
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department Allied and Healthcare, St. Soldier Institute of Pharmacy, Jalandhar-144011, Punjab, India
Lung cancer remains one of the leading causes of cancer-related morbidity and mortality worldwide, largely due to late-stage diagnosis and limited early symptoms. Radiology plays a critical role in the early detection, diagnosis, staging, treatment planning, and follow-up of lung cancer. Advances in imaging techniques such as chest radiography, computed tomography (CT), low-dose computed tomography (LDCT), positron emission tomography–computed tomography (PET-CT), and magnetic resonance imaging (MRI) have significantly improved the ability to identify lung tumors at earlier and more treatable stages. Among these modalities, LDCT screening has demonstrated substantial effectiveness in detecting small pulmonary nodules and reducing lung cancer–related mortality in high-risk populations. In addition, emerging technologies such as artificial intelligence (AI), radiomics, and machine learning are transforming thoracic imaging by enabling automated nodule detection, risk stratification, and personalized treatment planning. Despite these advances, several challenges remain, including false-positive findings, radiation exposure concerns, limited access to advanced imaging technologies in resource-limited settings, and ethical considerations surrounding AI implementation. This review highlights the evolving role of radiology in lung cancer care, focusing on imaging modalities used for early detection, current challenges, and future innovations that may enhance diagnostic accuracy and improve patient outcomes.
Across the world, lung cancer ranks among the most frequently occurring cancers and continues to be the leading cause of death associated with cancer1,2. Global lung cancer incidence and mortality continue to rise. According to GLOBOCAN 2018, approximately 2.09 million new cases and 1.76 million deaths were reported worldwide, accounting for 11.6% of all cancer diagnoses and 18.4% of cancer-related deaths. Compared with the 2012 estimates of 1.8 million cases and 1.6 million deaths, these figures demonstrate a growing disease burden3. Consequently, lung cancer remains the most frequently diagnosed cancer and the leading cause of cancer mortality worldwide, while in women it ranks as the third most common cancer and the second leading cause of cancer death4,5.
Tobacco smoking remains the most significant risk factor for lung cancer and is responsible for approximately 80–90% of cases worldwide. However, other risk factors such as secondhand smoke exposure, occupational carcinogens (asbestos, silica, radon), air pollution, and genetic susceptibility also contribute to the global burden of disease. Notably, an increasing proportion of lung cancer cases are now reported among never-smokers, particularly in Asian populations, suggesting the influence of environmental and molecular factors in disease pathogenesis6.
Temporal trends in lung cancer incidence and mortality demonstrate varying patterns between sexes and regions. In many high-income countries, lung cancer incidence and mortality among men have declined over the past few decades, largely due to reduced smoking prevalence following aggressive tobacco control policies. Conversely, lung cancer incidence among women has increased or plateaued in several regions, reflecting later adoption and cessation of smoking among female populations. These epidemiological patterns highlight the long latency period between tobacco exposure and the development of lung cancer7.
Future projections indicate that the global burden of lung cancer is expected to rise substantially over the coming decades due to population growth, aging demographics, and persistent exposure to risk factors. Estimates suggest that by 2050 the number of new lung cancer cases and deaths could increase by more than 60–70% globally, particularly in developing countries where tobacco control and early detection programs remain limited8. This projected increase underscores the urgent need for effective preventive strategies, early detection programs, and improved diagnostic technologies.
In this context, radiology has emerged as a crucial component in lung cancer control strategies. Imaging techniques such as low-dose computed tomography (LDCT) have demonstrated significant potential in detecting lung cancer at earlier stages, thereby improving survival outcomes. Understanding the global epidemiological trends of lung cancer provides an essential foundation for evaluating the role of radiological innovations in early detection and management.
Lung cancer is one of the most common malignancies worldwide and represents a major cause of cancer-related morbidity and mortality. According to global cancer statistics, lung cancer accounts for a significant proportion of newly diagnosed cancers and remains the leading cause of cancer deaths globally. The incidence and mortality rates vary widely across different regions due to differences in smoking prevalence, environmental exposures, occupational hazards, and socioeconomic factors. Higher incidence rates are typically observed in developed countries such as North America and Europe, although increasing trends are also being reported in several developing regions due to rising tobacco consumption and industrial pollution9,10. Epidemiological studies have also demonstrated variations based on sex and age, with historically higher incidence among men; however, the gap between men and women has been narrowing as smoking prevalence among women has increased in many countries11,12.
