
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
PES, Modern College of Pharmacy (For Ladies) Moshi, Pune, Maharashtra India.
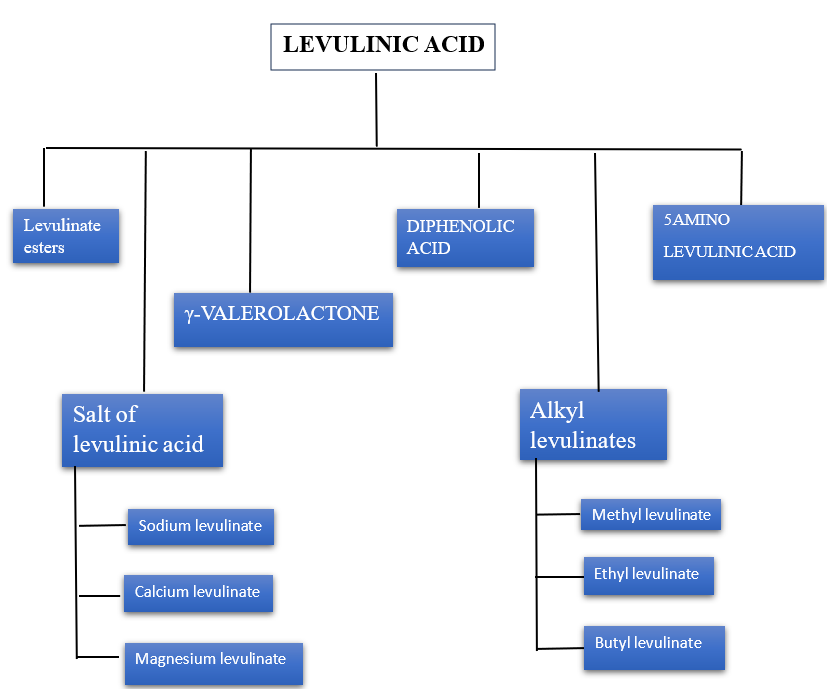
Levulinic acid (LA) is a bio-based platform chemical with wide-ranging applications in food, fuel, and pharmaceuticals. Its derivatives—such as sodium, calcium, methyl, and butyl levulinates etc. They exhibit antimicrobial, nutritional, and flavor-enhancing properties. LA-based esters and compounds like ?-valerolactone and diphenolic acid contribute to biofuel development and advanced materials. 5-aminolevulinic acid (5-ALA) shows promise in treating COVID-19 and skin cancer through photodynamic therapy. Levulinic acid (LA) is a versatile compound used in cosmetics, agriculture, and drug delivery. In cosmetics, it acts as an antimicrobial agent and a permeation enhancer for treatments like Photodynamic Therapy (PDT). LA also improves skin permeation in transdermal drug systems. In agriculture, aminolevulinic acid (ALA) exhibits herbicidal effects, with some plants showing tolerance. ALA also helps combat fungal infection in rosemary, boosting growth and essential oil production. Additionally, LA is used in sensors for sulfur dioxide detection. It has application in the development of sustainable bioplastics and plasticizers, enhancing material properties. Levulinic acid (LA) is a versatile, eco-friendly compound used in agriculture, biofuels, fragrances, and food industries. It enhance biodegradable polymer production and waste management. In the fragrance and food sectors, LA contributes to flavor and helps prevent pathogen spread. LA is a key feedstock for biofuels like ?-valerolactone, offer sustainable alternatives to fossil fuels. Levulinic acid (LA) can be converted to succinic acid (SA) and used in biocatalysis to produce pharmaceutical intermediates. LA also demonstrates significant antibacterial activity.
The first evidence for the formation of levulinic acid was obtained from the treatment of sugars with dilute acid solutions. This was one of the earliest known discoveries of levulinic acid. The initial proof of levulinic acid's synthesis was obtained through the treatment of sugars with dilute acid solutions, which induced the conversion of the sugars into this compound. Levulinic acid is a type of keto?carboxylic acid, featuring both a carbonyl group and a carboxyl group in its structure, making it a key intermediate in the production of various chemicals and materials. Top of FormBottom of FormLevulinic acid with formula CH 3C(O)CH2CH2COOH prepared in 1840 by Dutch chemist Gerardus Johannes Mulder. He synthesized it by heating fructose with hydrochloric acid. It is prepared by various method like acid hydrolysis of carbohydrates of various sugars like glucose, galactose, sucrose, fructose and polysaccharide like chitose, by wood, starch and agricultural wastes by lignocellulose, Microwave radiation-assisted synthesis of levulinic acid from microcrystalline cellulose, by steam explosion and oxidation-hydrolysis route it is produced by various method like isolation, hydrolysis, stram distillation. 