
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Smt. BNB. Swaminarayan Pharmacy College, affiliated to Gujarat Technological University, India
The development of robust, efficient, and reliable analytical methods is essential in modern pharmaceutical research and quality assurance. Traditional analytical method development is often time-consuming, resource-intensive, and largely dependent on trial-and-error experimentation. In recent years, Artificial Intelligence (AI) has emerged as a powerful approach to address these limitations in pharmaceutical analytics. AI-based techniques, including machine learning (ML), artificial neural networks (ANN), and deep learning (DL), enable rapid data analysis, pattern recognition, and predictive modeling for analytical method optimization. These tools are increasingly applied to optimize chromatographic parameters, predict retention behavior, improve peak resolution, and enhance method robustness while reducing the number of experimental trials. By learning from historical experimental data, AI systems support data-driven decision-making and continuous refinement of analytical conditions. As a result, AI-assisted method development can significantly reduce development time, solvent consumption, and human effort while improving analytical performance and reproducibility. This review discusses the fundamental concepts, applications, advantages, and future potential of AI-driven analytics in pharmaceutical analytical method development.
High-Performance Liquid Chromatography (HPLC) stands as one of the most extensively employed analytical methodologies within the pharmaceutical sector, attributable to its exceptional sensitivity, specificity, and adaptability in the examination of drug compounds, impurities, and finalized pharmaceutical products. The development of analytical methods constitutes a pivotal component in pharmaceutical research, quality control (QC), and quality assurance (QA), ensuring the precision, dependability, and reproducibility of analytical outcomes throughout the lifecycle of drug products.(1-2)
Conventionally, the development of HPLC methods has predominantly depended on empirical strategies and iterative trial-and-error experimentation. Analytical chemists manually adjust chromatographic parametersvincluding mobile phase composition, pH, flow rate, column chemistry, temperature, and detection wavelength through numerous experimental iterations. Despite advancements in chromatographic instrumentation and column technologies that have enhanced analytical capabilities, the overall method development process remains labor-intensive, time-consuming, and heavily reliant on the expertise of the analyst.(3)
The escalating complexity of pharmaceutical molecules, coupled with stringent regulatory mandates and the imperative for accelerated development timelines, has underscored the limitations inherent in traditional analytical method development approaches. These challenges have engendered a demand for more efficient, data-driven methodologies capable of managing complex analytical datasets and optimizing chromatographic conditions while minimizing experimental workload.
In this context, Artificial Intelligence (AI) has emerged as a promising avenue for contemporary pharmaceutical analytics. AI methodologies, including machine learning (ML), artificial neural networks (ANN), and deep learning (DL), facilitate the analysis of intricate experimental data, the elucidation of latent relationships among analytical variables, and the prediction of chromatographic behavior. By leveraging historical and experimental datasets, AI models can enable predictive optimization of analytical parameters, thereby substantially reducing the requisite number of experimental trials during method development.
The incorporation of AI into pharmaceutical analytical workflows signifies a paradigm shift from experience-driven experimentation toward predictive, data-centric method development. This review endeavors to elucidate the foundational principles of AI, explore its applications in pharmaceutical analytical method development, examine the benefits and challenges associated with AI-driven analytics, and consider the prospective advancements of intelligent analytical systems within pharmaceutical research.(4)
2. Fundamentals of Artificial Intelligence in Pharmaceutical Analytics
The escalating complexity of pharmaceutical analytical data necessitates the deployment of sophisticated computational methodologies capable of extracting substantive insights from extensive experimental datasets. Artificial Intelligence (AI) offers a robust framework for the analysis of intricate data derived from chromatographic, spectroscopic, and mass spectrometric techniques. Within the domain of pharmaceutical analytics, AI facilitates the identification of patterns, the modeling of interrelationships among analytical variables, and the prediction of optimal experimental parameters to enhance analytical method development. AI encompasses computational systems engineered to execute tasks traditionally requiring human cognitive functions, such as pattern recognition, data-driven learning, and predictive decision-making. Specifically, in pharmaceutical analytics, AI supports the processing and interpretation of complex experimental data generated during method development, thereby enabling data-driven optimization of analytical parameters.
Among the diverse subfields of AI, machine learning, artificial neural networks, and deep learning represent the most prevalently employed methodologies in pharmaceutical analytical research, owing to their capacity to model complex interactions between analytical variables and experimental outcomes.
