
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
1Assistant Professor, Department of Pharmaceutics, Pandaveswar School of Pharmacy, Pandaveswar, Paschim Bardhaman, West Bengal, India.
2Assistant Professor, Department of Pharmacy, Shri Venkateshwara University, Gajraula, Uttar Pradesh, India.
3Professor & HOD, Department of Pharmacognosy, School of Pharmacy, Rai University, Ahmedabad, India.
4Associate Professor, Department of Pharmacology, Calcutta Institute of Pharmaceutical Technology and AHS, Banitabla, Uluberia, Howrah, West Bengal, India.
5Associate Professor, Department of Bio Technology and Life Sciences, Mangalayatan University, India.
6Research Scholar, Department of Pharmacy, Maharishi Markandeshwar Deemed to be University, Mullana-Ambala, India.
7Assistant Professor, Department of Pharmaceutics, School of Pharmacy, Rai University, Ahmedabad, India.
8Assistant Professor, Department of Pharmacology, School of Pharmacy, Rai University, Ahmedabad, India.
9Assistant Professor, Department of Pharmacology, JKKMMRF 'S Annai Jkk Sampoorani Ammal College of Pharmacy, The Tamilnadu Dr. MGR Medical University, India.
Background-Polycystic ovary syndrome (PCOS) is a prevalent and complex endocrine disorder affecting reproductive-aged women. It is characterized by reproductive, metabolic, and psychological manifestations, with pathophysiology involving genetic, hormonal, metabolic, and inflammatory crosstalk. Despite significant research advances, the heterogeneity of PCOS continues to challenge effective diagnosis and management. Objective- This review aims to provide a comprehensive overview of the cellular, molecular, and systemic mechanisms underlying PCOS, with emphasis on hormonal, metabolic, and inflammatory interactions. It also highlights clinical manifestations, therapeutic implications, and research gaps to guide future strategies. Methods- A narrative synthesis was conducted by reviewing literature from PubMed, Scopus, and Web of Science databases. Studies addressing pathophysiology, hormonal and metabolic mechanisms, immune and inflammatory crosstalk, clinical consequences, and therapeutic interventions in PCOS were included. Priority was given to recent high-impact studies, systematic reviews, and international guidelines. Results- The findings reveal that PCOS arises from the interplay of hypothalamic-pituitary-ovarian (HPO) axis dysfunction, hyperandrogenism, insulin resistance, chronic low-grade inflammation, and gut microbiota dysbiosis. At the cellular level, theca and granulosa cell dysfunction, impaired insulin signalling, mitochondrial dysfunction, and altered adipose–liver–ovary communication contribute to disease progression. Clinically, PCOS is associated with reproductive abnormalities, metabolic syndrome, cardiovascular risks, endometrial dysfunction, and psychological consequences. Current therapies—lifestyle modification, insulin sensitizers, oral contraceptives, and anti-androgens—offer partial relief, while emerging therapies such as GLP-1 agonists, AMPK activators, mTOR inhibitors, and microbiota-targeted strategies show promise. Research gaps include the need for integrative omics, systems biology approaches, patient-specific therapies, and longitudinal studies. Conclusion- PCOS is a multi-system disorder with interlinked endocrine, metabolic, and immune axes. Understanding the cellular and molecular underpinnings is crucial for developing holistic, precision-based, and long-term management strategies. A multidisciplinary approach is required to improve reproductive, metabolic, and psychological outcomes in affected women.
1.1 Definition and Global Prevalence of PCOS
Polycystic ovary syndrome (PCOS) is one of the most common endocrine disorders affecting women of reproductive age, characterized by a constellation of reproductive, metabolic, and hormonal abnormalities (Teede et al., 2018). It is defined by chronic anovulation, hyperandrogenism, and polycystic ovarian morphology, though clinical presentation can vary significantly among individuals. Globally, PCOS affects approximately 6–20% of women, depending on the diagnostic criteria applied and population studied (Bozdag et al., 2016; Lizneva et al., 2016). Such variation in prevalence highlights the complexity and heterogeneity of the disorder across different ethnic and geographic populations.