5 -amino levulinic acid (5-ALA) is found in natural amino acid animals, plants, fungi and bacteria. [1] The preparation of Levulinic Acid (LA) has changed over the years. intial approaches uses acid for dehydrative treatment of biomass and carbohydrates it is reported by Dahlmann in 1968. Vaerman and Bertrand 1972 introduced the oxidation of ketones using manganese (III) acetate. Edwards in 1986 introduced the use of ozone for ketone oxidation to synthesize LA. Jow et al. in 1987 further advanced the dehydrative treatment method for LA production. Itaya et al. (1988) developed a process to produce LA by acid hydrolysis of furfuryl alcohol. Cavinato and Toniolo (1990) reported a novel method which involve palladium- catalyzed carbonylation of ketones for LA production. Farnleitner et al. in 1991 founded a method involving the hydrolysis of acetyl succinate esters. Lourvanij and Rorrer (1994) established the acid-based dehydrative treatment of biomass. Farone and Cuzens (1998) continued refining the acid dehydration approach for LA production from carbohydrates. The Biofine Corporation introduced LA synthesis with a two-reactor system designed to minimize side product formation. This process focuses on the acid hydrolysis of 6-carbon sugars.[2]
Synthesis From Lignocellulose
Lignocellulose, found in sources like wheat straw and municipal waste, is a renewable, low-cost feedstock for levulinic acid (LEV) production. Its components include cellulose (38–50%), which converts to glucose, HMF, and LEV; hemicellulose (23–32%), yielding sugars that also form LEV; and lignin (15–25%), a complex, degradation-resistant structure. Using low-lignin materials and efficient methods, lignocellulose can achieve up to 60% LEV yield[3]
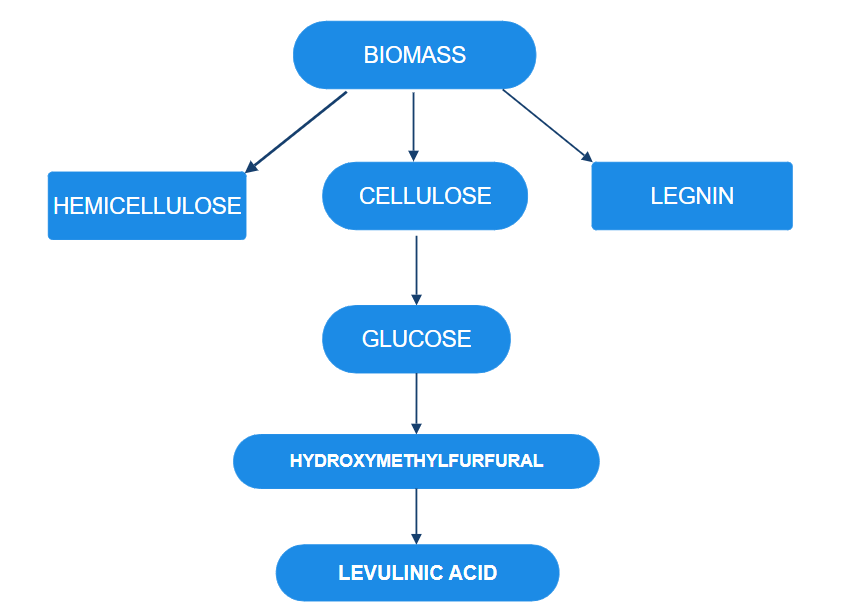
A Production from Glucose and Fructose
Levulinic acid (LA) production from glucose involves an acid-catalyzed reaction. First, glucose is isomerized to fructose via a C1-C2 hydride. It involves Lewis acid. Fructose then undergoes dehydration. Degradation of HMF in the presence of H?O and Brønsted acid produce LA. Homogeneous catalysts like inorganic acids, ionic liquids, and metal chlorides gives good results. Glucose gives a 70% LA using 1 M H?SO?. Conversion of cellulose to LA with Crcl? gives 67% yield. These systems face challenges due to purification and recycling issues. [4] Levulinic acid is very useful chemical in pharmaceutical industry. It is produced by dehydrating fructose and glucose. In this cation exchange resin in aqueous medium is used. Nacl,kcl,and cacl2 salt enhance the conversion rates of this process. In this process cl- ion is most imp. If condition are good then one can get,74.6% of la from 10% fructose at 110 degree celceous and from glucose. it is increased to 70%. This reaction gives high yield of lausing heterogeneous catalysis[5] Succinic acid, diphenolic acid, pentanol, 1,4-pentanediol, 5-nonanone, 5-methyl-2-pyrrolidone, and pentanoic acid represent a high-value chemicals obtainable from LA, renowned for their diverse applications as plasticizers, flavor enhancers, solvents, and monomeric precursors. [6].
