
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department Of Pharmaceutics, Durgamata Institute Of Pharmacy, Parbhani, India.
Objective -To formulate and optimize ketoconazole-loaded liposomes for topical application.Methods – To create a consistent, stable, and nanoscale vesicular system for improved topical distribution, ketoconazole-loaded liposomes were made by thin-film hydration and sonicationResult -The marketed product and the in vitro drug release profiles of ketoconazole-loaded liposomal gel formulations (G1–G10) were compared. Consistent with controlled release behavior, all formulations showed a progressive increase in drug release over time. Effective drug diffusion from the gel matrix was demonstrated by the developed formulations’ total drug release, which varied from 92.3% to 100% over 180 minutes.Conclusion - The successful integration of ketoconazole into the lipid bilayer was confirmed by the improved formulation’s effective drug encapsulation.
An innovative topical medication delivery method called ketoconazole-loaded liposomal gel was created to increase the therapeutic effectiveness of ketoconazole in the management of fungal infections.A common treatment for superficial mycoses like dermatophytosis, candidiasis, and seborrheic dermatitis is ketoconazole, a broad-spectrum imidazole antifungal medication. However, because of its weak water solubility, poor skin penetration, and brief residence time at the application site, its therapeutic efficacy in traditional topical formulations (such as creams and ointments) is frequently limited.Lipophilic and hydrophilic medicines can be encapsulated in liposomal drug delivery systems, which are vesicular carriers made of phospholipid bilayers. When ketoconazole is incorporated into liposomes, it becomes more soluble and has better stratum corneum penetration. Additionally, liposomes serve as drug reservoirs, improving antifungal efficacy while reducing systemic absorption and related side effects by enabling sustained release and better localization of the medication within the skin layers.Because of its broad-spectrum antifungal action and efficacy against a variety of pathogenic fungi, such as dermatophytes, yeasts, and molds, ketoconazole is an important part of the treatment of fungal infections. It works by preventing the creation of ergosterol, a crucial part of the fungal cell membrane, and is a member of the imidazole class of antifungal drugs. Ketoconazole is quite efficient in treating superficial mycoses because this impairment in ergosterol biosynthesis causes increased membrane permeability, cellular content leakage, and finally fungal cell death.Liposomes also enhance drug penetration through the skin by interacting with the lipid Components of the stratum corneum, thereby improving drug permeation and accumulation in Deeper skin layers. This is particularly beneficial in topical antifungal therapy, where effective Drug delivery to the site of infection is essential. Furthermore, liposomes can provide targeted Drug delivery, reducing off-target effects and minimizing systemic absorption, which enhances the Safety profile of the drug.To improve therapeutic efficacy, the idea of a liposomal gel combines the benefits of liposomal drug delivery systems with the desired qualities of topical gel formulations. Gels offer an appropriate basis for simple topical delivery, while liposomes are vesicular carriers made of phospholipid bilayers that can encapsulate both hydrophilic and lipophilic medicines. Liposomes are incorporated into a gel matrix to create a dual-controlled delivery system that increases skin penetration, prolongs drug retention at the application location, and improves drug stability.Furthermore, liposomal gels filled with ketoconazole improve therapeutic efficacy while minimizing side effects. The formulation lowers the risk of both local irritation and systemic toxicity by localizing the medicine within the skin layers and decreasing systemic absorption. Better clinical results and a lower risk of recurrence result from the enhanced antifungal efficacy brought about by the enhanced targeting and sustained release.