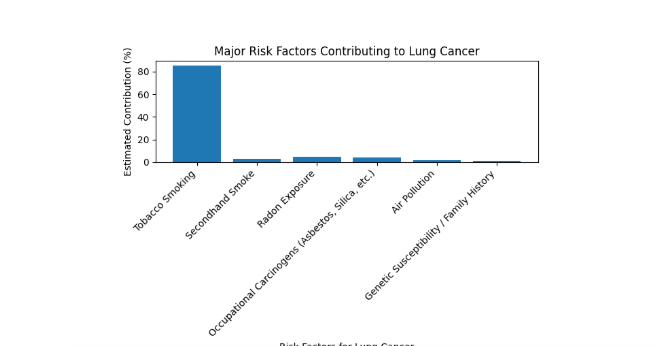
Graph 1: Major Risk Factor of Lung Cancer
Table 1: Major Risk Factors for Lung Cancer
|
Risk Factor |
Estimated Contribution (%) |
|
Tobacco smoking |
85 |
|
Secondhand smoke |
3 |
|
Radon exposure |
5 |
|
Occupational carcinogens (asbestos, silica, etc.) |
4 |
|
Air pollution |
2 |
|
Genetic susceptibility / family history |
1 |
Overall, lung cancer epidemiology reflects a complex interaction between lifestyle behaviors, environmental exposures, occupational hazards, and genetic factors. Among these, tobacco smoking remains the dominant and most preventable risk factor. Understanding these epidemiological patterns and risk determinants is essential for developing effective prevention strategies, public health interventions, and early detection programs aimed at reducing the global burden of lung cancer.
Radiology has become the cornerstone of lung cancer screening, fundamentally changing how the disease is detected at earlier and more treatable stages. Historically, screening relied on chest radiography; however, this approach showed limited sensitivity and did not significantly reduce mortality. The introduction of low-dose computed tomography (LDCT) marked a major turning point, offering higher resolution imaging capable of detecting small pulmonary nodules that are often missed on conventional radiographs19,20.
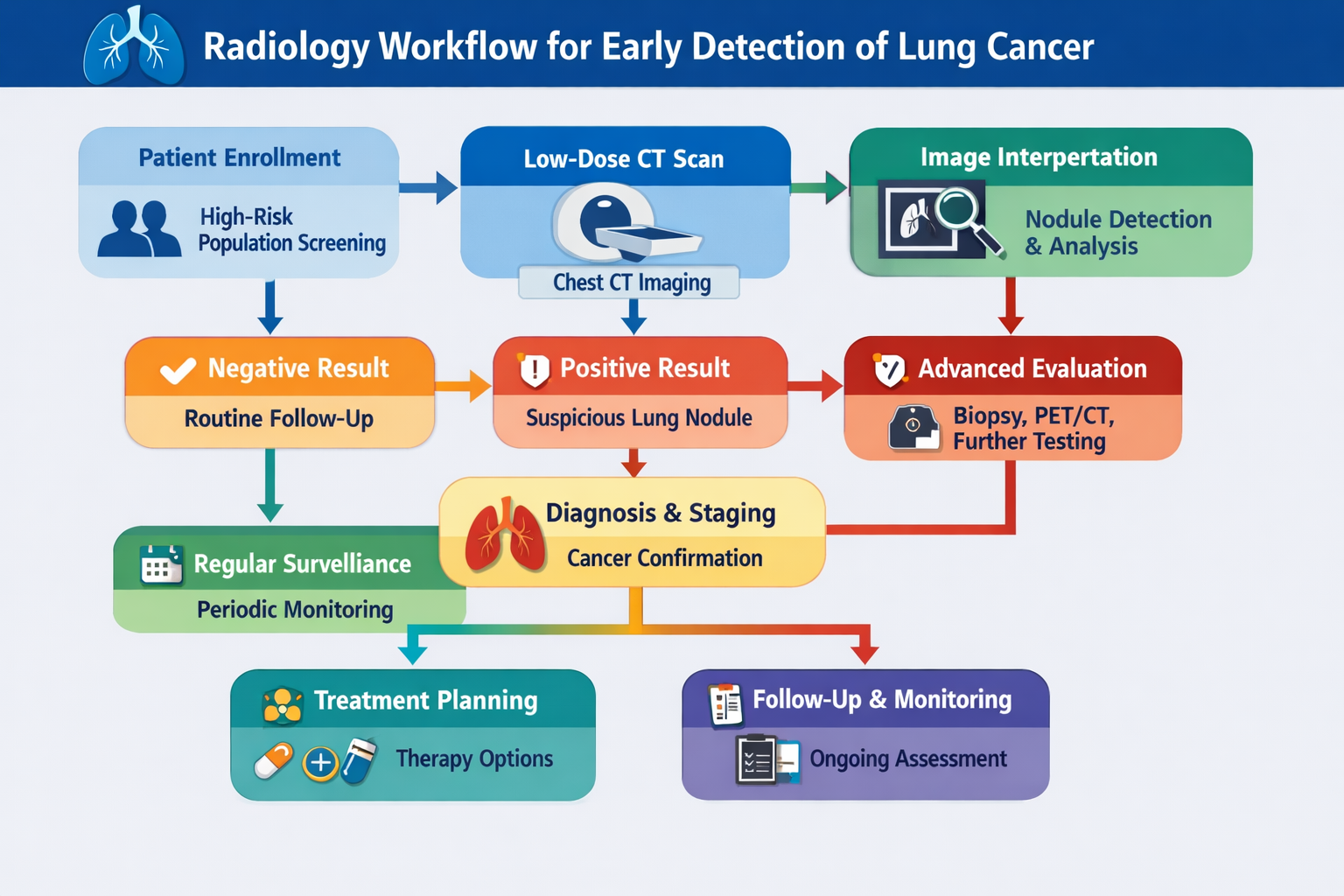
Figure 1: Role of Radiology in early detection of Lung Cancer
Large randomized controlled trials have firmly established the value of radiological screening. The National Lung Screening Trial (NLST) demonstrated that LDCT screening in high-risk individuals—primarily heavy smokers—resulted in approximately a 20% reduction in lung cancer–specific mortality compared to chest X-ray screening21,22. Similarly, the NELSON trial further strengthened this evidence, reporting even greater reductions in mortality and confirming that LDCT can detect cancers at earlier stages, when curative treatment is more feasible23,24. These findings have led to the widespread adoption of LDCT screening guidelines in several countries, particularly for high-risk populations.
From a practical perspective, radiology does more than simply detect lung nodules; it also plays a key role in risk stratification and longitudinal monitoring. Radiologists assess nodule size, shape, density, and growth over time to distinguish between benign and malignant lesions. Structured reporting systems and standardized protocols have been developed to improve consistency and reduce variability in interpretation25,26. In addition, follow-up imaging is often used to monitor indeterminate nodules, helping clinicians avoid unnecessary invasive procedures while ensuring timely diagnosis.