1.2 Clinical Heterogeneity and Diagnostic Criteria (Rotterdam, NIH, AE-PCOS)
PCOS is clinically heterogeneous, and multiple diagnostic frameworks have been developed. The NIH 1990 criteria define PCOS based on hyperandrogenism and chronic anovulation after exclusion of related disorders (Zawadski & Dunaif, 1992). The Rotterdam 2003 criteria expanded the definition by requiring the presence of any two of the following: oligo-/anovulation, clinical or biochemical hyperandrogenism, and polycystic ovarian morphology (ESHRE/ASRM, 2004). The Androgen Excess-PCOS Society (AE-PCOS) 2006 criteria emphasize androgen excess as a central feature, along with ovarian dysfunction (Azziz et al., 2006). While these differing criteria allow for broader identification, they also contribute to variability in reported prevalence and phenotypic subgroups.
1.3 Burden of PCOS as a Multi-System Disorder Beyond Reproduction
Although initially described as a reproductive condition, PCOS is now recognized as a multi-system disorder with implications extending beyond fertility. Women with PCOS often present with insulin resistance, obesity, dyslipidemia, and increased risk of metabolic syndrome and type 2 diabetes mellitus (Diamanti-Kandarakis & Dunaif, 2012). Cardiovascular risks such as hypertension and endothelial dysfunction are also heightened in this population (Escobar-Morreale, 2018). Moreover, PCOS has been associated with psychological consequences including anxiety, depression, and diminished quality of life (Cooney & Dokras, 2018). These systemic associations highlight the importance of understanding PCOS within a broader hormonal, metabolic, and inflammatory framework.
1.4 Aim and Scope of the Review
The aim of this review is to provide a comprehensive analysis of PCOS as a multi-system disorder, focusing on the interplay between hormonal, metabolic, and inflammatory pathways at the cellular and molecular levels. By synthesizing current knowledge, this review seeks to clarify mechanisms underlying disease pathophysiology, highlight clinical implications, and identify emerging therapeutic strategies. Particular emphasis is placed on the crosstalk among endocrine, metabolic, and immune systems, which collectively shape the heterogeneity and progression of PCOS.
2. Pathophysiology of PCOS: An Overview
PCOS pathophysiology is multifactorial, involving a complex interplay of genetic, hormonal, metabolic, and inflammatory mechanisms that disrupt ovarian function. This dysregulation leads to hyperandrogenism, anovulation, and systemic metabolic disturbances, manifesting as a multi-system disorder.
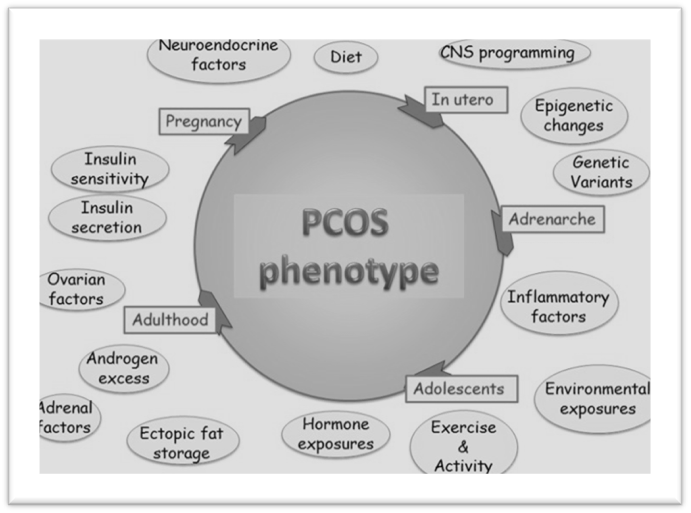
Figure 1. Schematic representation of the pathophysiology of polycystic ovary syndrome (PCOS)
2.1 Genetic Predisposition and Epigenetic Modifications
PCOS is considered a polygenic disorder with heritability estimates ranging from 40–70% (Day et al., 2018). Genome-wide association studies (GWAS) have identified susceptibility loci linked to gonadotropin secretion, insulin signaling, and androgen biosynthesis, including LHCGR, DENND1A, THADA, and FSHR genes (Goodarzi et al., 2015). Epigenetic modifications, such as DNA methylation, histone acetylation, and non-coding RNAs, have also been implicated in dysregulated ovarian and metabolic pathways (Xu et al., 2020).