Levulinic Acid Properties and Natural Occurrence
Chemical Properties
Levulinic Acid:- Molecular Weight: 116.11 g/mol, Estimated log Kow: -0.498, State: Solid with a low melting point and limited granularity, Functional Groups: Carboxylic acid and ketone
UV/Vis Absorption:- No significant absorption observed between 290–700 nm, Molar absorption coefficient below 1000 l/mol⋅cm, indicating no phototoxic effects, compound is highly soluble in water
Natural Occurrence
Sodium Levulinate
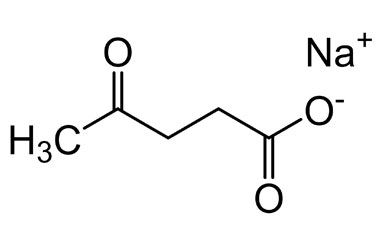
its weight is 138.1 g/mol, Estimated log Kow: -0.616, State: Solid, white to off-white powder, Functional Groups: Carboxylic acid and ketone.This research investigated the impact of 1.4% and 2.7% sodium levulinate on the aerobic bacterial count (APC), hue, (pH), and (TBA) levels in freshly prepared pork and turkey sausages. In experiment which is carried out on aerobic microorganism it is found that sodium levulinate shows inhibitory effect on growth of these microorganism on storage in comparision to control sample. It is Antimicrobial activity is independent of concentration its 1.4% concentration shows similar effect as its 2.7% .TBH, colour and pH of concentration remain same after treatment of sodium levulinate. We can conclude that it has potential to use antimicrobial agent for fresh sauag. it also provide financial sustainability, marketability. [8] It is also used incomercial product of name – aqualogica suncreen
Calcium Levulinate
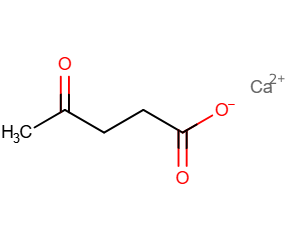
(4?ketopentanoate, LEV) is a dietary calcium supplement, Calcium levulinate is administered orally or intravenously as a supplement for calcium. Calcium levulinate is a novel calcium compound.it is synthesized from L- or levulinic acid, levulose, and calcium hydroxide. It Contains 14.8% more calcium than calcium lactate. It is a low molecular weight and organic calcium ion type.it is Highly bioavailable and easily absorbed through the intestinal wall. [9]
Applications: it Enhances calcium content in a various products, including- Sauces and condiments, Beer and other beverages, Softdrinks, Milk and milk-based products, Soy milk and soy-based products. It is use in the formulation of tablets, capsules, and injections, it Can be applied individually or in combination with other calcium compounds like calcium lactate and calcium chloride. [10] It is basically intended for improving calcium nutrition in foods, beverages, and pharmaceutical preparations. Calcium levulinate improve in calcium supplementation, it offer higher calcium content and Used as a calcium supplement.it may be used in veterinary medicine in hypocalcemic states [11]
Methyl Levulinate
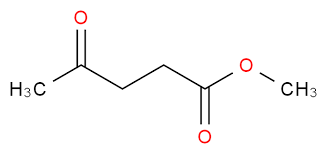
Methyl levulinate is also called as Methyl 4-oxopentanoat. It is used as flavoring agent or adjuvant. [12] It is used in the production of levulinic ketal like Glycerol Levulinate Ketal. it is a small chemical made from levulinic acid. It is used in medicine, fuel, and as a cleaner or solvent. ML is a lot like biodiesel. When it’s mixed in it helps the fuel burn cleaner. This mix also works better—it’s smoother, safer at high temperatures, and flows well in the cold.[13]
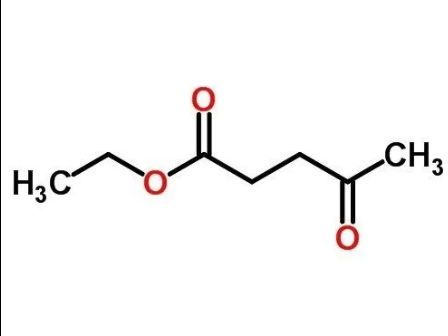
Ethyl Levulinate
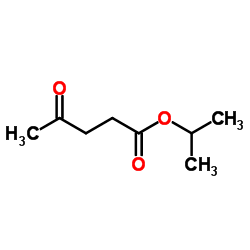
Ethyl 4-oxopentanoate, has a pleasant, fruity [14] Although it is related to ethyl pentanoate. it provides a richer and more complex aroma. The keto group increases its polarity, improving water solubility, which is beneficial for flavor systems where ingredients are divided between fat and water, such as in frozen desserts. it is recommended to use is at a 0.05% concentration in ready-to-drink beverages or simple broths. Ethyl levulinate enhances the complexity and depth of various fruit flavors at specific concentrations. Ethyl levulinate deepens fruit flavors 500 ppm for blackberry and raspberry, 150 ppm for blackcurrant, 800 ppm for blueberry, 500 ppm for cherry, and 100 ppm for strawberry. [15]
