
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shri Sai College of Pharmacy Khandala, Vaijapur, Chatrapati Sambhajinagar
Oral drug delivery remains the most preferred route of administration due to patient convenience and compliance; however, poor oral bioavailability continues to limit the therapeutic efficacy of many drugs, particularly those belonging to Biopharmaceutics Classification System (BCS) classes II and IV. Factors such as low aqueous solubility, inadequate intestinal permeability, extensive first-pass metabolism, and efflux transporter activity significantly compromise drug absorption following oral administration. To overcome these challenges, lipid-based nanocarrier systems have emerged as promising strategies, among which nanostructured lipid carriers (NLCs) represent a second-generation advancement over solid lipid nanoparticles. NLCs are composed of a blend of solid and liquid lipids stabilized by biocompatible surfactants, resulting in a structurally imperfect lipid matrix that enhances drug loading capacity and minimizes drug expulsion during storage. The unique composition and nanoscale size of NLCs facilitate improved drug solubilization in gastrointestinal fluids, promote lymphatic transport, reduce first-pass hepatic metabolism, and enhance intestinal permeability, collectively leading to improved oral bioavailability. This review provides a comprehensive overview of the principles of oral bioavailability, the relevance of BCS classification, and the design rationale of nanostructured lipid carriers.
Oral drug delivery remains the most preferred route of administration due to its convenience, non-invasiveness, cost-effectiveness, and high patient compliance[1]. It is particularly advantageous for chronic therapies and long-term disease management. Despite these benefits, the oral route presents significant challenges in achieving adequate and reproducible drug bioavailability[3].
Bioavailability is defined as the fraction of an administered dose of a drug that reaches the systemic circulation in an unchanged form[5]. In the context of oral drug delivery, bioavailability is influenced by several factors including drug solubility, dissolution rate, permeability across the intestinal membrane, stability in gastrointestinal fluids, and the extent of first-pass metabolism[6]. Poor oral bioavailability often results in reduced therapeutic efficacy, increased dose requirements, and higher inter-individual variability in drug response [36].Consequently, improving oral bioavailability is a critical objective in pharmaceutical formulation development[16].
To better understand and predict the factors affecting drug absorption, the Biopharmaceutics Classification System (BCS) was introduced[2]. The BCS categorizes drugs into four classes based on their aqueous solubility and intestinal permeability. BCS Class I drugs exhibit high solubility and high permeability and generally show good oral bioavailability[3]. BCS Class II drugs possess low solubility but high permeability, where dissolution rate is the major limiting factor for absorption. BCS Class III drugs show high solubility but low permeability, with absorption limited by intestinal transport[2]. BCS Class IV drugs exhibit both low solubility and low permeability and represent the most challenging group in terms of oral drug delivery[3].
A significant proportion of newly developed drug candidates belong to BCS Class II and IV, highlighting the growing need for advanced formulation strategies to enhance solubility and absorption[36]. Conventional dosage forms and traditional formulation approaches, such as salt formation, particle size reduction, and use of solubilizing agents, often fail to adequately address these challenges[16]. Moreover, physiological barriers such as enzymatic degradation, efflux transporters like P-glycoprotein, and extensive hepatic first-pass metabolism further reduce the oral bioavailability of many drugs[18].
In recent years, nanotechnology-based drug delivery systems have emerged as promising tools to overcome the limitations of conventional oral formulations[11]. Nanocarriers offer advantages such as small particle size, large surface area, enhanced drug solubilization, and improved interaction with biological membranes [12]. Among various nanocarrier systems, lipid-based drug delivery systems have gained considerable attention due to their biocompatibility, biodegradability, and ability to exploit natural lipid digestion and absorption pathways[19].
Nanostructured lipid carriers (NLCs) represent a second-generation lipid nanoparticle system developed to overcome the limitations associated with solid lipid nanoparticles (SLNs)[9]. SLNs, although effective in protecting drugs and providing controlled release, suffer from drawbacks such as low drug loading capacity and drug expulsion during storage due to the highly ordered crystalline structure of solid lipids[30]. NLCs were designed by incorporating a mixture of solid and liquid lipids, resulting in a less ordered lipid matrix with structural imperfections that allow higher drug loading and improved formulation stability[30].