Despite its proven benefits, radiological screening is not without challenges. False-positive findings are relatively common, leading to additional imaging and sometimes invasive diagnostic procedures. However, advances in imaging techniques, volumetric analysis, and the integration of artificial intelligence (AI) are helping to improve diagnostic accuracy and reduce unnecessary interventions27,28. AI-based tools can assist in automated nodule detection, characterization, and risk prediction, potentially enhancing the efficiency and reliability of screening programs.
Table 1: The role of radiologists in lung cancer screening is complex and encompasses multiple critical responsibilities, ranging from image interpretation and early detection to risk assessment and ongoing patient monitoring.

Low-Dose CT in Lung Cancer Screening
In any screening program targeting individuals without symptoms, the benefits of detecting disease early must clearly outweigh any potential harm. In lung cancer screening (LCS), one of the primary concerns is exposure to radiation from CT imaging. Although the overall risk of developing radiation-induced cancer is relatively low compared to the life-saving potential of early detection, this risk increases with higher radiation doses. Fortunately, low-dose computed tomography (LDCT) protocols allow radiation exposure to be minimized while still maintaining sufficient image quality, largely because lung tissue naturally provides high contrast for detecting nodules.
To ensure safety and effectiveness, carefully designed LDCT protocols are essential. These protocols must strike a balance between reducing radiation dose and preserving diagnostic accuracy. Achieving this requires close collaboration between thoracic radiologists and medical physicists, who work together to optimize scanning parameters. Despite established recommendations—such as those from the American College of Radiology (ACR)—studies have shown considerable variation in radiation doses across institutions. For example, a large U.S. cohort study found that some centers exceeded recommended dose levels, particularly when there was limited collaboration between radiologists and physicists. In contrast, facilities with coordinated protocol development consistently achieved lower and safer radiation doses.
Medical physicists play a crucial role in maintaining radiation safety within screening programs. Their expertise includes equipment testing, quality assurance, and staff training in radiation protection. They also help monitor radiation doses and compare them with national reference standards, ensuring that imaging practices remain within safe limits. Their active involvement supports continuous improvement in both patient safety and image quality.
