
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
1Faculty, Department of Pharmacology, Kamalakshi Pandurangan College of Pharmacy, Ayyampalayam, Tiruvannamalai 60603, Tamilnadu, India.
2B. Pharm Student, Department of Pharmacology, Kamalakshi Pandurangan College of Pharmacy, Ayyampalayam, Tiruvannamalai 60603, Tamilnadu, India.
3Vice Principal, Departmen of Pharmacology,Kamalakshi Pandurangan College of Pharmacy, Ayyampalayam, Tiruvannamalai 60603, Tamilnadu, India.
4Principal, Department of Pharmaceuticl Chemistry,Kamalakshi Pandurangan College of Pharmacy, Ayyampalayam, Tiruvannamalai 60603, Tamilnadu, India.
Metformin is an oral antihyperglycemic agent belonging to the biguanide class and is widely accepted as the first-line pharmacological therapy for type 2 diabetes mellitus. It exerts its primary action by reducing hepatic glucose production and improving peripheral insulin sensitivity without causing significant hypoglycemia. Over several decades of clinical use, metformin has demonstrated an excellent safety profile along with additional benefits such as weight neutrality, cardiovascular protection, and potential anticancer effects. This article provides a comprehensive review of the history, chemistry, mechanism of action, pharmacokinetics, pharmacodynamics, therapeutic uses, adverse effects, contraindications, drug interactions, and future perspectives of metformin
Diabetes mellitus is a chronic metabolic disorder characterized by persistent hyperglycemia due to defects in insulin secretion, insulin action, or both. The prevalence of type 2 diabetes mellitus has increased rapidly worldwide due to changes in lifestyle, urbanization, obesity, and aging populations. Effective glycemic control is essential to prevent long-term complications such as cardiovascular disease, nephropathy, neuropathy, and retinopathy. Among the available antidiabetic drugs, metformin has gained prominence due to its proven efficacy, safety, affordability, and additional metabolic benefits. It is recommended by most international guidelines as the initial drug of choice in patients with type 2 diabetes mellitus
2. Historical Background
The origin of metformin can be traced back to the medicinal plant Galega officinalis, also known as French lilac, which was traditionally used to treat symptoms of diabetes. The plant contains guanidine, a compound known to lower blood glucose levels. During the early twentieth century, synthetic guanidine derivatives were developed, leading to the discovery of biguanides such as phenformin, buformin, and metformin. Although phenformin was initially popular, it was withdrawn due to a high risk of lactic acidosis. Metformin, however, showed a significantly lower risk and was introduced into clinical practice in Europe during the 1950s. It was later approved by the United States Food and Drug Administration in 1995 and has since become the most widely prescribed antidiabetic drug globally.
3. Chemical Structure and Physicochemical Properties
Metformin is chemically known as 1,1-dimethylbiguanide hydrochloride. It has a molecular formula of C?H??N?·HCl and a molecular weight of 165.63 g/mol. The drug appears as a white or off-white crystalline powder that is freely soluble in water and practically insoluble in organic solvents. Metformin is a strong base and exists predominantly in an ionized form at physiological pH, which influences its absorption and distribution characteristics. Its hydrophilic nature limits passive diffusion across biological membranes and necessitates transport via specific organic cation transporters.
