy [30, 31].
Suzetrigine is a recently developed kind of pain medication which can be administered orally. It does not fall under the NSAID or opioid drugs. Its mechanism of action involves having a selective inhibitory effect on a sodium channel known as Na V1.8, which is involved in the transmission of pain in the body. The characteristic of suzetrigine is that it is used to treat pain by targeting the pain signals in the body, but does not influence opioid receptors, which will lower the risks of getting addicted and the side effects on the brain and nerves will also be reduced. This renders suzetrigine a suitable choice in treating pain in the current medical practice [26,28,32].
Suzetrigine Chemical Nature.
Suzetrigine is an artificial, tiny molecule analgesic medication that was designed to inhibit a particular sodium-channel known as NaV1.8. It is an organic compound that has a ring structure in it with various atoms and it acts by attacking some pain-sensing nerves in the body. It consists of an oily part and a water-loving part that collaborate to ensure that the molecule manages to move across membranes with ease and select the appropriate target. Suzetrigine may be orally administered and is able to withstand a body, thus being an effective pain reliever that can act on the body[31,33].
Suzetrigine chemical structure.
The structure of Suzetrigine is a small-molecule NaV1.8 sodium channel inhibitor and it can be described as follows:
• It has a heterocyclic core ring system.
• Contains aromatic (phenyl/heteroaryl) rings that offer hydrophobic binding.
• It has a basic amine group (essential in interaction with sodium channels)
• Has lipophilic and polar functional groups, which are well orally bioavailable.
Structurally dissimilar to:- Opioids (no morphinan nucleus), NSAIDs (no COX-inhibitory scaffold)
Its structure is designed to:
Selective binding of NaV1.8 channels.
NaV1.5 (cardiac): avoid NaV1.5 (cardiac)
NaV1.7 (CNS): NaV1.5 (cardiac): avoid NaV1.5 (cardiac) and NaV1.7 (CNS)
Restrict CNS penetration - reduced risk of addiction[26,28,34,35].
Structural Activity Relationship:
Research on the mechanism of action in relation to the structure of suzetrigine reveals that its pain-killer properties are primarily due to its specific antagonism of the NaV1.8 sodium channel [36,37].
Key SAR features include:
Aromatic/Hetero aromatic ring system.
The aromatic or heterocyclic rings are useful in increasing the binding of a molecule to the NaV1.8 channel, as more hydrophobic bonds can be formed within the pore of the channel[36,39].
Basic amine functionality
The protonatable amine functionality is significant to forming an electrostatic bond with negatively charged currents of the sodium channel that obstructs the channel and renders it selective[39,40].
Optimal lipophilicity
The correct proportion of lipophilicity also allows a drug to infiltrate peripheral sensory nerves, but not to reach the central nervous system which reduces the chances of side effects involving the brain and spinal cord [36,38].
Spatial arrangement and steric bulk
The location of the substituents as well as the size of the molecule influences the ions that can get into the channel. Big domains attached to the molecule render it less probable to interact with other varieties of sodium channels, such as NaV1.5 in heart tissue and NaV1.7 in brain neurons, which increases the safety of the drug [36,37,38].
Substituent modifications
Even minor modifications to the chain of side length or the functionality of the group type can significantly affect the effectiveness of the work of the drug and its circulation in the body. This demonstrates that the structure must be perfectly correct in order to get the drug to the NaV1.8 in order to have a prolonged working time with the purpose of pain management [39,41].