Butyl Levulinate
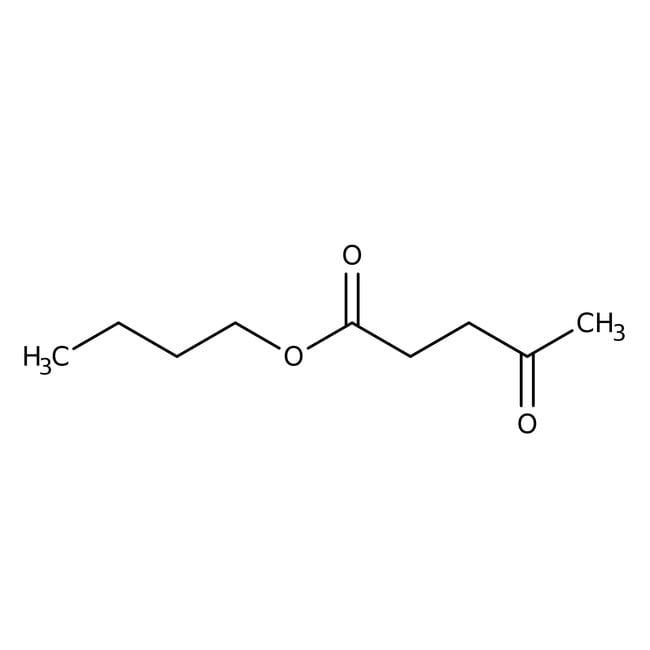
butyl 4-oxopentanoatefunctions as important component in both aroma and scent compositions that is flavoring agent [16]It proves valuable across a range of palatable uses, imparting profiles with subtle smoky undertones. Furthermore, it enhances fruit essences, including melon, banana, and cherry, with exceptional sophistication. [17]
Levulinate Esters
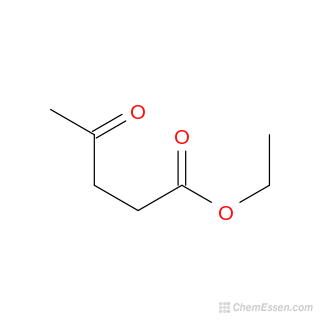
Levulinate esters are derived from levulinic acid via esterification, it employ an alcohol as the reactive counterpart. Levulinate esters show potential contributions in the fragrance and flavoring industries and as additives for diesel and biodiesel fuels. Its Several technoeconomic assessmnts have been conducted regarding the use of levulinate esters as fuel additives and for the increased production of liquid hydrocarbon fuels[18]. Cellulose levulinate ester (CLE), has an acetyl propyl ketone moiety, it serves as a precursor for fully biobased derivatives (CLEDs) via aldol condensation and with lignin-derived phenolic aldehydes and it is catalyzed by DL-proline. CLEDs exhibit a phenolic α, β-unsaturated ketone structure, it provide UV absorption, antioxidant activity, fluorescence, and biocompatibility. [19] If it combined with tunable substitution of CLE and diverse aldehyde choices, it will show a pathway to a various cellulose polymers and advanced polymeric architectures. Levulinate esters have significant applications, from flavoring and food additives to gasoline additives and bio-liquid fuels as it is a versatile compound. It is primaraly focued on its use as blending agents with petrochemical diesel and other transportation fuels, enhancing performance and sustainability.[20]
Gamma Varololactone
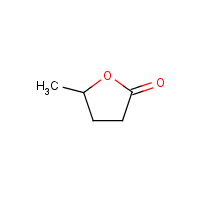
GVL's works as a reaction medium, a component in fuel formulations, and a building block for jet fuel and polymer synthesis. [22]The process of the transformation of GVL into high value chemicals such as valeric acid and 1,4-pentanediol, as well as polymers like α-methylene-γ-valerolactone and ε-caprolactam, involves catalytic processes. it enable selective functionalization and derivatization of it. GVL is highly regarded as an outstanding medium for the fractionation of lignocellulosic biomass. It attributed to its efficacy in dissolving lignin and promoting the deconstruction of GLV. GVL is precursor for the synthesis of carbon-based chemicals, polymeric materials, and liquid fuels. Its utility extends to the production of liquid fuels, such as C8–C18 branched and linear hydrocarbons, derived from GVL. [23]
Diphenolic Acid
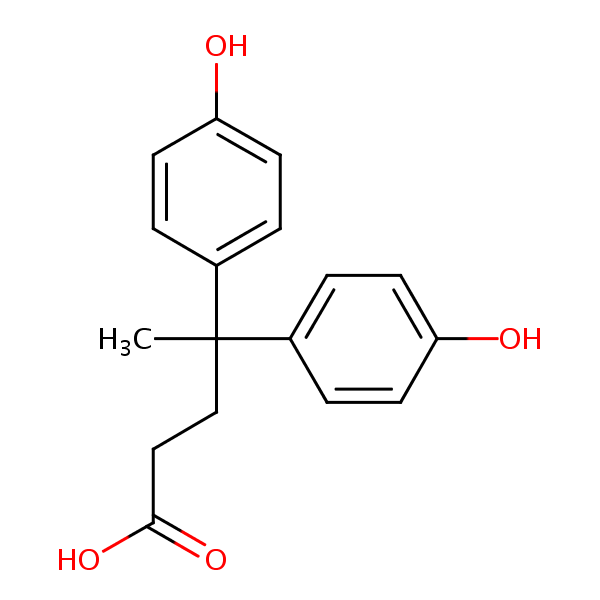
Diphenolic acid is synthesized through a condensation reaction. it involve phenol and levulinic acid, and Brønsted acid as catalyst. Diphenolic acid (DPA) shows performance in augmenting the UV stability of coatings by effectively inhibiting photodegradation and color deterioration during extended sunlight exposure. This property can be applied in architectural surfaces and automotive exteriors. DPA has antioxidant property which can be utilize in a wide range of industries. In packaging, it can increase product shelf life, in cosmetics, its shows anti-aging properties. DPA and its derivatives can be used in the development of paint systems, decorative coatings, lubricant additives, plasticizers, cutting-edge surfactants, personal care formulations, and in the textile sector [24]
5 Amino Levulinic Acid
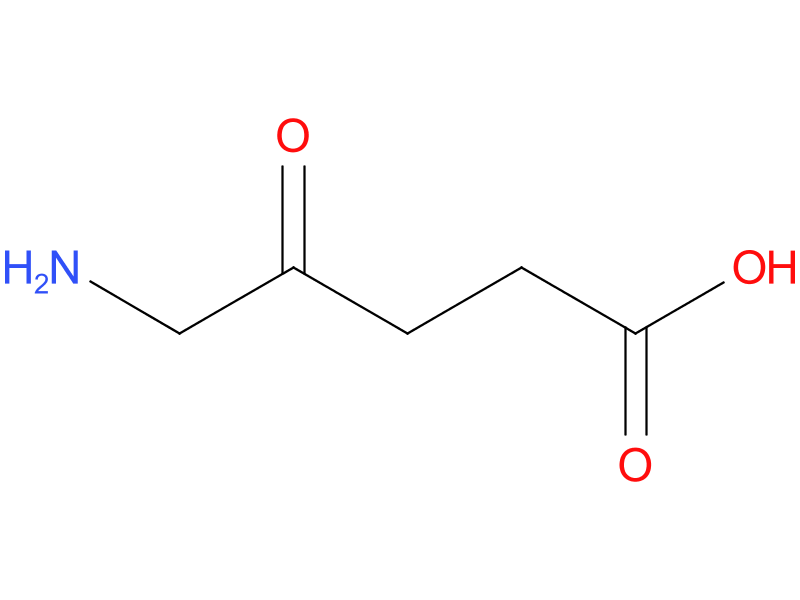
IN Corona Virus
In recent studies it was found that 5ALA can be used to treat corona virus which is severe respiratory acute syndrome (SARS). Eight 5 ALA conjugate to for protoporphyrin IX (PPIX) and PPIX.it generate heme, by inseration of ferrous ion. 5ALA used to increase the metabolism in disease like diabetics. The photosensitive properties of PPIX used 5-ALA to help in cancer detection and therapy, its application can enhance human health. The accumulation of PPIX in VeroE6 cells treated with 5-ALA aligned with observation of a time-dependent increase in 5-ALA's anti-SARS-CoV-2 efficacy in these cells.[25]