MATERIALS AND METHODS
Materials
|
Material |
Category |
Purpose/Use |
Source/Supplier |
|
|
1 |
Ketoconazole |
Active Pharmaceutical Ingredient (API) |
Antifungal drug |
Pharma company |
|
2 |
Phosphatidylcholine |
Lipid |
Formation of liposomal bilayer |
Sigma-Aldrich |
|
3 |
Cholesterol |
Lipid stabilizer |
Provides rigidity to liposomal membrane |
Loba Chemie |
|
4 |
Carbopol 934 |
Gelling agent |
Preparation of gel base |
Loba Chemie |
|
5 |
Hydroxypropyl Methylcellulose (HPMC) |
Gelling agent |
Alternative gel base polymer |
HiMedia |
|
6 |
Triethanolamine |
Neutralizing agent |
pH adjustment and gel formation |
Merck |
|
7 |
Glycerin |
Humectant |
Improves moisture retention and spreadability |
Loba Chemie |
|
8 |
Chloroform |
Organic solvent |
Lipid dissolution (thin film formation) |
Merck |
|
9 |
Methanol |
Organic solvent |
Co-solvent for lipid and drug |
Merck |
|
10 |
Phosphate buffer (pH 7.4) |
Buffer solution |
Hydration medium and dissolution studies |
Prepared in laboratory |
|
11 |
Distilled water |
Vehicle |
Preparation of formulations |
Laboratory grade |
|
12 |
Dialysis membrane |
Semi-permeable membrane |
In vitro drug release studies |
HiMedia |
|
13 |
Nutrient agar / Sabouraud agar |
Culture media |
Antifungal activity studies |
HiMedia |
|
14 |
Fungal strains (C. albicans, A. niger) |
Test organisms |
Evaluation of antifungal activity |
Microbiology laboratory |
Collection of Material
The active pharmaceutical ingredient (API) for this investigation was ketoconazole, which was obtained as a gift sample from a reputable pharmaceutical manufacturing company. The phospholipids needed to make liposomes, like cholesterol and phosphatidylcholine, were purchased from reputable commercial vendors and utilized without additional purification. For the creation of the topical gel basis, gel-forming polymers such as hydroxypropyl methylcellulose (HPMC) and carbopol were chosen due to their appropriateness for cutaneous application, biocompatibility, and capacity to change viscosity. All other excipients were obtained from standard chemical sources and were of analytical grade, including organic solvents (methanol and chloroform), preservatives, buffering agents, and analytical-grade reagents. Throughout the investigation, double-distilled water was utilized for both formulation and analysis. Unless otherwise noted, all materials were used exactly as they were received.
Drug Characterization
In order to gather preliminary data about ketoconazole’s identity, purity, and suitability for formulation development, its physical appearance was assessed as a crucial preformulation characteristic. The drug sample’s color, odor, and physical condition were all closely inspected visually in daylight. Since this could affect the medicine’s solubility and formulation behavior, special attention was paid to determining whether the drug was crystalline or amorphous.
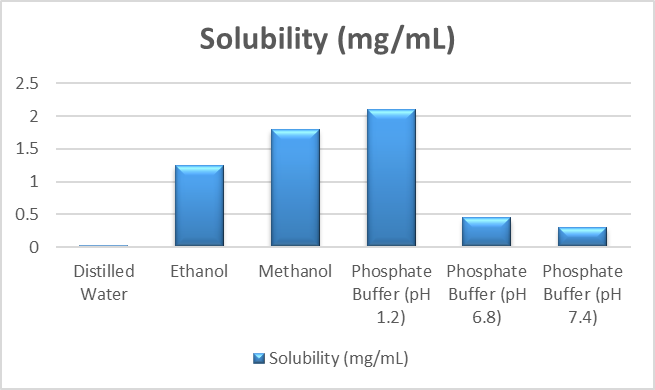
Ketoconazole’s solubility was identified as a crucial preformulation metric to comprehend its dissolution behavior and to help choose appropriate solvents and formulation ingredients. Distilled water, ethanol, methanol, and phosphate buffer solutions with varying pH values (1.2, 6.8, and 7.4) were among the solvents to which an excess of ketoconazole was added.
Formulation Development
1 Preparation of Ketoconazole-Loaded Liposomes
To create a consistent, stable, and nanoscale vesicular system for improved topical distribution, ketoconazole-loaded liposomes were made by thin-film hydration and sonication.This approach involved dissolving precisely weighed volumes of cholesterol and phosphatidylcholine in a round-bottom flask using an appropriate organic solvent system (chloroform:methanol in the right ratio).To guarantee consistent entrapment within the lipid bilayer, ketoconazole was added to the lipid phase. In order to achieve desired properties like smaller particle size, narrow polydispersity index (PDI), higher entrapment efficiency, and sufficient zeta potential, liposomes were optimized by adjusting the lipid composition (phosphatidylcholine and cholesterol ratio) and drug-to-lipid ratio.Based on its overall physicochemical performance, the improved formulation was chosen to be further included into the gel foundation.