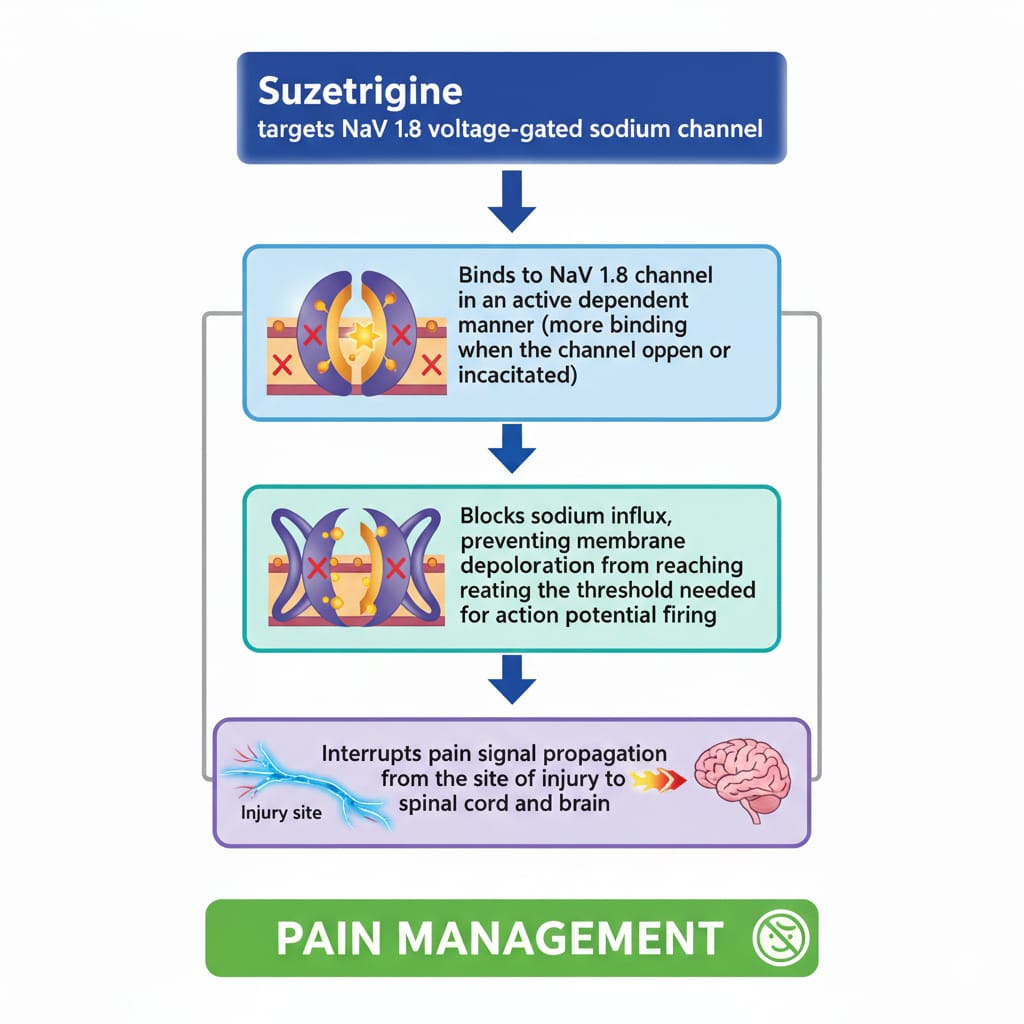
Mechanism of Action:
Suzetrigine is a medication that inhibits particular kind of voltage-gated sodium channel or NaV1.8. These are predominantly located in the pain nerves of the dorsal root ganglia. These are the channels that are instrumental in initiating and transmitting the electrical impulses that convey pain messages particularly in the case of tissue damage or swelling [42,43]. The attachment of Suzetrigine to NaV1.8 channels is conditioned by the state of the channel and prevents the entry of sodium ions into neurons in case they are depolarized. This leads to reduced production and transmission of electrical impulses on the pain nerves in the body [42,45]. Consequently, the body pain receptors which are relayed to the spinal cord and then to the brain to be processed are significantly diminished. The impact of suzetrigine on other sodium channels is minimal such as NaV1.5 in the heart, and other sodium channels in the central nervous system hence, reducing the possibility of cardiovascular toxicity and other adverse side effects in the heart and nervous system. Besides, it does not work on μ-opioid receptors or cyclooxygenase enzymes, which distinguishes it as an opioid pain reliever and NSAIDs [43,45]. Suzetrigine is a good pain killer, which modulates the activity of nerve cells in the body. It accomplishes it without producing the side effects that are commonly associated with opioids, such as a decrease in breathing, decreased efficacy with continued use, and developing an addiction to the substance. It also does not cause stomach and kidney issues that are prevalent with the NSAIDs [42,44].