IN Skin Cancer
Several reports have been published with evidence that ALA, in various concentrations, can be used to treat skin cancer. The results of one study showed its effectiveness in treating nail psoriasis. three intractable cases of palmoplantar pustulosis (PPP) were treated with PDT using 20% 5-aminolevulinic acid and a 630 ± 50 nm LED device. The intensity was 30 mW/cm², and the dose was 15 J/cm². After treatment, all cases exhibited mild to substantial improvement. Photodynamic therapy (PDT) destroys tumors through three main mechanisms:- ROS Generation ,Microvascular Damage, Immune Activation.[26] ALA is a small molecule with no intrinsic photosensitizing properties. It bypasses a regulatory checkpoint in the heme synthesis pathway, leading to the accumulation of in cells. Malignant and premalignant cells accumulate PPIX at a rate 2 to 10 times higher than normal cells. ALA-PDT targets these cells due to both biochemical factors and the increased permeability of the stratum corneum in superficial lesions, which enhances ALA penetration.
Cancer Detection and Prevention: ALA-PDT is better treatment for Bowen’s disease and it serves as an effective, non-invasive therapy for superficial basal cell carcinoma (sbcc). It may also be beneficial in treating nodular basal cell carcinoma (nbcc), other skin malignancies, and localized cutaneous lymphomas. Its Benefit is Minimal scarring, Outstanding cosmetic results, Capability to treat multiple lesions at once.in Preventive role, ala-PDT may also serve as a preventive treatment for new skin cancers in high-risk individuals. [27]
Application Of Levulinic Acid
Levulinic Acid to Succinic Acid
Levulinic acid can be converted to succinic acid (SA) using 30% aqueous hydrogen peroxide (H?O?). in this process trifluoroacetic acid (TFA) at 90 °C is used. The reaction completes in 2 hours.it yields 62% SA, 43% acetic acid, 45% formic acid, and 9% hydroxypropionic acid (HPA), by ¹H NMR integration.In a transesterification reaction, the monomethyl ester of SA reacts with TFA, produce methyl trifluoroacetate and its yield is 45% . This volatile compound can be trapped in a cold trap. For purification process, the crude product undergoes trituration with ether. ether remove soluble impurities and leave purified SA as an insoluble solid. The isolated yield of pure SA is approximately 60%. The ether-soluble fraction contains hydroxypropionic acid, small amounts of SA lost during washing, and minor unidentified by-products. [29]
Use Of Nanoflower
Levulinic acid (LA) was converted into enantiopure (S)-4-aminopentanoic acid through a recyclable enzymatic cascade with two transaminases (TAs). The first TA catalyzed the amination of LA using (S)-α-Methylbenzylamine [(S)-α-MBA] as the amino donor. it produce acetophenone as a byproduct. The second TA recycled acetophenone by reacting it with isopropyl amine (IPA), acetone as byproduct. Conversion can be ranged from 30% and 80% depending on the reaction system. Hybrid nanoflowers (HNF) of fusion proteins were developed to achieve complete conversion, with good stability for over a month and to ues in seven cycles. In a preparative reaction (100 mL), the cascade system conversion, results in a 62% isolated yield. The method was tested on various β-keto ester substrates, yielding 18%-48% of the corresponding β-amino acids. it can synthesis a pharmaceutical intermediate with an 82% conversion rate, showing its application in sustainable biocatalysis and industrial applications. [30]
Antibacterial Agent
Therapeutics and prophylactic strategies are being planed to fight with bacteria which are linked to dental caries, acne vulgaris, and the pathogenesis of various species like domesticated livestock, avian poultry, and aquatic organisms.