Modern guidelines emphasize the use of standardized dose metrics such as CTDIvol, which provides a more consistent measure of radiation exposure than previously used estimates like effective dose. Current European recommendations suggest maintaining CTDIvol within a low range, adjusted according to patient body size. Technical factors are also carefully controlled: scans should be completed quickly in a single breath-hold, use automatic exposure control to adapt to patient anatomy, and avoid unnecessary adjustments that could increase image noise. High-resolution imaging is achieved using thin-slice reconstruction, while advanced reconstruction techniques—such as iterative and deep learning-based methods—have largely replaced older approaches to improve image clarity at lower radiation doses.
Beyond technical considerations, accessibility remains an important challenge in lung cancer screening. Many individuals, particularly those in underserved or remote areas, face practical barriers such as travel distance to screening centers. To address this, mobile LDCT units have been introduced, bringing screening services closer to communities. Programs like those implemented in the United Kingdom and the Manchester “lung health check” initiative have demonstrated that mobile units can significantly improve participation rates, especially among high-risk populations such as smokers. However, it is essential that these mobile systems meet the same quality and safety standards as hospital-based scanners.
Baseline Radiological Examination: Chest X-ray
Chest X-ray (CXR) is usually the initial imaging test performed when lung cancer is suspected. Its widespread use is largely due to its availability, simplicity, low cost, and minimal radiation exposure, making it a practical first-line investigation. However, while CXR is useful for detecting abnormalities, it provides only limited detail, and any suspicious finding typically requires further evaluation with more advanced imaging techniques.
On CXR, lung cancer may appear in different ways depending on its location and stage. Tumors can present as central or peripheral masses, and in some cases, early forms such as in situ adenocarcinoma may appear as persistent areas resembling chronic airspace disease. Central tumors may be associated with enlarged hilar lymph nodes, mediastinal widening, or airway obstruction, which can lead to partial or complete lung collapse. In some cases, secondary findings such as consolidation or infection may obscure the tumor or be the first clue to an underlying malignancy. Radiologists should be particularly cautious when such abnormalities persist over time, as this raises suspicion for cancer.
Mediastinal involvement may be suggested on CXR by a widened mediastinal silhouette, although the technique has clear limitations in assessing deeper or more complex invasion, such as involvement of the phrenic nerve or major vessels like the superior vena cava. Many lung cancers detected incidentally, especially in asymptomatic individuals, appear as solitary pulmonary nodules, which may have smooth, irregular, or spiculated margins. Occasionally, additional features such as internal nodularity or air crescent signs may be observed.
Despite its usefulness, CXR has significant limitations. It cannot reliably differentiate between benign and malignant lesions, especially in the absence of features like rib destruction. While it can identify associated findings such as pleural effusion, it does not provide enough information to determine whether these are cancer-related. For these reasons, chest X-ray serves primarily as a screening or preliminary tool, with computed tomography (CT) being essential for further characterization and accurate diagnosis.
4.1 Computed Tomography (CT)
Computed tomography (CT) remains the primary imaging modality in the evaluation of lung cancer due to its high spatial resolution and ability to provide detailed anatomical information. It plays a crucial role in detecting pulmonary nodules, characterizing tumor morphology, and assessing tumor extent. Contrast-enhanced CT is particularly useful for evaluating mediastinal structures, vascular invasion, and lymph node involvement. In addition, CT is widely used for image-guided biopsies and longitudinal monitoring of disease progression or treatment response.
4.2 Positron Emission Tomography–Computed Tomography (PET/CT)
PET/CT integrates metabolic and anatomical imaging, significantly improving diagnostic accuracy in lung cancer. Using 18F-fluorodeoxyglucose (FDG), PET/CT detects increased glucose metabolism in malignant tissues, helping differentiate benign from malignant lesions. It is highly valuable in staging, particularly for identifying nodal involvement and distant metastases. PET/CT also reduces unnecessary surgical interventions by detecting advanced disease early and plays a role in radiotherapy planning and response assessment (Owens et al., 2023; Evangelista et al., 2023).
4.3 Magnetic Resonance Imaging (MRI)
Magnetic resonance imaging (MRI) is not routinely used for initial lung cancer detection but is essential in specific clinical scenarios. Its superior soft tissue contrast makes it valuable for evaluating tumor invasion into the chest wall, mediastinum, and spine. MRI is the modality of choice for detecting brain metastases. Advanced techniques such as diffusion-weighted imaging (DWI) provide functional information about tumor cellularity and may aid in assessing treatment response.