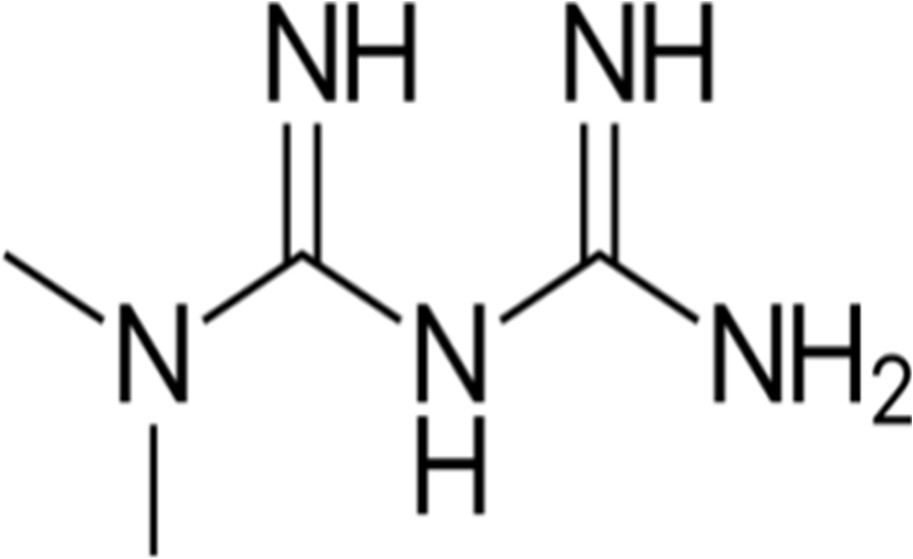
Fig.1 Structure of metformin
4. Classification
Metformin is pharmacologically classified as a biguanide antihyperglycemic agent, chemically distinct from sulfonylureas and thiazolidinediones due to its unique mechanism of action that does not involve the direct stimulation of insulin secretion. Instead, it functions primarily as an insulin sensitizer, reducing hepatic glucose production (gluconeogenesis) and enhancing peripheral glucose uptake and utilization in muscle tissues. Within the therapeutic hierarchy, it is the first-line pharmacotherapeutic intervention for the management of Type 2 Diabetes Mellitus (T2DM). From a biopharmaceutical perspective, Metformin is categorized under the Biopharmaceutics Classification System (BCS) as a Class III drug, characterized by high aqueous solubility but low intestinal permeability. This classification presents specific challenges in drug delivery, often necessitating high doses and specialized formulation strategies to overcome limited absorption windows in the upper gastrointestinal tract.
Key Classification Breakdown
5. Mechanism of Action
The glucose-lowering effect of metformin is mediated through multiple mechanisms that act synergistically. The primary action of metformin is the inhibition of hepatic gluconeogenesis, which leads to reduced endogenous glucose production. This effect is largely attributed to the inhibition of mitochondrial respiratory chain complex I, resulting in decreased ATP production and increased intracellular AMP levels. Elevated AMP activates adenosine monophosphate-activated protein kinase (AMPK), a key regulatory enzyme involved in cellular energy homeostasis.
Activation of AMPK leads to enhanced insulin sensitivity, increased glucose uptake in peripheral tissues such as skeletal muscle, and reduced lipogenesis. Metformin also decreases intestinal glucose absorption and alters gut microbiota, contributing to improved glycemic control. Importantly, because metformin does not increase insulin secretion, it carries a minimal risk of hypoglycemia when used as monotherapy.
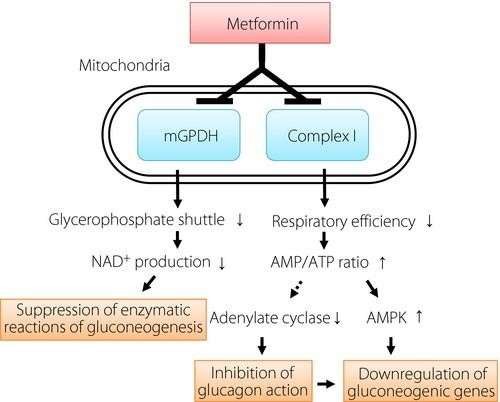
Fig .2 mechanism of action for metformin
5. Pharmacokinetics
After oral administration, metformin is absorbed mainly from the small intestine, with an absolute bioavailability of approximately 50 to 60 percent. Peak plasma concentrations are typically achieved within two to three hours for immediate-release formulations. The drug exhibits negligible plasma protein binding and is widely distributed in body tissues, particularly the liver, kidneys, and gastrointestinal tract. Metformin is not metabolized by the liver and does not undergo biotransformation. It is eliminated unchanged through renal excretion via glomerular filtration and active tubular secretion. The elimination half-life of metformin is approximately six hours in individuals with normal renal function. Renal impairment significantly reduces metformin clearance and increases the risk of accumulation.