with sodium dodecyl sulfate (SDS) in recent years has shown potential to use as an antimicrobial studies S. Aureus. Staphylococcus aureus (KCTC 1621), Pseudomonas aeruginosa (KCTC 1636), Enterococcus faecium (KCTC 3095) LA shows antibacterial activity for thisbacteria. Levulinic acid shows potent antibacterial activity, it exhibit minimum inhibitory concentrations (mics) ranging from 1.5 to 4.5 mg/ml against multidrug-resistant bacterial strains. Its efficacy against Pseudomonas aeruginosa KCTC 1636 and resistant Pseudomonas aeruginosa is remarkable. Levulinic acid can be used as a active pharmacological component in the formulation of antibiotics targeting drug-resistant pathogens. By observing various Streptococcus mutans strains, like HS-6, BHT, GS-5, Ingbritt, and OMZ-176, it is conclude that levulinic acid has antibacterial properties. It can mitigat the pathogenicity of these strains. Levulinic acid as a antimicrobial agent can fight Streptococcus infections. Levulinic acid shows significant antimicrobial activity, with minimum inhibitory concentrations (mics) ranging from 0.37 to 3.5 mg/ml against pathogens infecting aquatic organisms and avian species, including Edwardsiela tarda GYO1, Vibrio anguillarum YT-85805, and Salmonella typhimurium KCTC 1925. Levulinic acid can be utilized as a potent component in antibiotic formulations targeting pathogenic organisms affecting aquatic life and poultry. The MIC against acne-causing bacteria was 6 mg/ml, and against pathogens linked to stomach ulcers, duodenal ulcers, and gastric cancer was also determined. [31] [32]
Levulinic Acid In Cosmetic
Levulinic acid is basically used in cosmetics to inhibit microbial growth while maintaining the pH and color stability of beauty products. Studies have shown that levulinic acid show no short-term toxicity in rats or adult male humans.
Permeation Enhancer
Microneedling, a cosmetic technique. it creates micro-injuries in the stratum corneum, potentially enhancing Levulan (delta amino levulinic acid) penetration in Photodynamic Therapy (PDT) and improve both treatment efficacy and cosmetic outcomes. [33]
To study the effects of different chemical penetration enhancers on skin permeation rate and adhesion properties in a transdermal drug delivery system.
Case study
Methodology:- A series of drug-in-adhesive transdermal patches were formulated with varying concentrations (0-8%) of levulinic acid, lauryl alcohol, and Tween 80 as chemical penetration enhancers. The Box-Behnken experimental design, a three-level, three-variable approach, was employed to evaluate the effects on dependent variables:-Skin permeation rate (flux),Adhesion properties (peel strength and tack value).Buprenorphine was selected as the model drug for testing.
Skin Permeation Rate:- Incorporation of 20% penetration enhancers resulted in the highest skin permeation flux for buprenorphine through rat abdominal skin. Flux values for individual enhancers for Levulinic acid: 1.594 μg/cm²·h, Tween 80: 1.473 μg/cm²·h, Lauryl alcohol: 0.843 μg/cm²·h A synergistic effect was observed when enhancers were combined. Adhesion Properties like Tack Value is Increased with all enhancers and Peel Strength Improved by levulinic acid and lauryl alcohol but it is Reduced by Tween 80.
Conclusion:- Levulinic acid effective chemical penetration enhancer for buprenorphine patches. Among all formulations, the patch containing 12% (wt/wt) of enhancers achieved the best balance of skin permeation and adhesion properties. These results shows the potential of levulinic acid in optimizing transdermal drug delivery systems for buprenorphine. [34] According to some report, By itself, levulinic acid, a short-chain fatty acid, does not alter the lipid structure of the stratum corneum. But in situations where lipid organization is already impaired, like high temperatures or when nerolidol is present, it improves lipid fluidity. This suggests that levulinic acid only works as a conditional permeation enhancer in the presence of disorder in the lipid matrix.[35]
Herbicidal Activity of Aminolevulinic Acid (ALA)
Sang-Uk Chon discussed about Herbicidal Activity of Aminolevulinic Acid (ALA) on Different Plant Species in which it is concluded that Aminolevulinic acid (ALA), an intermediate in tetrapyrrole biosynthesis (e.g., chlorophyll, heme, bacteriochlorophyll, and vitamins), was studied for its phytotoxic effects on various plant species under different application methods.
Seed-Soaking Treatment:- Low ALA concentrations showed no effect on shoot and root lengths, High concentrations reduced growth by 20–30%, Alfalfa exhibited the highest tolerance to ALA.
Pre-Emergence Application:- Chinese cabbage cotyledons were severely bleached at 0.5 mM ALA within 24 hours, Root growth of rice, barnyard grass, and alfalfa was significantly inhibited with increasing ALA concentrations, Alfalfa and rice showed higher tolerance compared to other species.