Nanostructured lipid carriers typically consist of solid lipids, liquid lipids, and suitable surfactants for stabilization[10]. The presence of liquid lipids disrupts the crystalline lattice of solid lipids, creating void spaces within the matrix that can accommodate greater amounts of drug[12]. This unique structure enhances encapsulation efficiency and reduces the risk of drug expulsion during storage[32]. Additionally, the nanoscale size of NLCs enhances surface area, leading to improved dissolution and solubilization of poorly water-soluble drugs in gastrointestinal fluids[38].
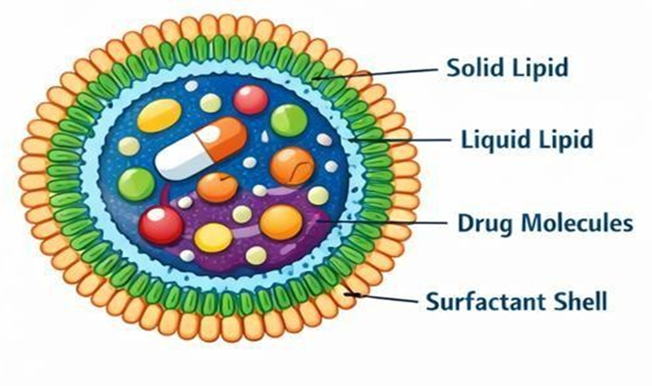
Fig. Structure of Nanostructured Lipid Carrier(NLCs)[10]
NLCs enhance oral bioavailability through multiple mechanisms[18]. Upon oral administration, lipid components undergo enzymatic digestion, forming mixed micelles that facilitate drug transport across the intestinal membrane[19]. NLCs can also promote lymphatic uptake of lipophilic drugs, thereby bypassing hepatic first-pass metabolism and increasing systemic drug exposure[49]. Furthermore, certain lipid excipients and surfactants used in NLC formulations have been reported to inhibit efflux transporters, further improving intestinal absorption[37]. NLCs also provide protection to encapsulated drugs against harsh gastrointestinal conditions, such as acidic pH and enzymatic degradation[32].
Due to these advantages, nanostructured lipid carriers have been extensively investigated for the oral delivery of BCS Class II and IV drugs, including anticancer, antidiabetic, anti-inflammatory, and cardiovascular agents[38]. Numerous preclinical studies have demonstrated improved pharmacokinetic profiles and enhanced therapeutic efficacy of NLC based formulations compared to conventional dosage forms[34].
Concept of Bioavailability
Oral bioavailability is defined as the fraction of an administered drug dose that reaches the systemic circulation in an unchanged form. It is a critical pharmacokinetic parameter influencing therapeutic effectiveness[2].
Bioavailability is influenced by several factors:
Formulation-related factors also play a vital role. The dosage form, particle size, crystalline state, and excipients used can influence drug dissolution and absorption[16]. Conventional oral dosage forms often fail to provide adequate bioavailability for drugs with poor solubility or stability[4].
Low bioavailability can result in reduced therapeutic effectiveness, higher dose requirements, and increased variability in patient response[36]. Therefore, improving bioavailability is a major objective in pharmaceutical formulation development[16]. Advanced drug delivery systems, including lipid-based and nanotechnology-based carriers, have been extensively explored to enhance oral bioavailability by improving solubility, protecting drugs from dgradation, and facilitating efficient absorption[19].
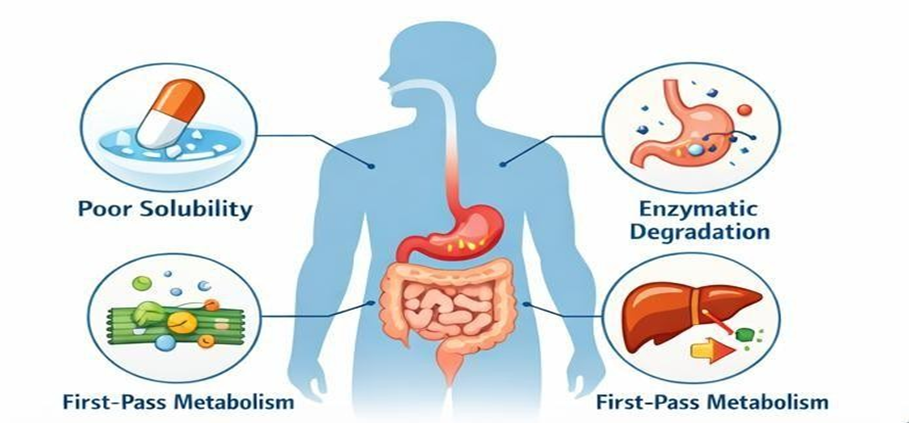
Fig. Challenges in Oral Bioavailability[1]
Lipid-Based Drug Delivery Systems
Lipid-based formulations have been extensively explored to enhance oral drug absorption[18].These systems improve drug solubilization and promote absorption via lipid digestion and micelle formation[49].