|
Formulation Code |
Ketoconazole (mg) |
Phosphatidylcholine (mg) |
Cholesterol (mg) |
Drug:Lipid Ratio |
PC:Chol Ratio |
|
F1 |
50 |
100 |
20 |
1:2.4 |
5:1 |
|
F2 |
50 |
120 |
20 |
1:2.8 |
6:1 |
|
F3 |
50 |
140 |
20 |
1:3.2 |
7:1 |
|
F4 |
50 |
100 |
40 |
1:2.8 |
2.5:1 |
|
F5 |
50 |
120 |
40 |
1:3.2 |
3:1 |
|
F6 |
50 |
140 |
40 |
1:3.6 |
3.5:1 |
|
F7 |
50 |
100 |
60 |
1:3.2 |
1.6:1 |
|
F8 |
50 |
120 |
60 |
1:3.6 |
2:1 |
|
F9 |
50 |
140 |
60 |
1:4.0 |
2.3:1 |
|
F10 |
50 |
160 |
60 |
1:4.4 |
2.6:1 |
Composition of Ketoconazole-Loaded Liposomal Formulations (F1–F10)
Characterization of Liposomes
Particle size and Polydispersity Index (PDI)
In order to assess the vesicle size distribution and homogeneity of the formulation—two crucial factors affecting drug release, skin penetration, and overall system stability—the particle size and polydispersity index (PDI) of ketoconazole-loaded liposomes were measured. The measurements were performed under regulated settings utilizing a particle size analyzer and the dynamic light scattering (DLS) technique.
Zeta potential
A zeta potential analyzer based on the electrophoretic light scattering method was used to do the measurements. The liposomal dispersion was put into a transparent foldable capillary cell for examination after being appropriately diluted with distilled water or a suitable buffer to prevent multiple scattering effects. The electrophoretic mobility of the vesicles was determined after the sample was exposed to an electric field, and the instrument software was then used to transform the results into zeta potential values.
Entrapment efficiency
The amount of drug effectively contained inside the vesicular lipid bilayer in relation to the total drug employed in the formulation was measured using the entrapment efficiency of ketoconazole-loaded liposomes. It is a crucial factor that directly represents the liposomes’ loading capacity and affects the system’s stability, sustained release behavior.
Composition of Ketoconazole Liposomal Gel Formulations (G1–G10)
|
Formulation Code |
Optimized Liposomes (equivalent to 1% drug) |
Carbopol 934 (% w/w) |
HPMC (% w/w) |
Triethanolamine (q.s.) |
Glycerin (% w/w) |
Distilled Water (q.s.) |
|
G1 |
Equivalent to 1% Ketoconazole |
0.5 |
— |
q.s. |
5 |
100% |
|
G2 |
Equivalent to 1% Ketoconazole |
1.0 |
— |
q.s. |
5 |
100% |
|
G3 |
Equivalent to 1% Ketoconazole |
1.5 |
— |
q.s. |
5 |
100% |
|
G4 |
Equivalent to 1% Ketoconazole |
2.0 |
— |
q.s. |
5 |
100% |
|
G5 |
Equivalent to 1% Ketoconazole |
0.75 |
— |
q.s. |
5 |
5 |
|
G6 |
Equivalent to 1% Ketoconazole |
— |
0.5 |
— |
5 |
100% |
|
G7 |
Equivalent to 1% Ketoconazole |
— |
1.0 |
— |
5 |
100% |
|
G8 |
Equivalent to 1% Ketoconazole |
— |
1.5 |
— |
5 |
100% |
|
G9 |
Equivalent to 1% Ketoconazole |
— |
2.0 |
— |
5 |
100% |
|
G10 |
Equivalent to 1% Ketoconazole |
0.5 |
0.5 |
q.s. |
5 |
100% |
Evaluation of Liposomal Gel
Physicochemical Properties
Appearance, color, homogeneity
The generated ketoconazole-loaded liposomal gel’s appearance, color, and homogeneity were assessed as the main physicochemical parameters to gauge its general quality, consistency, and patient acceptability. Under normal lighting, the formulation’s physical characteristics, such as texture, smoothness, and the existence of any particle matter or phase separation, were visually examined. The gel was carefully examined to make sure there were no lumps, grittiness, or indications of liposomal vesicle aggregation.
pH measurement
The pH of the ketoconazole-loaded liposomal gel was determined as an important physicochemical Parameter to ensure skin compatibility, stability of the formulation, and prevention of irritation upon Topical application. Since the normal skin pH ranges from approximately 4.5 to 6.5, it is essential that The prepared formulation maintains a pH.
Viscosity
To assess the ketoconazole-loaded liposomal gel’s rheological behavior, consistency, and ease of skin application, its viscosity was measured. Viscosity is a crucial factor for topical formulations since it directly affects patient acceptance, spreadability, and retention time at the application site. An ideal viscosity guarantees that the gel is both easily spreadable during application and stable during storage.