Post-Emergence Application:- ALA at 2–4 mM completely inhibited shoot and root growth in Chinese cabbage and barnyard grass, Alfalfa remained the most tolerant species.
Combination with 2,2-Dipyridyl:- Herbicidal effects were enhanced in barnyard grass and Chinese cabbage when ALA was applied with 2,2-dipyridyl compared to ALA alone.
Conclusion: Alfalfa shows high resistance to ALA across treatments, while post-emergence application showed the strongest herbicidal effects, through photodynamic activity.[36]. Some study shows how rice plants respond to photodynamic stress caused by 5-aminolevulinic acid (ALA). ALA treatment resulted in white necrosis, a significant decrease in photochemical efficiency (Fv/Fm), and increased superoxide dismutase activity. ALA help in detoxification processes, like those for transport, cellular homeostasis, and xenobiotic conjugation, with unique upregulation of serine/threonine kinase and chaperone genes. These findings highlight the role of antioxidant and detoxification systems in treating ALA-induced photodynamic stress and protecting plants from damage. [37]. ALA treats Fungal Stress in Rosemary - This study explored the effects of 5-aminolevulinic acid (ALA) (3–10 ppm, eight weekly applications) on Salvia rosmarinus which is infected by Alternaria dauci and Rhizoctonia solani. ALA improved plant growth, branch number, biomass and enhanced essential oil production (1,8-cineole, linalool, camphor, borneol). It increased antioxidant enzyme activities (SOD, CAT), total phenolics, chlorophyll, sugars, and proline levels. ALA also enhanced gas exchange, water content, and antioxidant (FeSOD, Cu/ZnSOD, MnSOD) gene expression. The effects were more incresed in fertile soils, focusing ALA's potential as a biotic stress mitigator. [38]
Probe Design
Levulinic acid – Serves as the SO2 recognition site, DCM-OH – A dicyanomethylene-benzopyran (DCM) fluorophore known for its outstanding photophysical properties,so it is selected for its NIR fluorescence capabilities. The combination of the levulinic acid moiety and the DCM fluorophore able to the detect SO2 levels in vivo, in tumor-bearing mice. A novel NIR fluorescence probe, DCM-SO2, is designed and synthesized for SO2 detection. Its properties like large Stokes Shift, high selectivity, strong interference resistance, and low cytotoxicity. these properties make it an effective tool for SO2 Detection in Biological System and tumor Research. [39]. Sulfites (HSO3−), commonly used as preservatives. It can cause health issues like asthma and accumulate in the body. A levulinic acid-based sensor, SO-2, was developed to detect SO2 with high sensitivity (detection limit: 2.0 × 10−8 M) and a fast response time (<10 min). The probe successfully detect bisulfite in cells and analyzed natural water samples, making it suitable for environmental and biomedical applications. [40]
Levulinic Ketal
Levulinic acid is a chemical that can be derived from lignocellulosic biomass. It can be further converted into various valuable compounds, including levulinic ketal. Levulinic ketals are synthesized by the reaction of levulinic acid or its esters with alcohols like ethyl levulinate, using an acid catalyst. This forms a cyclic acetal structure [41]. The preparation of levulinic ketals, using glycerol as the alcohol component, has been explored as a way to valorize byproducts from the biodiesel industry [42]. Successfully prepared ketal intermediates from levulinic acid derivatives using a recyclable Na-β catalyst. It Achieves 97–100% yields from alkyl levulinates (methyl and butyl) with glycerol and ethylene glycol. The Dean-Stark method enhanced conversion and selectivity by removing water by-products. Even crude glycerol gave a 97% yield, highlighting the method's value for biodiesel by-products.[42] . ethyl levulinate react with glycerol to form a five-membered ketal, using H,Y-zeolite as a catalyst. The reaction was studied in a batch reactor and a continuous milli-reactor. Key ingredients are ethyl levulinate, glycerol, and H,Y-zeolite catalyst.[43] Glycerol levulinate ketal (GLK) is an innovative biobased compound. It has significant potential for industrial applications, as a green solvent, in polymer production and polyurethane foam production.[ 44]
Plasticizer
Levulinic acid can be used as a raw material in the synthesis of polymers and as an initiator of polymerization reactions.[45] Levulinic acid-based plasticizers show great potential for polylactide (PLA). Ketone end groups lower PLA’s Tg, while ketal groups enhance thermal stability. Ethylene glycol dilevulinate improves flexibility, achieving 546% elongation at break, competing traditional plasticizers like ATBC[46] Levulinic acid can be used to synthesize novel bioplasticizers through a protecting-group-free three-step process. A bioplasticizer was synthesized through solvent-free, little esterification. It utilizes levulinic acid and glycerol, two industrial by-products. This plasticizer effectively reduces the brittleness of PHB and optimizes its viscosity.[47] Levulinic acid-based bioplasticizers were synthesized via a three-step process and tested with poly(3-hydroxybutyrate) (PHB). These plasticizers enhance PHB’s flexibility, reduce brittleness, and lower glass transition and melting temperatures, improving processability without compromising biodegradability or biocompatibility.[48] A various plasticizers was developed using the levulinic acid (lea) as a building block. Combined with other current or emerging biobased components, lea esters and derivatives offer versatile plasticization solutions for the biodegradable thermoplastic polylactide (PLA).[16]