Common lipid-based systems include emulsions, microemulsions, and solid lipid nanoparticles (SLNs)[20]. Although SLNs offer advantages such as controlled drug release and protection from degradation, they suffer from limitations including low drug loading capacity, drug expulsion during storage, and crystalline instability[30]. These drawbacks led to the development of nanostructured lipid carriers[8].
Solid lipid nanoparticles (SLNs) represent the first generation of lipid nanoparticles developed to overcome the limitations of liquid lipid systems[7]. SLNs consist of solid lipids stabilized by surfactants and offer advantages such as controlled drug release, protection of drugs from chemical degradation, and improved stability[25]. However, SLNs exhibit inherent drawbacks including low drug loading capacity, drug expulsion during storage, and crystalline instability due to the highly ordered lipid matrix[30].
To address the limitations of SLNs, nanostructured lipid carriers (NLCs) were introduced as second-generation lipid nanoparticles[9]. NLCs are composed of a blend of solid and liquid lipids, creating a less ordered lipid matrix with structural imperfections that allow higher drug loading and enhanced formulation stability[10]. These systems provide superior performance in terms of encapsulation efficiency, controlled drug release, and long-term stability compared to SLNs[32].
Overall, lipid-based drug delivery systems offer a versatile and promising platform for improving oral bioavailability of poorly soluble drugs[18]. Their biocompatibility, ability to exploit physiological lipid absorption pathways, and flexibility in formulation design make them attractive candidates for advanced oral drug delivery[1]. Among lipid-based systems, nanostructured lipid carriers have gained significant attention due to their improved drug loading, stability, and bioavailability enhancement potential[32].
Nanostructured Lipid Carriers (NLCs)
1. Definition and Structure
Nanostructured lipid carriers are submicron colloidal carriers composed of a mixture of solid lipid, liquid lipid, and surfactant[10]. The incorporation of liquid lipids into the solid lipid matrix creates structural imperfections, allowing higher drug accommodation and enhanced stability[12].
2. Types of NLCs
Advantages of NLCs for Oral Bioavailability Enhancement
NLCs improve the apparent solubility of poorly water-soluble drugs by incorporating them into a lipid matrix, increasing drug availability in gastrointestinal fluids[1].
The nanoscale particle size provides a large surface area, leading to faster dissolution and enhanced absorption[22].
The lipid matrix protects drugs from acidic pH, enzymatic degradation, and chemical instability in the gastrointestinal tract[32].
NLCs promote lymphatic transport of lipophilic drugs, bypassing hepatic first-pass metabolism and increasing systemic bioavailability[18].
Lipidic nature and small size of NLCs improve interaction with the intestinal membrane, facilitating drug transport across the epithelium[37].
Certain lipids and surfactants used in NLCs inhibit P-glycoprotein (P-gp) efflux, reducing drug expulsion back into the intestinal lumen[6].
The presence of liquid lipids creates imperfections in the lipid matrix, allowing higher drug incorporation compared to solid lipid nanoparticles[12].
NLCs minimize drug expulsion during storage and show better long-term stability than conventional lipid carriers[30].
Selection of Components for Oral NLCs
Commonly used solid lipids include glyceryl monostearate, Compritol®, and stearic acid due to their safety and biocompatibility[10].
Medium-chain triglycerides and oleic acid are frequently used to enhance solubilization and drug loading[12].
Non-ionic surfactants such as Tween 80 and Poloxamers are preferred owing to their low toxicity and oral safety[20].
Methods of Preparation of NLCs
Nanostructured lipid carriers (NLCs) are colloidal carriers composed of a mixture of solid and liquid lipids stabilized by surfactants[10]. They are designed to overcome limitations of conventional lipid carriers such as low drug loading and stability issues[30]. The method of preparation plays a crucial role in determining particle size, drug loading, stability, and bioavailability[23]. NLCs are generally prepared by emulsifying the lipid phase into an aqueous surfactant solution, followed by particle size reduction and lipid solidification[25].