6. Pharmacodynamics
Metformin functions primarily as an antihyperglycemic agent by improving glucose tolerance in patients with type 2 diabetes, lowering both basal and postprandial plasma glucose. Its principal mechanism of action involves the inhibition of mitochondrial complex I in hepatocytes, which leads to a reduction in cellular energy levels (ATP) and a subsequent rise in the AMP:ATP ratio; this shift activates AMP-activated protein kinase (AMPK), a cellular metabolic sensor that inhibits key enzymes involved in gluconeogenesis and glycogenolysis, thereby significantly decreasing hepatic glucose production. Furthermore, metformin enhances peripheral insulin sensitivity, facilitating glucose uptake and utilization by skeletal muscle via increased translocation of GLUT4 transporters, while simultaneously delaying intestinal glucose absorption. Unlike sulfonylureas, metformin does not stimulate insulin secretion from pancreatic beta cells, which minimizes the risk of hypoglycemia and avoids weight gain, making it a distinct class of biguanide therapy focused on fixing metabolic inefficiency rather than increasing insulin levels.
Spectrum of Activity :
Metformin, a biguanide agent, exhibits a broad spectrum of metabolic and pleiotropic activity primarily characterized by its potent antihyperglycemic effects in Type 2 Diabetes Mellitus without the inherent risk of hypoglycemia common to sulfonylureas. Its pharmacological profile is defined by a multifaceted mechanism that centers on the inhibition of mitochondrial respiratory chain Complex I and the subsequent activation of AMP-activated protein kinase (AMPK), a cellular energy sensor; this cascade results in the suppression of hepatic gluconeogenesis, the enhancement of peripheral insulin sensitivity and glucose uptake in skeletal muscle, and the reduction of intestinal glucose absorption. Beyond its primary role in glucose homeostasis, the drug’s activity spectrum extends to endocrine regulation, making it a first-line intervention for Polycystic Ovary Syndrome (PCOS) by ameliorating insulin resistance and lowering androgen levels, while emerging research highlights its potential antineoplastic and geroprotective properties through the inhibition of the mTOR pathway and modulation of inflammatory markers.
Resistance Mechanisms:
Metformin resistance is primarily driven by pharmacokinetic variability and genetic polymorphisms that impair the drug's cellular uptake and intracellular accumulation, preventing it from exerting its therapeutic effects on hepatic gluconeogenesis. Since metformin is a hydrophilic cation at physiological pH, it cannot passively diffuse across cell membranes and relies heavily on specific transporters, most notably the organic cation transporter 1 (OCT1), encoded by the SLC22A1 gene. Resistance often stems from loss-of-function polymorphisms in SLC22A1 that reduce hepatic uptake, or variations in multidrug and toxin extrusion proteins (MATE1/MATE2) that alter renal elimination; without sufficient intracellular concentration, metformin fails to inhibit mitochondrial respiratory chain complex I, which is necessary to disrupt ATP production, increase the AMP:ATP ratio, and subsequently activate AMP-activated protein kinase (AMPK) to suppress glucose production.
Adverse Effects and Safety:
Like all drugs, metformin might have negative side effects, but most patients tolerate it well. The most often reported adverse effects are diarrhea, nausea, vomiting, and abdominal discomfort, occurring in up to 30% of patients. These are often dose-dependent and transient. Lactic Acidosis: A rare but serious metabolic complication (MALA) defined by high blood lactate levels (>5 mmol/L) and acidosis (pH < 7.35). It is most likely to occur in patients with significant renal impairment or hypoxic conditions. Vitamin B12 Deficiency: Long-term use is associated with reduced intestinal absorption of Vitamin B12, potentially leading to anemia or neuropathy. Hypoglycemia: Rare when used as monotherapy, as it does not increase insulin secretion.