In vitro Drug Release
To assess the drug’s release profile from the created formulation and compare it with a traditional ketoconazole gel, an in vitro drug release study of ketoconazole-loaded liposomal gel was carried out. This research is crucial to comprehending how liposomal encapsulation affects drug release behavior and verifying the system’s capability for sustained release.A suitable diffusion method, such as a dialysis membrane diffusion technique, was used to conduct the release investigation. A Franz diffusion cell’s donor and receptor sections were separated by a pre-treated dialysis membrane.
RESULT AND DISCUSSION
Drug Characterization
Physical appearance
The physical appearance of ketoconazole was evaluated as a preliminary step in drug Characterization. The drug was observed to be a white to off-white, odorless, crystalline powder With a fine and uniform texture.
|
Parameter |
Observation |
|
Color |
White to off-white |
|
Odor |
Odorless |
|
Physical State |
Crystalline powder |
|
Texture |
Fine, non-gritty |
|
Impurities |
No visible impurities observed |
Solubility
To comprehend ketoconazole’s dissolving behavior, its solubility profile was assessed in a variety of solvents and buffer systems. The drug’s extremely low solubility in distilled water confirmed that it is hydrophobic. It did, however, exhibit far greater solubility in organic solvents like ethanol and methanol, suggesting a strong preference for organic phases. In an acidic medium (pH 1.2), there was a noticeable increase in solubility, whereas at higher pH values (6.8 and 7.4), there was a significant drop.
Particle size
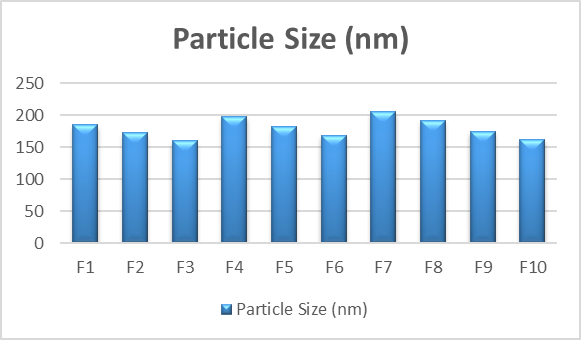
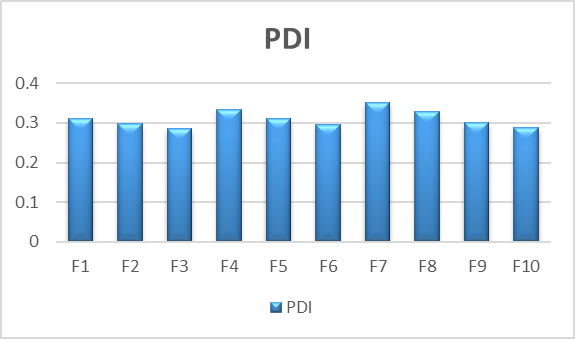
The polydispersity index (PDI) and particle size of liposomes loaded with ketoconazole were measured. Dynamic light scattering was used to measure the particle size and polydispersity index (PDI) of ketoconazole-loaded liposomes (F1–F10) in order to assess vesicle size distribution and homogeneity. The formulations’ particle sizes, which varied from 160.3 to 205.6 nm, showed that nanosized liposomes appropriate for topical medication administration had been successfully formed. As seen in formulations F3 and F10, smaller vesicle sizes are beneficial for increased skin penetration and better medicinal efficiency. The PDI values showed a somewhat uniform to uniform size distribution among the formulations, ranging from 0.285 to 0.352.
pH Measurement
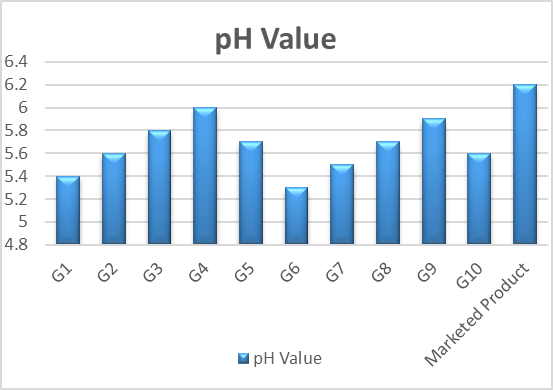
The pH of ketoconazole-loaded liposomal gel formulations (G1–G10) and the marketed product Was evaluated to ensure skin compatibility. The pH values of the developed formulations ranged From 5.3 to 6.0, falling within the normal skin pH range (4.5–6.5), indicating suitability for topical Application. Formulations G3 and G10 showed pH values closest to ideal skin conditions, Suggesting better compatibility. Minor variations in pH were due to differences in polymer Concentration and neutralization, especially in Carbopol-based gels.