Methodology
Study Design
This is a narrative literature review which examines the effectiveness of suzetrigine as a pain reliever, and in this instance a non-NSAID and non-opioid analgesic in the management of pain [44,45,46]. It was aimed to collect, examine, and compile with the published research on its chemical composition, its effect on the body, its effects on health, and its potential use as a treatment [50,51,52].
Data Sources
Some online databases such as PubMed, Scopus, ScienceDirect, and Google Scholar were utilized by the researchers to collect scientific articles. The reason why these databases were selected was to ensure that any significant publications on pharmaceuticals, biomedical research and clinical research are not left out.
Search Strategy
These words were searched in a planned manner: Suzetrigine, VX-548, NaV1.8 sodium channel inhibitor, non-opioid analgesic and pain management. Search was narrowed down by adding such words as AND and OR to target results and narrow it out. Associated articles had reference lists that were verified to identify additional studies [46,47,48].
Inclusion criteria:
• Studies that describe the action of suzetrigine in the body, its influence on brain chemicals and its efficacy in treating a disease.
A research conducted before human testing, such as a test in the laboratory and on living animals, and research conducted on humans to comprehend the process of pain management.
Exclusion criteria:
Other studies that are outside the topic of suzetrigine or inhibition of the sodium channel NaV1.8.
• Articles that merely discuss opioids, NSAIDs, or other pain medications that are not related to the subject matter.
• Commentaries, non-scientific reports and editorials.
• Repetitive works and those that lack adequate data.
Study Selection Process
The articles that were located were initially sieved by glancing at the title and summaries to determine whether they were of relevancy. The articles that were included in the inclusion criteria were subsequently verified in full. Only the studies providing clear and specific information regarding the effects of medicine suzetrigine on the body and its capacity to decrease pain were included in the final analysis.
Data Extraction
Based on the selected studies, valuable information was selectively collected, which included:
• Pharmacological and chemical properties of suzetrigine.
• Mechanism of action that includes NaV1.8 channel inhibition.
• Pain evaluation experimental models.
• Clinical efficacy outcomes
• Safety and tolerability data.
• Data Synthesis and Analysis
The data that were extracted were reviewed in a more detailed manner and were placed under various broad categories such as the type of chemical it is and how its structure correlates with its effects, the way it acts in the body, what it does in the medical field, and its medical safety. The comparison was made using the results of preclinical studies and clinical trials to indicate the effectiveness of suzetrigine in the treatment of pain.
Ethical Considerations
This is a review of existing information that has already been published and no experiments were carried out on humans or animals, which is why no ethical approval was required.
RESULTS
Literature Search Outcome
Literature search identified a number of preclinical and clinical trials that investigated suzetrigine as a new form of pain reliever, which is neither NSAID nor opioid. We selected the articles which discussed the chemical composition, the mechanism of action, its action on the body and the safety of the chemical on the body after reading the titles, the abstract and the entire text of the articles based on the inclusion and exclusion criteria [51,52,53].
Pharmacological Activity
The selected studies indicated that suzetrigine is effective in terms of administration of pain in both sudden and inflamed pain tests[44]. The principal action of the drug involves inhibition of particular sodium channel, NaV1.8, which is significant in transmission of pain in the body. Suzetrigine was used effectively compared to normal pain medicines in decreasing pain without having an impact on the opioid receptors and the cyclooxygenase pathways [50,51,52].
Preclinical Evidence
The preclinical trials indicated that suzetrigine significantly reduced the occurrence of pain behavioral responses in animals including heat, pressure, and inflammation [48,49]. The compound was highly effective on the NaV1.8 channels as opposed to NaVr and other types of sodium channels implying that it selects this type of channel. These findings demonstrate that it may be a pain reliever that is essentially acting on the outer section of the body and not putting a significant strain on the brain or spinal cord [57,65].