11. Therapeutic Indications
Metformin is primarily indicated as a first-line pharmacotherapeutic agent for the management of type 2 diabetes mellitus, utilized as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients aged 10 years and older. It functions effectively as an antihyperglycemic agent by decreasing hepatic glucose production (gluconeogenesis), decreasing intestinal absorption of glucose, and improving insulin sensitivity by increasing peripheral glucose uptake and utilization. Unlike sulfonylureas, metformin does not stimulate endogenous insulin secretion and is therefore rarely associated with hypoglycemia when used as monotherapy. While its FDA-approved labeling is strictly for type 2 diabetes, clinical practice frequently employs metformin off-label for the management of polycystic ovary syndrome (PCOS) to address insulin resistance and induce ovulation, as well as for the treatment of pre-diabetes and prevention of weight gain in patients taking antipsychotics. It is contraindicated in patients with severe renal impairment, acute or chronic metabolic acidosis (including diabetic ketoacidosis), and conditions predisposing to tissue hypoxia due to the risk of lactic acidosis.
12. Dosage and Administration
The dosage of metformin must be individualized based on the patient's glycemic control and tolerance, adhering to a "start low, go slow" titration strategy to mitigate gastrointestinal (GI) adverse effects. For immediate-release (IR) formulations, therapy is typically initiated at 500 mg once or twice daily or 850 mg once daily, administered with meals. The dosage is incrementally increased by 500 mg weekly or 850 mg every two weeks, up to a maintenance target of 2000 mg daily in divided doses. The maximum recommended daily dose for the IR formulation is 2550 mg in the United States, though clinical efficacy rarely increases significantly beyond 2000 mg. For extended-release (XR or ER) formulations, the starting dose is 500 mg once daily with the evening meal, titrating weekly to a maximum of 2000 mg once daily. Administration with food is critical for both formulations to improve bioavailability and reduce the incidence of nausea, vomiting, and diarrhea. Patients should be advised to swallow XR tablets whole, without crushing or chewing, to preserve the dual hydrophilic polymer matrix system responsible for prolonged drug release.
13. Adverse Effects
The adverse effect profile of metformin is primarily dominated by gastrointestinal (GI) disturbances, which affect approximately 20% to 30% of patients, particularly during the initiation of therapy. These symptoms typically include diarrhea, nausea, vomiting, abdominal bloating, flatulence, and a characteristic metallic taste (dysgeusia). While often transient and dosedependent, these effects can lead to drug discontinuation in nearly 5% of patients; however, their severity can be mitigated by gradual dose titration and administration with meals. Of greater clinical concern, though rare (incidence <10 cases per 100,000 patient-years), is MetforminAssociated Lactic Acidosis (MALA). This life-threatening metabolic complication arises from the drug’s inhibition of gluconeogenesis, leading to lactate accumulation, and is most prevalent in patients with pre-existing renal impairment, tissue hypoxia, or hepatic failure. Additionally, chronic metformin use is significantly associated with Vitamin B12 malabsorption, potentially resulting in reduced serum B12 levels, elevated homocysteine, and subsequent megaloblastic anemia or peripheral neuropathy. Unlike sulfonylureas, metformin monotherapy carries a negligible risk of hypoglycemia, as it does not stimulate endogenous insulin secretion.