Based on the amount of energy applied for emulsification, NLC preparation methods can be classified into three main categories:
1. High-Energy Methods
High-energy methods rely on mechanical energy to reduce droplet size and generate stable lipid nanoparticles [23].
Mechanism / Steps:
Advantages: Reproducible , narrow particle size distribution, scalable[30].
Limitations: Not suitable for heat-sensitive drugs [25].
Examples:
2. Low-Energy Methods
Low-energy methods utilize the intrinsic chemical energy of the system, such as phase inversion or spontaneous emulsification, rather than external mechanical forces[47].
Mechanism / Steps:
Advantages: Gentle on thermolabile drugs, requires less equipment [20].
Limitations: May produce slightly larger particles, less uniform than high-energy Methods [47].
Examples:
3. Solvent-Based Methods
Solvent-based methods are suitable for heat-sensitive drugs and involve dissolving lipids in organic solvents [29].
Mechanism / Steps:
Advantages: Suitable for thermolabile drugs, controlled particle size [29].
Limitations: Requires complete solvent removal, environmental and toxicity Concerns[50].
Examples:
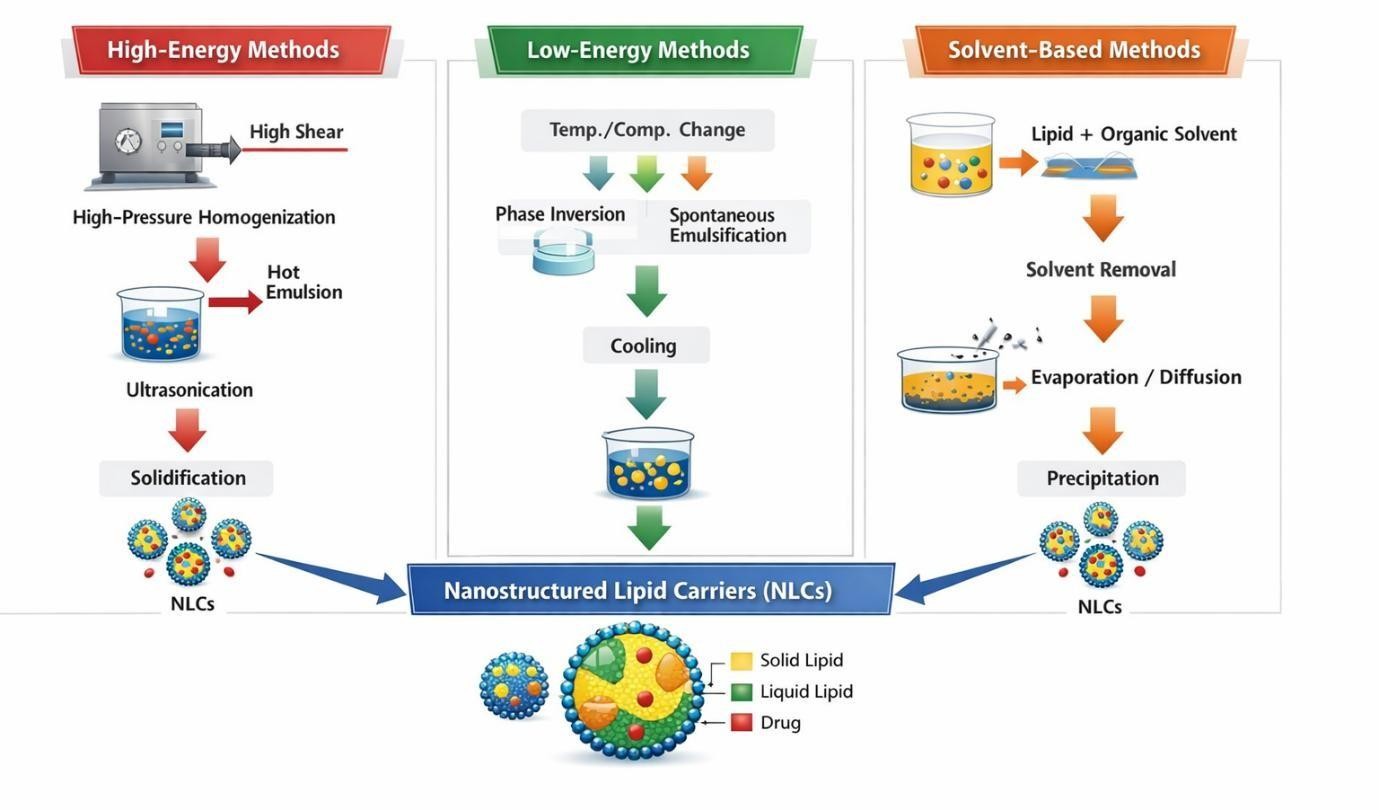
Fig. Method of Preparation of NLCs[23]
Characterization of NLCs
1. Particle Size and Polydispersity Index (PDI)
2. Zeta Potential
3. Morphology and Surface Structure
4. In Vitro Drug Release
In Vitro and Ex Vivo Evaluation
In vitro evaluation of NLCs includes dissolution studies and drug release kinetics, which predict drug release behavior in gastrointestinal conditions[48]. Lipolysis models simulate enzymatic digestion of lipids to assess drug solubilization and absorption potential [4]. Ex vivo studies use isolated intestinal tissues (e.g., rat or porcine intestines) to measure permeability and transport of the encapsulated drug[18]. These studies help determine intestinal uptake, membrane interaction, and potential bioavailability, providing critical data before in vivo testing. Overall, in vitro and ex vivo evaluations guide formulation optimization for effective oral delivery [26].
In Vivo Evaluation for Oral Bioavailability
In vivo studies are essential to assess the oral bioavailability of drugs delivered via NLCs. Pharmacokinetic parameters such as Cmax, Tmax, AUC, and half-life are measured after oral administration in animal models (e.g., rats or rabbits) and compared with conventional formulations[38]. NLCs often show higher systemic drug exposure, prolonged circulation, and reduced first-pass metabolism[14]. Studies also evaluate lymphatic uptake, tissue distribution, and therapeutic efficacy[31]. Overall, in vivo evaluation confirms the enhanced absorption, improved bioavailability, and sustained drug release provided by NLC-based formulations [32].