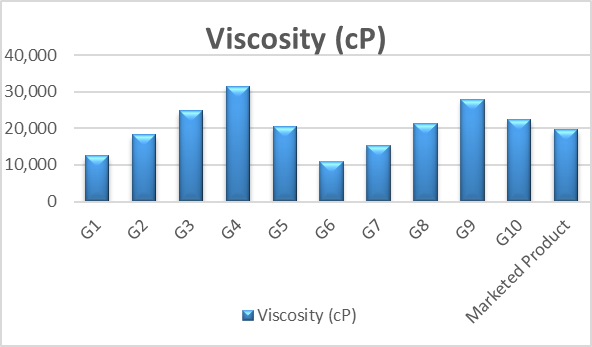
Viscosity
The viscosity of ketoconazole-loaded liposomal gel formulations (G1–G10) and the marketed Product was evaluated to assess rheological behavior and suitability for topical application. The Viscosity ranged from 10,800 to 31,500 cP, mainly influenced by polymer type and concentration,
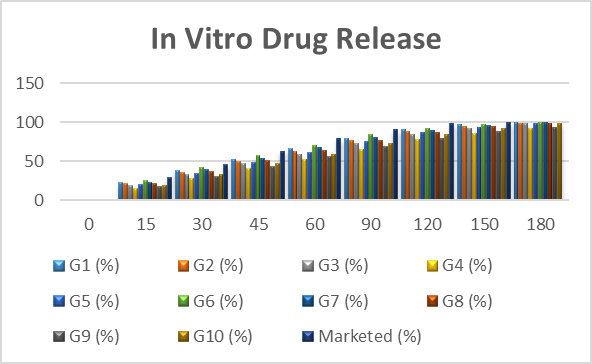
In Vitro Drug Release Studies
The marketed product and the in vitro drug release profiles of ketoconazole-loaded liposomal gel formulations (G1–G10) were compared. Consistent with controlled release behavior, all formulations showed a progressive increase in drug release over time. Effective drug diffusion from the gel matrix was demonstrated by the developed formulations’ total drug release, which varied from 92.3% to 100% over 180 minutes.The balanced release profile of the improved formulations G3 and G10 allowed for prolonged medication release without unduly postponing drug availability. In about 150 minutes, the marketed formulation achieved almost full drug release, demonstrating quick release characteristics.
SUMMARY AND CONCLUSION
Using polymers like Carbopol and HPMC, which are frequently utilized in topical administration systems because of their biocompatibility and capacity to improve rheological properties, the improved liposomal formulation was further integrated into a semisolid gel matrix (G1–G10). Numerous physicochemical properties were assessed for the produced gels. Every formulation's pH stayed within the skin-compatible range of 6.0–7.0, guaranteeing that there was little chance of irritation after application. The viscosity measurements showed that the gel had the right consistency, making application simple and retaining sufficient retention on the skin's surface. Studies on spreadability verified that the formulations could be readily dispersed throughout the impacted area, which is crucial for consistent drug distribution. Additional evidence of consistency was provided by extrudability and drug content analysis, where drug content remained continuously high (above 95%), showing dose uniformity. In order to improve topical distribution and boost antifungal activity, the current study successfully developed and assessed a ketoconazole-loaded liposomal gel system (F1–F10 and G1–G10). In addition to having appropriate PDI and zeta potential values, the liposomal formulations made using the thin-film hydration approach displayed nanosized vesicles with particle sizes ranging from 160.3 to 205.6 nm, indicating uniformity and strong physical stability. The successful integration of ketoconazole into the lipid bilayer was confirmed by the improved formulation’s effective drug encapsulation. Carbopol and HPMC were used to successfully incorporate the tailored liposomes into a gel base, resulting in a semisolid formulation with desired physicochemical attributes. The gel showed good spreadability, enough extrudability, a skin-compatible pH (~6.0–7.0), and the right viscosity for topical administration.
REFERENCES



Rohini Pundge, Sameer Sheaikh, Milke U. R., Design And Evaluation of Ketoconazole-Loaded Liposomal Gel for Enhanced Topical Antifungal Therapy, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 2314-2324, https://doi.org/10.5281/zenodo.19591926
 10.5281/zenodo.19591926
10.5281/zenodo.19591926