4.4 Radiomics and Artificial Intelligence
Radiomics is an emerging field that converts medical images into quantitative data, enabling deeper analysis of tumor characteristics such as heterogeneity, texture, and shape. When combined with artificial intelligence (AI), radiomics can assist in early diagnosis, risk stratification, prediction of genetic mutations, and treatment response evaluation. These technologies hold promise for personalized medicine, although challenges related to standardization and clinical implementation remain (Tang et al., 2023; Bianconi et al., 2020).
Table 2: Comparison of Advanced Imaging Modalities in Lung Cancer
|
Modality |
Principle |
Key Role |
Advantages |
Limitations |
|
CT |
X-ray cross-sectional imaging |
Detection, staging, biopsy guidance |
High spatial resolution, widely available, fast |
Radiation exposure, limited functional data |
|
PET/CT |
Metabolic + anatomical imaging (FDG uptake) |
Staging, metastasis detection, prognosis |
Detects occult disease, improves staging accuracy |
High cost, false positives (inflammation) |
|
MRI |
Magnetic field and radiofrequency signals |
Soft tissue evaluation, brain metastasis |
Excellent soft tissue contrast, no radiation |
Limited availability, motion artifacts |
|
Radiomics/AI |
Quantitative image feature analysis |
Prediction, personalization, decision support |
Non-invasive biomarkers, precision medicine potential |
Lack of standardization, requires validation |
Radiomics and imaging biomarkers are rapidly transforming the field of lung cancer imaging by enabling a more detailed and quantitative assessment of tumors beyond what is visible to the human eye. Traditional radiological interpretation relies largely on visual evaluation of features such as size, shape, and density. In contrast, radiomics extracts a large number of quantitative features from standard imaging modalities like CT, PET/CT, and MRI, converting images into mineable data that can reveal underlying tumor biology.
Radiomic features include parameters related to tumor texture, intensity, shape, and heterogeneity, which reflect important biological characteristics such as cellular density, angiogenesis, and genetic alterations. These features can be analyzed using advanced computational methods and machine learning algorithms to assist in distinguishing benign from malignant nodules, predicting tumor aggressiveness, and assessing prognosis. Importantly, radiomics offers a non-invasive approach to capture tumor heterogeneity, which is often difficult to assess through biopsy alone due to sampling limitations29.
Imaging biomarkers derived from radiomic analysis are increasingly being explored for their role in personalized medicine. They have shown potential in predicting treatment response, particularly in patients undergoing targeted therapy or immunotherapy. For example, certain radiomic signatures from FDG PET/CT have been associated with treatment outcomes and survival in non-small cell lung cancer (NSCLC). These biomarkers may also help predict genetic mutations such as EGFR status, which is critical for selecting appropriate targeted therapies30.
Another promising application of radiomics is in early detection and risk stratification. By integrating radiomic features with clinical and demographic data, predictive models can be developed to identify high-risk individuals and guide screening strategies. Furthermore, radiomics can assist in differentiating indolent from aggressive tumors, thereby helping to reduce overdiagnosis and unnecessary interventions.
Despite its significant potential, the clinical adoption of radiomics faces several challenges. These include lack of standardization in imaging protocols, variability in feature extraction methods, and the need for large, well-validated datasets. Reproducibility and generalizability remain key concerns, and further multicenter studies are required before widespread clinical implementation can be achieved31.