Applications of Nanostructured Lipid Carriers (NLCs) in Oral Drug Delivery
NLCs improve the solubility and gastrointestinal dissolution of lipophilic drugs, leading to increased oral bioavailability and systemic exposure [18].
Poorly water-soluble drugs classified under BCS class II benefit from NLC-based formulations due to enhanced dissolution rate and absorption[27].
NLCs enhance intestinal permeability and promote lymphatic uptake of BCS class IV drugs, thereby improving oral absorption [43].
Lipid-based NLC systems facilitate lymphatic transport of drugs, reducing hepatic firstpass metabolism and increasing bioavailability [31].
NLCs enable controlled drug release, improved therapeutic efficacy, and reduced systemic toxicity for orally administered anticancer drugs [44].
Encapsulation of antidiabetic agents in NLCs improves oral bioavailability and enhances pharmacological response [33].
NLC formulations provide prolonged drug release profiles, reducing dosing frequency and improving patient compliance[48].
The lipid matrix of NLCs protects encapsulated drugs from chemical and enzymatic degradation in the gastrointestinal tract[34].
Certain lipids and surfactants used in NLC formulations inhibit P-glycoprotein-mediated drug efflux, enhancing intestinal drug absorption [6].
NLCs improve the stability of chemically sensitive drugs by shielding them from harsh gastrointestinal conditions[38].
Limitations and Challenges of NLCs
Despite their numerous advantages, NLCs face several limitations and challenges that need to be addressed for successful formulation and clinical translation [30].
Future Perspectives
lipid carriers (NLCs) represent a promising platform for oral drug delivery, and Nanostructured ongoing research continues to expand their potential [38]. Future directions aim to overcome current limitations, improve clinical translation, and enable personalized therapy [39].
CONCLUSION
Nanostructured lipid carriers represent a versatile and efficient oral drug delivery system capable of significantly improving the bioavailability of poorly soluble drugs [14]. Their ability to overcome multiple physiological barriers makes them a promising platform for future oral pharmaceutical formulations [39].
REFERENCES



Ishwari Shinde, Prachi Udapurkar, Rupesh Tagad, Nanostructured Lipid Carriers as an Advanced Oral Drug Delivery System for Improving Bioavailability, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 4282-4294. https://doi.org/10.5281/zenodo.18790365
 10.5281/zenodo.18790365
10.5281/zenodo.18790365