Agriculture
Levulinic acid (LA) is an eco-friendly option that can enhance waste management in sustainable agriculture. Its derivatives have diverse commercial uses, including pharmaceuticals, fuels, food additives, solvents, biopolymers, and agricultural products. [49]
Coating And Textile
Fabrics treated with Levulinic acid /Sodium Dodecyl sulfate coatings can be utilized in food processing plants and retail settings to help prevent the spread of airborne pathogens. [50]
Feedstock For Pha Biosynthesis
levulinic acid (LVA) is a well-known platform chemical widely used in the synthesis of beneficial materials. Conventional feedstocks like glucose have often been combined with LVA for PHA production. LVA offers a cost-effective alternative because it is easily produced via acid catalysis from abundant renewable materials. Its cost is significantly lower than commonly used alternatives like propionic acid or pentanoic (valeric) acid. By pairing LVA with low-cost carbon sources such as glycerine, whey permeate, and xylose, unique PHA polymers develop.it has diverse compositions and structures. This approach Enhance Properties of these polymers. Improved economics of production make renewable PHA polymers. LVA in combination with other inexpensive feedstocks enhances the economic and functionalality of PHA polymers. it support their broader industrial application. [57]
Fragrance
Levulinic acid is a versatile compound used to enhance both savory and sweet flavors. It is present in fish sauces, a key element of many Asian dishes, and plays a significant role in the distinctive taste of Vietnamese cuisine, such as nuoc cham. Its sweet, syrup-like aroma makes it valuable for sweet applications, particularly in cola flavoring, where it rounds out the profile in combination with caramel color, gum Arabic, and kola nut extract. Levulinic acid is also useful in confectionery, like "Kola Kubes," when diluted in triacetin. Furthermore, it supports sugar reduction in low-calorie colas, especially when combined with sweetness-enhancing compounds like maltol. [16]. Levulinic acid has Sweet, creamy, acidic, buttery, and guaiacol-like odour. It is commonly used in creating flavors such as: Caramel, Butterscotch, Brown sugar, Maple.Levulinic Acid Esters used as niche fruity flavor and fragrance ingredients. Ethyl Levulinate has potential replacement for valencene (a flavor extracted from oranges), especially in beverages. Levulinate esters serve as flavoring agents, Alkali earth metal salts of levulinic acid inhibit microbial growth in foods, Helps regulate the pH of food ingredients, Used for disinfecting fruit surfaces. [52]
Biofuel
LA, derived from C5 and C6 carbohydrates, is crucial for biofuel production. LA and alkyl levulinates are converted into γ-valerolactone, 2-methyltetrahydrofuran, and valeric acid. Efficient conversion relies on advanced catalysts influenced by additives, particle size, and surface properties. Levulinate esters, produced from levulinic acid, are renewable fuels that improve combustion efficiency and reduce emissions. Ionic liquids catalyze the conversion process, making it a sustainable and efficient method for producing eco-friendly transportation fuels. [53] Levulinic acid serves as a crucial fuel additive owing to its versatile functionality, encompassing both keto and carboxylic functional groups. Additionally, it holds the potential to yield various other fuel additives, including γ-valerolactone, ethyl levulinate, and 2-methyl tetrahydrofuran. These compounds can be seamlessly blended with petroleum-derived transportation fuels, akin to bioethanol, without necessitating modifications to the engine infrastructure. [54]. Levulinic acid (LA) is a crucial chemical for biofuel production. LA is converted into 2-MTHF through hydrodeoxygenation a promising biofuel with higher energy density and stability than ethanol. It achieves 73.4% yield using a NiCo/γ-Al2O3 catalyst. LA hydrogenation produces valeric acid, which serves as the basis for valeric biofuels. LA dimers, such as TMDFA and MOTOA, are potential biojet fuel precursors. These are synthesized via dimerization using an H-Beta 19 catalyst, it achieve high selectivity (>98 mol%). these biofuels offer sustainable alternatives to fossil fuels.[55]
CONCLUSION:
In conclusion, after reviewing various research and review paper it can be conclude that Levulinic Acid (LA) is a highly versatile and sustainable bio-based chemical.it has applications across various industries, including pharmaceuticals, agriculture, food, and biofuels. Its derivatives, such as levulinate esters and γ-valerolactone, offer significant potential in enhancing antimicrobial properties, promoting sustainable biofuel development, and contributing to advanced materials.LA-based compounds, like 5-aminolevulinic acid (5-ALA), have shown promise in therapeutic areas, such as photodynamic therapy for skin cancer and COVID-19 treatment. The role of LA in agriculture, enhancing growth and combating fungal infections, shows its value. Its eco-friendly characteristics and diverse applications, Levulinic Acid stands out as an essential compound for the future of sustainable technologies and drug discovery. Further research and development will unlock more opportunities in various sectors.
REFERENCES


Shubhangi Shinde*, Dr. Raksha Mhetre, Levulinic Acid: Types and Versatile Applications in Science and Industry, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 8, 794-811. https://doi.org/10.5281/zenodo.16777357
 10.5281/zenodo.16777357
10.5281/zenodo.16777357