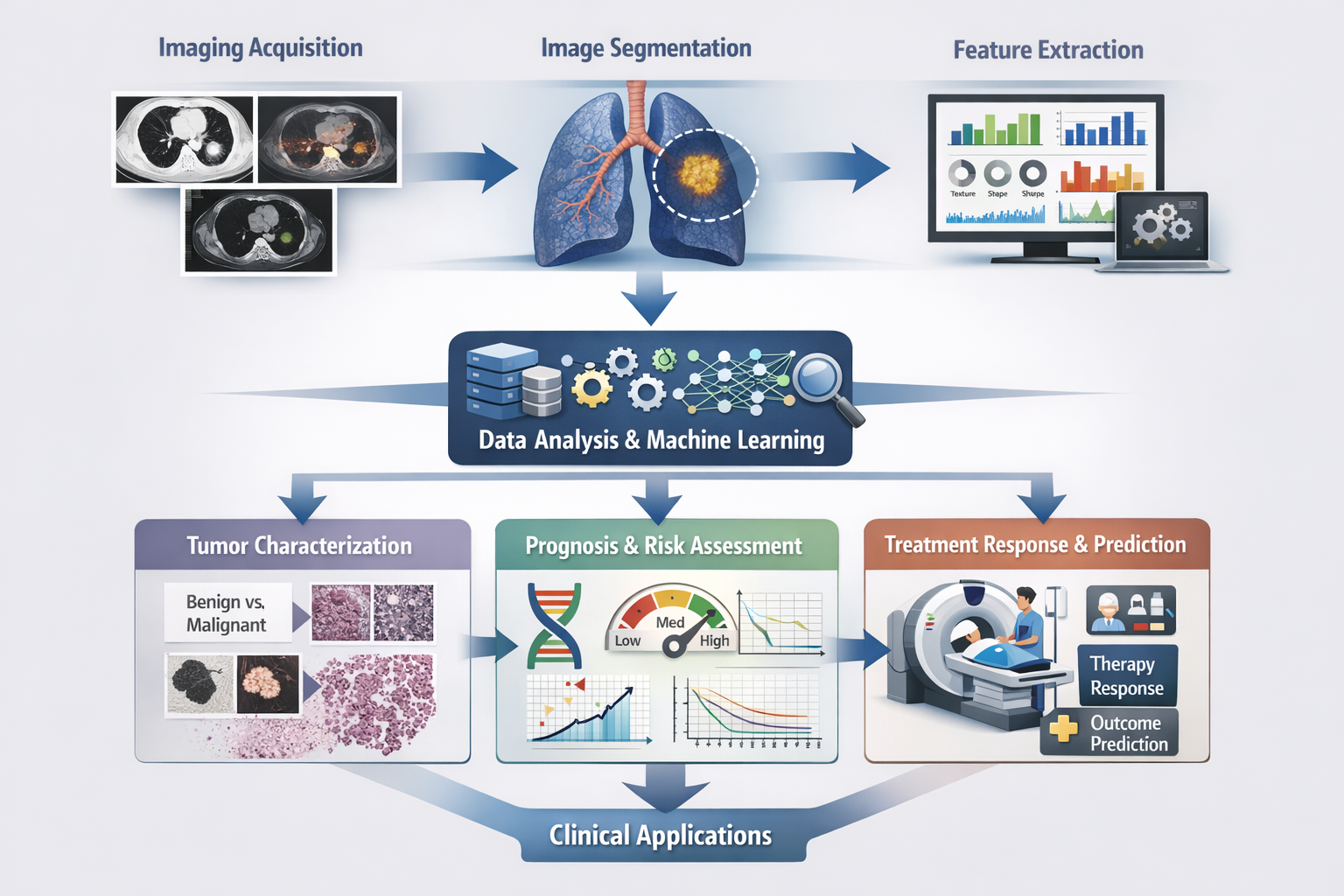
Figure 2: Radiomics workflow in lung cancer imaging.
This diagram illustrates the stepwise process of radiomics in lung cancer, beginning with image acquisition using modalities such as CT, PET/CT, or MRI, followed by tumor segmentation to define the region of interest. Quantitative features related to texture, shape, and intensity are then extracted and analyzed using machine learning algorithms. The resulting data are applied to clinical tasks, including tumor characterization (benign vs. malignant), prognosis and risk assessment, and prediction of treatment response, ultimately supporting personalized clinical decision-making.
Radiology plays a central role not only in the diagnosis of lung cancer but also in monitoring treatment response and long-term surveillance. Imaging allows clinicians to evaluate how tumors respond to therapy, detect complications, and identify disease recurrence at an early stage, thereby guiding timely clinical decisions.
Computed tomography (CT) remains the most commonly used modality for treatment monitoring due to its wide availability and high spatial resolution. Serial CT scans are used to measure changes in tumor size based on standardized criteria such as the Response Evaluation Criteria in Solid Tumors (RECIST). A reduction in tumor size typically indicates a favorable response, whereas stable or increasing size may suggest resistance or disease progression. However, size-based assessment alone has limitations, particularly with newer therapies such as immunotherapy, where atypical response patterns like pseudoprogression may occur32.
To overcome these limitations, functional imaging techniques, particularly FDG PET/CT, are increasingly used. PET/CT evaluates metabolic activity within the tumor, allowing earlier detection of treatment response compared to anatomical imaging. A decrease in FDG uptake often precedes visible size reduction, making it a valuable tool for assessing therapeutic effectiveness. PET/CT is also helpful in distinguishing viable tumor tissue from post-treatment fibrosis or necrosis, which can appear similar on CT scans33.
Magnetic resonance imaging (MRI) has a more limited but important role in treatment monitoring, especially in assessing brain metastases and soft tissue involvement. Advanced MRI techniques, such as diffusion-weighted imaging (DWI), can provide insights into tumor cellularity and may help evaluate response to therapy at an earlier stage.