14. Contraindications
Metformin is contraindicated in patients with conditions that increase the risk of lactic acidosis, a rare but potentially fatal complication. It should not be used in individuals with severe renal impairment, typically defined as an estimated glomerular filtration rate (eGFR) below 30 mL/min/1.73 m², because reduced renal clearance leads to drug accumulation and increased lactic acid levels. Metformin is also contraindicated in patients with acute or chronic metabolic acidosis, including diabetic ketoacidosis, with or without coma. Conditions associated with tissue hypoxia, such as acute heart failure, recent myocardial infarction, severe respiratory disease, shock, or sepsis, are contraindications because hypoxia promotes anaerobic metabolism and lactate production. Severe hepatic impairment is another contraindication, as liver dysfunction reduces lactate clearance. Excessive alcohol consumption, either acute or chronic, contraindicates metformin use due to alcohol’s potentiation of lactate accumulation. Metformin should be temporarily discontinued in patients undergoing iodinated contrast imaging procedures, particularly in those with impaired renal function, dehydration, or heart failure, to prevent contrast-induced nephropathy and lactic acidosis. It is also contraindicated in patients with known hypersensitivity to metformin or any of its components. Careful evaluation of these conditions is essential before initiating metformin therapy to ensure patient safety and optimal therapeutic outcomes
15. Drug Interactions
Metformin’s drug interactions are predominantly governed by its pharmacokinetics—specifically its reliance on active renal tubular secretion rather than hepatic metabolism—and its pharmacodynamics regarding lactate metabolism. The most clinically significant interactions involve agents that compete for the Organic Cation Transporter 2 (OCT2) and Multidrug and Toxin Extrusion (MATE) proteins in the renal tubules; drugs such as cimetidine, dolutegravir, ranolazine, and vandetanib inhibit these transporters, significantly reducing metformin clearance and elevating plasma concentrations, which heightens the risk of toxicity. Concurrently, medications that impair renal perfusion or glomerular filtration, including NSAIDs, ACE inhibitors, ARBs, and diuretics, can precipitate acute renal insufficiency, leading to hazardous metformin accumulation and a subsequent risk of lactic acidosis. This specific risk also necessitates the temporary withholding of metformin during the administration of iodinated contrast media in vulnerable patients (usually those with eGFR <45 mL/min/1.73m²), as contrast-induced nephropathy can trigger a rapid spike in metformin levels. Beyond renal mechanisms, alcohol poses a severe pharmacodynamic interaction by inhibiting lactate clearance and gluconeogenesis, synergistically increasing the likelihood of lactic acidosis and hypoglycemia. Finally, while metformin monotherapy carries a low risk of hypoglycemia, its combination with insulin or insulin secretagogues (like sulfonylureas) requires careful dose calibration due to additive glucose-lowering effects.
Fig.3 Drug interaction
CONCLUSION
Metformin stands as a singular entity in the pharmacopoeia of metabolic medicine. Despite the rapid proliferation of novel antidiabetic agents—such as SGLT2 inhibitors and GLP-1 receptor agonists—Metformin retains its status as the undisputed first-line therapy for the management of Type 2 Diabetes Mellitus (T2DM). Its enduring dominance is attributed not merely to its historical precedence, but to a unique and multifaceted pharmacological profile that addresses the core pathophysiology of insulin resistance. The primary efficacy of Metformin lies in its ability to lower basal and postprandial plasma glucose without inducing hypoglycemia, a critical advantage over insulin secretagogues. By inhibiting hepatic gluconeogenesis and enhancing peripheral insulin sensitivity, primarily through the activation of AMP-activated protein kinase (AMPK), it offers a physiological approach to glucose control. Furthermore, its weight-neutral or modest weight-loss properties, combined with proven cardiovascular safety benefits, make it an ideal intervention for the overweight and obese demographic most affected by T2DM. While the drug is generally well-tolerated, clinical vigilance remains necessary. The prevalence of gastrointestinal side effects affects patient adherence, though these can often be mitigated through dose titration and extended-release formulations. Clinicians must also remain cognizant of the risk of Vitamin B12 deficiency during long-term use and the rare but serious risk of lactic acidosis, particularly in patients with significant renal impairment.
REFERENCES






R. V. Sivaprakash1, M. Gowtham2, S. Arunagiri2, S. Mahalakshmi2, N. Gnanasekar3, D. Rajalingam4, Metformin: A Comprehensive Clinical and Pharmacological Review of Its Spectrum, Efficacy, Resistance, and Safety Profile, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 518-525. https://doi.org/10.5281/zenodo.18484164
 10.5281/zenodo.18484164
10.5281/zenodo.18484164