Table 1. Genetic and Epigenetic Factors Implicated in PCOS
|
Mechanism |
Key Genes / Epigenetic Changes |
Functional Consequence |
References |
|
Genetic loci |
LHCGR, DENND1A, THADA, FSHR |
Altered gonadotropin signaling, folliculogenesis |
Goodarzi et al., 2015 |
|
Epigenetics |
DNA methylation in granulosa cells |
Dysregulated steroidogenesis |
Xu et al., 2020 |
|
Non-coding RNAs |
miR-93, miR-222, lncRNAs |
Insulin signaling and inflammation modulation |
Xu et al., 2020 |
|
Histone changes |
Histone acetylation defects |
Gene transcription dysregulation |
Day et al., 2018 |
2.2 Environmental and Lifestyle Influences
Beyond genetics, environmental and lifestyle factors significantly influence PCOS onset and severity. Sedentary lifestyle, obesity, and dietary habits exacerbate insulin resistance and hyperandrogenism (Lim et al., 2019). Prenatal androgen exposure and endocrine-disrupting chemicals (EDCs), such as bisphenol A (BPA), have been linked to altered ovarian development and neuroendocrine programming (Rattan et al., 2021). These findings support the “developmental origins” hypothesis, suggesting that early-life exposures may predispose women to PCOS later in life.
2.3 Central Role of Ovarian Dysfunction and Neuroendocrine Abnormalities
Ovarian dysfunction remains central to PCOS pathogenesis. Theca cells exhibit increased steroidogenic activity, producing excess androgens due to upregulation of enzymes such as CYP17A1 (Nelson et al., 2019). Granulosa cells show impaired aromatase activity, disrupting estrogen production and follicular maturation. Neuroendocrine alterations include increased GnRH pulsatility and elevated LH/FSH ratio, leading to chronic anovulation (Pastor et al., 2016). This dysregulation establishes a vicious cycle of androgen excess and impaired folliculogenesis.
2.4 PCOS as an Interplay of Hormonal, Metabolic, and Inflammatory Factors
PCOS is increasingly recognized as a multi-system disorder where hormonal, metabolic, and inflammatory pathways converge. Hyperinsulinemia amplifies ovarian androgen production and suppresses sex hormone-binding globulin (SHBG), worsening hyperandrogenism (Diamanti-Kandarakis & Dunaif, 2012). Chronic low-grade inflammation, characterized by elevated CRP, TNF-α, and IL-6, further contributes to insulin resistance and ovarian dysfunction (Escobar-Morreale, 2018). Adipose tissue dysfunction, oxidative stress, and gut microbiota dysbiosis are emerging factors reinforcing this systemic interplay.
Table 2. Interplay of Hormonal, Metabolic, and Inflammatory Pathways in PCOS
|
Pathway |
Key Features |
Cellular/Molecular Effect |
References |
|
Hormonal |
Hyperandrogenism, ↑ LH/FSH ratio |
Disrupted folliculogenesis |
Nelson et al., 2019 |
|
Metabolic |
Insulin resistance, dyslipidemia |
↑ Theca cell androgen synthesis, ↓ SHBG |
Diamanti-Kandarakis & Dunaif, 2012 |
|
Inflammatory |
↑ CRP, TNF-α, IL-6, oxidative stress |
Worsened insulin resistance, ovarian dysfunction |
Escobar-Morreale, 2018 |
|
Adipose tissue |
Dysregulated adipokines (leptin, adiponectin) |
Systemic metabolic dysfunction |
Lim et al., 2019 |
3. Hormonal Crosstalk in PCOS
3.1 Hypothalamic–Pituitary–Ovarian (HPO) Axis Dysfunction
The HPO axis plays a central role in PCOS pathogenesis. Women with PCOS exhibit altered GnRH pulsatility, favoring rapid pulses that preferentially stimulate luteinizing hormone (LH) over follicle-stimulating hormone (FSH) secretion (Pastor et al., 2016). This results in an elevated LH/FSH ratio, promoting theca cell androgen synthesis while impairing granulosa cell function. Consequently, folliculogenesis is disrupted, leading to anovulation and the accumulation of immature follicles within the ovary (Rosenfield & Ehrmann, 2016).