Radiology is equally critical in the post-treatment surveillance phase, where the goal is to detect recurrence or metastasis as early as possible. Follow-up imaging protocols typically involve periodic CT scans, with PET/CT reserved for cases where recurrence is suspected but not clearly defined. Imaging also helps identify treatment-related complications, such as radiation-induced lung injury or chemotherapy-associated changes.
Emerging approaches, including radiomics and artificial intelligence, are further enhancing the role of imaging in treatment monitoring. These techniques can analyze subtle imaging changes over time, potentially predicting treatment response and patient outcomes more accurately than conventional methods31.
Table 3: Comparison of RECIST, iRECIST, and PERCIST Criteria in Lung Cancer
|
Criteria |
Full Form |
Imaging Modality |
Basis of Assessment |
Key Features |
Limitations |
|
RECIST 1.1 |
Response Evaluation Criteria in Solid Tumors |
CT (primarily) |
Tumor size (unidimensional measurement) |
Standardized and widely used; defines CR, PR, SD, PD based on size changes |
Does not account for metabolic activity; limited in immunotherapy (e.g., pseudoprogression) |
|
iRECIST |
Immune RECIST |
CT |
Tumor size with modified criteria for immunotherapy |
Accounts for atypical responses (e.g., pseudoprogression); requires confirmation of progression |
More complex; requires follow-up confirmation; not yet universally adopted |
|
PERCIST |
PET Response Criteria in Solid Tumors |
FDG PET/CT |
Metabolic activity (SUV changes) |
Detects early response based on tumor metabolism; useful for assessing treatment efficacy before size changes occur |
Limited availability; variability in SUV measurement; less standardized than RECIST |
Recent advances in imaging technologies are rapidly reshaping the landscape of lung cancer diagnosis and management. These innovations aim to improve early detection, enhance diagnostic accuracy, and support personalized treatment strategies through the integration of advanced computational and molecular techniques.
One of the most significant developments is the application of artificial intelligence (AI) and deep learning in medical imaging. AI algorithms can assist radiologists in detecting small pulmonary nodules, reducing interpretation time, and improving diagnostic consistency. Computer-aided detection (CAD) systems have demonstrated high sensitivity in identifying early-stage lung cancers, particularly in low-dose CT screening programs. Moreover, AI models are increasingly being used to predict malignancy risk and clinical outcomes based on imaging features34.
Another promising innovation is radiogenomics, which combines imaging data with genomic information to uncover associations between imaging phenotypes and underlying genetic mutations. This approach enables non-invasive prediction of molecular characteristics such as EGFR and ALK mutations, which are essential for targeted therapy selection. Radiogenomics has the potential to reduce the need for repeated biopsies and provide a more comprehensive understanding of tumor heterogeneity35.
Advanced functional imaging techniques are also gaining importance. Techniques such as dynamic contrast-enhanced imaging, diffusion-weighted MRI, and dual-energy CT provide additional information about tumor perfusion, cellularity, and vascularity. These methods improve lesion characterization and may allow earlier assessment of treatment response compared to conventional imaging.
The emergence of hybrid imaging modalities, such as PET/MRI, represents another important advancement. PET/MRI combines the metabolic information of PET with the superior soft tissue contrast of MRI, offering comprehensive tumor evaluation in a single examination. Although still limited in availability, this modality shows promise in staging, treatment planning, and follow-up of lung cancer patients32.
In addition, liquid biopsy imaging correlation and molecular imaging probes are being explored to enhance disease monitoring. Novel radiotracers targeting specific tumor receptors or metabolic pathways may improve the specificity of imaging and enable better assessment of tumor biology.
Despite these advancements, challenges remain, including high costs, limited accessibility, and the need for large-scale validation studies before widespread clinical adoption. Standardization of imaging protocols and integration into clinical workflows are also essential for maximizing the benefits of these technologies.
In conclusion, emerging innovations in lung cancer imaging are driving a shift toward more precise, data-driven, and personalized care. As these technologies continue to evolve, they are expected to significantly improve early detection, treatment planning, and patient outcomes.
Despite major advances in imaging technologies, several challenges and limitations remain in the use of radiology for the detection and management of lung cancer. Issues such as false-positive findings, radiation exposure, limited accessibility to advanced imaging modalities, and ethical concerns related to artificial intelligence (AI) continue to influence the effectiveness and implementation of radiological techniques in clinical practice.