1. Machine Learning (ML): Machine learning constitutes a fundamental aspect of AI, empowering computational systems to discern patterns and relationships directly from data and to generate predictions without explicit programming. In the context of pharmaceutical analytical method development, ML serves as a pivotal data-analytic tool for modeling the relationships between experimental input variables such as mobile phase composition, pH, flow rate, temperature, and column characteristics and analytical responses, including retention time, resolution, and peak morphology. The ML workflow commences with the acquisition of experimental data produced during method development, followed by data preprocessing and feature selection. Subsequently, the refined dataset is employed to train an ML model, which internalizes the underlying patterns and correlations inherent in the data. Upon completion of training, the model undergoes validation and can be utilized to predict optimal analytical conditions, thereby minimizing reliance on empirical trial-and-error approaches and enhancing the efficiency of method development. In pharmaceutical method development, ML models are increasingly applied to forecast chromatographic retention times, optimize mobile phase compositions, and identify critical analytical variables that influence separation performance.(5)
2. Artificial Neural Networks (ANN): Artificial Neural Networks (ANNs) are computational frameworks modeled after the neural architecture of the human brain. These networks are especially proficient in capturing non-linear relationships between analytical variables and their corresponding responses. In the field of pharmaceutical analytics, ANNs are extensively utilized to optimize chromatographic conditions, where multiple factors concurrently affect separation efficiency. Structurally, ANNs comprise input layers that represent experimental variables, hidden layers that process intricate interactions, and output layers that produce predicted analytical results. Their capacity to manage noisy and complex datasets enables ANNs to achieve superior predictive accuracy relative to traditional regression techniques. Consequently, owing to their robust ability to model complex and non-linear interactions, ANNs are particularly advantageous in chromatographic method development, where various analytical parameters simultaneously impact retention behavior, peak morphology, and resolution.(6-7)
3. Deep Learning and Advanced AI Models: Deep learning represents a sophisticated subset of machine learning characterized by the use of multiple hidden layers to derive high-level features from extensive datasets. While its application in pharmaceutical analytics remains in the nascent stages, deep learning models demonstrate considerable potential in managing the large volumes of analytical data produced by contemporary instruments and automated laboratory systems. These models possess the capability to discern complex relationships among chromatographic variables, thereby facilitating real-time decision-making during method development processes. The adoption of deep learning techniques is anticipated to expand in tandem with advancements in computational resources and the increased availability of high-quality analytical data. Furthermore, deep learning methodologies exhibit promise for the automated interpretation of extensive analytical datasets, encompassing spectral data analysis and the recognition of intricate chromatographic patterns within modern pharmaceutical laboratories.(8)
3. Digital Libraries and Software Tools in AI-Driven Pharmaceutical Analytics
Table 1: Summarizes commonly used software platforms that support AI-driven data analysis and optimization in pharmaceutical analytical method development.
|
Software |
AI / Analytics Role (ML / ANN / DL) |
Application Area |
Method Development Benefits |
|
MATLAB® |
ML, ANN, DL modeling |
Analytical data modeling and prediction |
Development of predictive models, handling non-linear chromatographic relationships, optimization of analytical parameters |
|
Python (scikit-learn, TensorFlow, Keras) |
ML, ANN, DL |
Advanced pharmaceutical data analytics |
Customizable machine learning models, deep learning based prediction, integration of digital libraries |