Clinical Evidence
The clinical trials indicated that suzetrigine produced increased pain relief with increasing dosage, particularly on individuals with moderate to severe pain, e.g. after surgery or during sudden episodes of intense pain [43]. The analgesic effect was rapid and the pain ratings were significantly lower than analgesic effects with placebo. Noteworthy, suzetrigine was pain relieving without resulting in opioid side effects, such as slowed breathing, drowsiness, or a sensation of extreme happiness [53,54,55,56,57].
Safety and Tolerability
Suzetrigine was generally tolerable in preclinical and clinical trials. The side effects were not serious and most common and included such things as headaches, feeling sick, and being lightheaded [46,50]. There were no significant indicators of abuse, breathing issues, or stomach issues. In addition, the minor effect on cardiac sodium channel indicated a low risk of heart toxicity [56,60,61,62].
Comparative Findings
Suzetrigine is different when compared to the conventional NSAIDs and opioid pain medicines [44,51]. It prevents sodium channels within the body, rather than preventing the production of prostaglandins or opioid receptors. This distinct separation of the mechanisms resulted in improved analgesia with minimal risk of gastroenteric, renal and addiction issues [52,59,63,69].
DISCUSSION
The current review dwells on suzetrigine as a novel form of pain reliever not a NSAID nor an opioid. It operates by attacking certain sodium channels of nerve cells which are involved in the transmission of pain. Suzetrigine is a good analgesic agent that modulates the effects of the nerve cells to pain transmission rather than preventing inflammation or opioid receptors [48,49,51]. This will minimize the possibility of side effects such as stomach problems, kidney complications, and brain and spinal cord related challenges the other pain medicines have [52]. This medication choice is effective in individuals who are unable to use NSAIDs or have the risk of developing issues with opioids.
The findings also indicate that suzetrigine may be useful when applied in combination with other painkillers that may minimize the use of opioids and result in improved pain control [56,57,58]. Its action acts on the area around the main part of the body, which may limit the possibility of misuse and easy to tolerate particularly in cases where it is applied in sudden or pain following surgery. Targeting sodium channels, which are involved in transmitting pain signals, is appropriate in the modern day tendency to deal with pain by its underlying mechanisms, which offers a more specific and rational approach to pain [60,61,62].
But the majority of the recent evidence examines the performance of something in the short-term, but there are few details on the safety over an extended time. The treatment of various pain conditions may be altered in the extent of sodium channels present, and this might influence the effectiveness of the treatment [63,64]. This implies that further studies are required to learn the contribution of sodium channels in the management of chronic pain. A major finding in the development of safer, non-opioid analgesics is the opportunity of Suzetrigine, which shows the growing importance of ion channel therapy in the development of new pain relief drugs [65].
CONCLUSION
Suzetrigine is an important breakthrough in pain medicine. It is also a good way of controlling pain without NSAIDs or opioids. It is more specific in the relief, acting on the particular voltage-monitored sodium channels that mediate transmission of pain impulses [44,46,48]. The method also minimizes the side effects that are not desired with the conventional pain medications. Due to its mechanism of action, suzetrigine could be a powerful option in combination pain management strategies since it allows decreasing the use of opioids and make the management of the pain process safer [51,53,55]. A method-focused approach illustrates how an approach-based approach can be applied in the modern management of pain, as suzetrigine alters the life-focus in treating pain, shifting attention from the need to reduce inflammation and stimulate opioid systems in the brain to the activation of certain nerve pathways when managing pain [58,59]. Although the treatment is promising, it is good to conduct long-term studies to ensure that it remains effective, safe, and more effective than other treatments in the long-term. To sum up, suzetrigine is a promising new pain reliever, which contributes to the growing interest towards using medicines that are not opioids and NSAIDs to treat pain in a safer and more effective way [64,65,66].
REFERENCES







Dineshkumar Palanivel, Mouleeswaran Rajendran, Sakthi Sundaram Subramaniyam, Harinee Murugesan, Yamuna Rameshbabu, Dhivya Rajamanickam, Gowri Saravanan, Pharmacological Activity of Suzetrigine (Non- Opiod, Non- NSAIDS) In Pain Management., Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 1608-1621. https://doi.org/10.5281/zenodo.19045203
 10.5281/zenodo.19045203
10.5281/zenodo.19045203