8.1 False Positives and Overdiagnosis
One of the major limitations of lung cancer screening, particularly with low-dose computed tomography (LDCT), is the occurrence of false-positive results. Small pulmonary nodules detected on imaging may not always represent malignant lesions and can instead be caused by benign conditions such as infections, inflammatory diseases, or granulomas. These false-positive findings may lead to unnecessary follow-up imaging, invasive diagnostic procedures, patient anxiety, and increased healthcare costs. In addition, overdiagnosis may occur when slow-growing or indolent tumors are detected that might not have caused clinical symptoms or affected patient survival if left undetected.
8.2 Radiation Exposure Concerns
Radiological imaging techniques, particularly computed tomography, involve exposure to ionizing radiation. Although the radiation dose in low-dose CT screening is significantly lower than that of standard CT scans, repeated imaging during screening programs or follow-up assessments may increase cumulative radiation exposure. This raises concerns about the potential long-term risk of radiation-induced malignancies, especially in populations undergoing regular screening. Therefore, balancing the benefits of early detection with the risks associated with radiation exposure remains an important consideration in lung cancer screening programs.
8.3 Accessibility of Advanced Imaging
Access to advanced imaging technologies such as PET-CT, high-resolution CT, and MRI can be limited in many regions, particularly in low- and middle-income countries. These modalities require specialized equipment, trained personnel, and substantial financial resources, which may restrict their availability in resource-limited healthcare settings. As a result, disparities in access to diagnostic imaging can lead to delays in diagnosis and treatment, ultimately affecting patient outcomes.
8.4 Ethical and Regulatory Issues of Artificial Intelligence
The integration of artificial intelligence (AI) in radiology has the potential to improve the detection and characterization of lung nodules; however, it also raises several ethical and regulatory concerns. Issues related to data privacy, algorithm transparency, bias in training datasets, and accountability for diagnostic errors remain important challenges. In addition, the clinical implementation of AI-based tools requires strict regulatory approval, validation across diverse populations, and continuous monitoring to ensure reliability and patient safety. Addressing these concerns is essential for the responsible and effective use of AI in lung cancer imaging.
FUTURE PERSPECTIVES
Rapid advancements in radiological technology are expected to significantly improve the early detection and management of lung cancer in the coming years. Emerging innovations such as artificial intelligence (AI), radiomics, and radiogenomics are increasingly being integrated into thoracic imaging to enhance diagnostic accuracy and enable personalized treatment strategies. AI-based algorithms can assist radiologists in detecting subtle pulmonary nodules, reducing diagnostic errors, and improving the efficiency of lung cancer screening programs. Radiomics, which involves the extraction of quantitative imaging features from CT or PET scans, has shown promising potential for predicting tumor behavior, treatment response, and patient prognosis. Furthermore, the integration of imaging data with genomic and clinical information may support precision medicine approaches and facilitate individualized treatment planning. Future research should focus on the development of standardized imaging protocols, validation of AI-based tools through large multicenter studies, and the establishment of clear regulatory frameworks to ensure safe and ethical clinical implementation.
CONCLUSION
In conclusion, radiology plays a critical role in the early detection, diagnosis, staging, treatment planning, and follow-up of lung cancer. Imaging modalities such as low-dose computed tomography (LDCT), PET-CT, MRI, and advanced imaging techniques have significantly improved the ability to identify lung cancer at earlier stages, thereby enhancing survival outcomes. Despite current challenges including false positives, radiation exposure, and limited access to advanced imaging technologies in certain regions, continuous technological progress and interdisciplinary collaboration are likely to overcome these barriers. The incorporation of advanced imaging analytics, AI-assisted diagnostics, and precision imaging strategies will further transform the role of radiology in lung cancer care, ultimately contributing to earlier diagnosis, improved treatment outcomes, and reduced mortality.
AUTHORS CONTRIBUTION
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be an author as per the international committee of medical journal editors (ICMJE) requirements/guidelines.
ACKNOWLEDGMENT
It’s our privilege to express profound sense of gratitude and cordial thanks to our respected chairman Mr. Anil Chopra, Vice Chairperson Ms. Sangeeta Chopra, St. Soldier Educational Society, Jalandhar for providing the necessary facilities to complete this work.
CONFLICTS OF INTEREST
No Conflict of interest
REFERENCES




Aruna Kumari, Dr. Saruchi, Dr. Ajeet Pal Singh and Dr. Amar Pal Singh, Role Of Radiology in Early Detection and Management of Lung Cancer, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 3413-3427, https://doi.org/10.5281/zenodo.19238122
 10.5281/zenodo.19238122
10.5281/zenodo.19238122