|
R |
ML, ANN, DL |
Statistical and AI-driven analytics |
Data preprocessing, predictive modeling, visualization of analytical trends |
|
Minitab® |
ML-assisted statistics |
Experimental data analysis |
Statistical modeling, trend analysis, support for data-driven optimization |
|
Design-Expert® |
ML-assisted optimization |
Experimental design and optimization |
Reduced experimental trials, systematic evaluation of analytical parameters |
|
Empower® |
AI-assisted analytics |
Chromatographic data acquisition and processing |
Reliable data generation, improved peak integration, structured datasets for AI modeling |
|
Chromeleon® |
AI-assisted analytics |
Chromatographic data management |
High-quality data acquisition, support for advanced data analysis |
4. Role of AI in Pharmaceutical Analytical Method Development
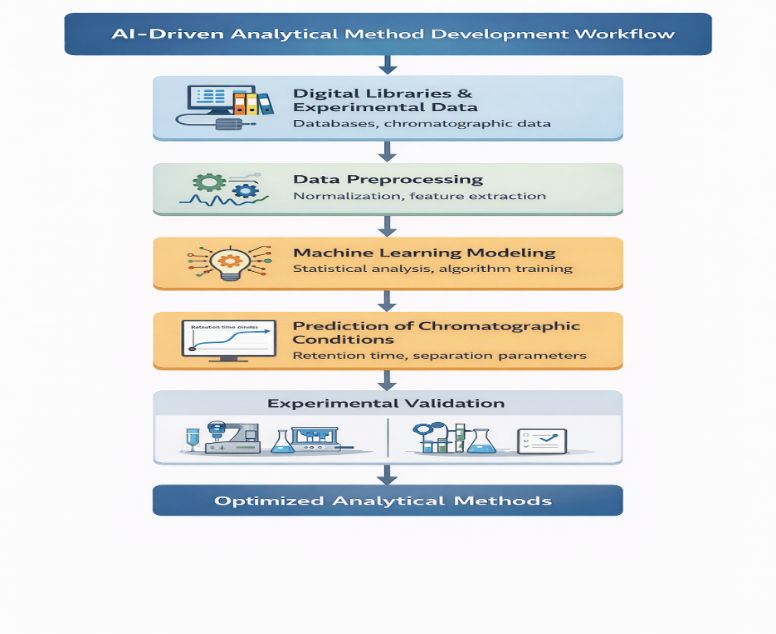
Figure 1: AI-Driven Analytical Method Development Workflow
1. Optimization of Chromatographic Parameters:
Artificial intelligence (AI) methodologies, including Machine Learning (ML), Artificial Neural Networks (ANN), and Deep Learning (DL), have proven particularly effective in refining chromatographic variables such as mobile phase composition and pH, flow rate and temperature, as well as column selection and particle size. By leveraging historical experimental datasets and identifying patterns within analytical results, AI models can forecast the optimal combination of method parameters to achieve targeted separation, retention, and resolution. Machine learning algorithms have been successfully employed to predict chromatographic retention times and optimize mobile phase compositions, thereby facilitating more rapid determination of appropriate separation conditions during analytical method development. Empirical evidence from multiple studies underscores the efficacy of AI approaches in this domain. For instance, Xu et al. (2023) developed a machine learning model that utilized molecular descriptors alongside chromatographic data to predict the retention times of chiral compounds in liquid chromatography. This model accurately anticipated retention behavior and aided in the identification of optimal separation conditions, substantially decreasing the number of experimental trials necessary during method development.
2. Reduction in the Number of Experimental Trials:
Artificial intelligence-driven analytics substantially decreases the quantity of experimental trials necessary during method development. Predictive models constructed using machine learning (ML) or artificial neural networks (ANN) can anticipate analytical responses under varying conditions, thereby reducing the reliance on exhaustive laboratory testing. This approach not only conserves time and resources but also improves methodological efficiency and promotes sustainability. By leveraging previously acquired experimental datasets, AI models are capable of computationally simulating multiple analytical scenarios, which further diminishes the demand for extensive empirical experimentation.
3. Addressing Complex and Non-linear Interactions:
Numerous analytical systems exhibit complex, non-linear interdependencies among variables, such as the interplay between buffer pH, flow rate, and column characteristics. Conventional statistical techniques often prove inadequate in capturing these intricate relationships. For instance, D’Archivio et al. (2019) employed an ANN model to predict the retention behavior of ionizable compounds in reversed-phase high-performance liquid chromatography (HPLC) across diverse chromatographic conditions. Their findings demonstrated that neural network models can effectively characterize the complex associations between chromatographic parameters and retention behavior. AI methodologies, particularly ANNs and deep learning algorithms, excel in discerning non-linear patterns within large datasets, thereby enabling precise predictions of method performance under varied experimental conditions. Specifically, artificial neural networks are adept at modeling non-linear interactions among chromatographic variables such as pH, solvent composition, and flow rate, which collectively influence separation efficiency.
4. Integration with Digital Libraries and Data Management Systems:
AI models depend on access to high-quality, well-structured data derived from both experimental records and digital repositories. Chromatographic data systems (CDS) such as Empower®, Chromeleon®, and OpenLab® furnish raw analytical data, while chemometric and statistical software tools facilitate data preprocessing and organization. The integration of these resources with AI technologies enables automated data analysis, real-time predictive capabilities, and intelligent optimization of analytical methods. This synergy between AI, chromatographic data systems, and digital analytical libraries supports the automated processing of extensive datasets, thereby enhancing the accuracy of predictions and the refinement of analytical conditions.(11-12)
5. Advantages of AI-Driven Analytics
Artificial Intelligence (AI) has markedly advanced the development of pharmaceutical analytical methods by offering intelligent, data-driven solutions that overcome the inherent limitations of traditional trial-and-error methodologies. The incorporation of AI technologies into analytical workflows confers multiple benefits that enhance efficiency, reliability, and overall analytical performance.
A. Accelerated Method Development: AI-driven predictive models can analyze both historical and experimental datasets to determine optimal chromatographic parameters, including mobile phase composition, pH, and flow rate. This capability substantially expedites the method development process relative to conventional experimental optimization techniques.
B. Decreased Experimental Burden: Machine learning algorithms and neural network models enable the computational simulation of numerous analytical conditions, thereby diminishing the necessity for extensive laboratory experimentation. This reduction leads to lower reagent consumption, decreased instrument utilization, and minimized overall experimental workload.
C. Enhanced Precision and Reproducibility: AI algorithms facilitate the identification of critical analytical variables that impact method performance. By forecasting the effects of these parameters, AI-assisted methods exhibit improved reproducibility and reduced analytical variability.
D. Data-Driven Decision Making: AI empowers analytical scientists to base their decisions on patterns discerned from large-scale datasets rather than relying exclusively on empirical experience. This approach strengthens the reliability and scientific rigor underpinning analytical method development.
E. Deeper Insight into Complex Analytical Systems: Pharmaceutical analytical systems frequently involve intricate interactions among multiple variables. AI models, particularly those employing machine learning and artificial neural networks, are capable of capturing non-linear relationships among chromatographic parameters, thereby providing enhanced understanding of system behavior and facilitating more robust method optimization.
F. Improved Method Robustness: AI-supported analytical strategies assist in identifying critical method parameters and their interactions, enabling the formulation of more robust analytical methods that maintain reliability despite minor variations in experimental conditions.(13-14-15)
6. Challenges and Limitations of AI-Driven Analytics
While the integration of Artificial Intelligence (AI) into pharmaceutical analytical method development offers considerable benefits, it also introduces several challenges that must be meticulously addressed to ensure effective implementation.
I. Data Quality and Availability: The performance of AI models is highly dependent on the quality and volume of input data. In the context of pharmaceutical analytics, datasets that are incomplete, inconsistent, or contain noise can substantially compromise model accuracy and predictive reliability. Consequently, the generation of high-quality experimental data is critical for the development of robust AI-based analytical models.
II. Necessity for Large Datasets: Many AI methodologies, particularly machine learning and deep learning algorithms, demand extensive datasets to facilitate effective model training. Within analytical method development, the production of sufficiently large and diverse experimental datasets can be both time-consuming and resource-intensive.
III. Model Interpretability: Advanced AI models, notably deep learning architectures, often operate as “black-box” systems, wherein the internal decision-making processes are not readily interpretable. This opacity poses challenges for the validation of analytical models and the elucidation of results, especially within the highly regulated pharmaceutical sector.
IV. Limited Regulatory Familiarity: The incorporation of AI into pharmaceutical analytical workflows introduces regulatory complexities. Regulatory bodies mandate that analytical methods be thoroughly understood, validated, and reproducible. The deployment of sophisticated AI models may necessitate additional validation procedures and the development of specific regulatory guidelines.
V. Requirement for Skilled Personnel: The successful adoption of AI-driven analytical approaches requires expertise spanning analytical chemistry, data science, statistics, and computational modeling. The scarcity of professionals possessing such interdisciplinary skills may impede the widespread implementation of AI technologies in pharmaceutical laboratories.(16-17-18)
7. FUTURE PERSPECTIVES
CONCLUSION
REFERENCES



Hiral Bhavsar, Shailesh Luhar, Sachin Narkhede, Artificial Intelligence Applications in Pharmaceutical Analytical Method Development, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 2601-2610. https://doi.org/10.5281/zenodo.19182269
 10.5281/zenodo.19182269
10.5281/zenodo.19182269