3.2 Hyperandrogenism
i) Ovarian Theca Cell Hyperactivity
Theca cells in PCOS exhibit intrinsic abnormalities, including upregulated expression of CYP17A1 (17α-hydroxylase/17,20-lyase), leading to excess androgen production (Nelson et al., 2019).
ii) Role of Insulin in Androgen Synthesis
Insulin acts synergistically with LH to stimulate theca cell steroidogenesis, while simultaneously suppressing hepatic sex hormone–binding globulin (SHBG), thereby increasing bioavailable testosterone (Diamanti-Kandarakis & Dunaif, 2012).
iii) Molecular Pathways
Dysregulation of CYP17A1, steroidogenic acute regulatory protein (STAR), and downstream AKT/PI3K signaling pathways has been implicated in enhanced androgen biosynthesis. Crosstalk between insulin receptor signaling and LH receptor pathways amplifies androgen excess, contributing to follicular arrest (Sen et al., 2020).
3.3 Progesterone Resistance and Impaired Endometrial Receptivity
Progesterone resistance is a hallmark of PCOS, characterized by reduced responsiveness of the endometrium to progesterone signaling (Piltonen et al., 2015). Impaired expression of progesterone receptors and altered downstream gene transcription lead to endometrial dysfunction, contributing to infertility, recurrent miscarriage, and increased risk of endometrial hyperplasia and cancer (Hu et al., 2018).
3.4 Adrenal Contribution to Androgen Excess
Although ovarian hyperandrogenism predominates, the adrenal glands contribute up to 30% of circulating androgens in women with PCOS (Azziz, 2016). Dysregulated adrenal steroidogenesis, involving 21-hydroxylase and 11β-hydroxylase pathways, results in elevated dehydroepiandrosterone sulfate (DHEAS) levels in a subset of patients (Yildiz et al., 2010). This adrenal contribution is particularly evident in lean PCOS phenotypes.
Table 3. Hormonal Dysregulation in PCOS
|
Hormonal Axis |
Key Abnormalities |
Cellular/Molecular Mechanism |
Clinical Implications |
References |
|
HPO Axis |
↑ GnRH pulsatility, ↑ LH/FSH ratio |
Excess LH drives theca cell androgen synthesis |
Anovulation, polycystic ovaries |
Pastor et al., 2016 |
|
Ovarian Androgens |
↑ CYP17A1, ↑ STAR, ↑ AKT/PI3K signaling |
Enhanced theca cell steroidogenesis |
Hirsutism, acne, infertility |
Nelson et al., 2019; Sen et al., 2020 |
|
Insulin Signaling |
Insulin–LH synergy, ↓ SHBG |
Amplified androgen bioavailability |
Metabolic dysfunction |
Diamanti-Kandarakis & Dunaif, 2012 |
|
Progesterone Axis |
Progesterone resistance in endometrium |
↓ PR expression, altered gene transcription |
Infertility, endometrial hyperplasia |
Piltonen et al., 2015 |
|
Adrenal Androgens |
↑ DHEAS production |
Dysregulated 21- and 11β-hydroxylase activity |
Hirsutism in lean PCOS |
Yildiz et al., 2010 |
4. Metabolic Dysregulation in PCOS
4.1 Insulin Resistance: Systemic and Tissue-Specific Mechanisms
Insulin resistance (IR) is present in up to 70% of women with PCOS, independent of obesity (Dunaif, 2012). It manifests in multiple tissues, including muscle, adipose, and liver, leading to impaired glucose utilization and compensatory hyperinsulinemia.
4.2 Hyperinsulinemia and Its Impact on Ovarian and Adrenal Androgen Production
Hyperinsulinemia acts synergistically with LH to enhance ovarian theca cell androgen biosynthesis through upregulation of CYP17A1 and STAR (Nelson et al., 2019). Insulin also decreases hepatic sex hormone–binding globulin (SHBG), increasing free testosterone levels (Diamanti-Kandarakis et al., 2006). In the adrenal cortex, hyperinsulinemia enhances androgen secretion by sensitizing steroidogenic enzymes, thereby contributing to elevated DHEAS in a subset of women (Azziz, 2016).
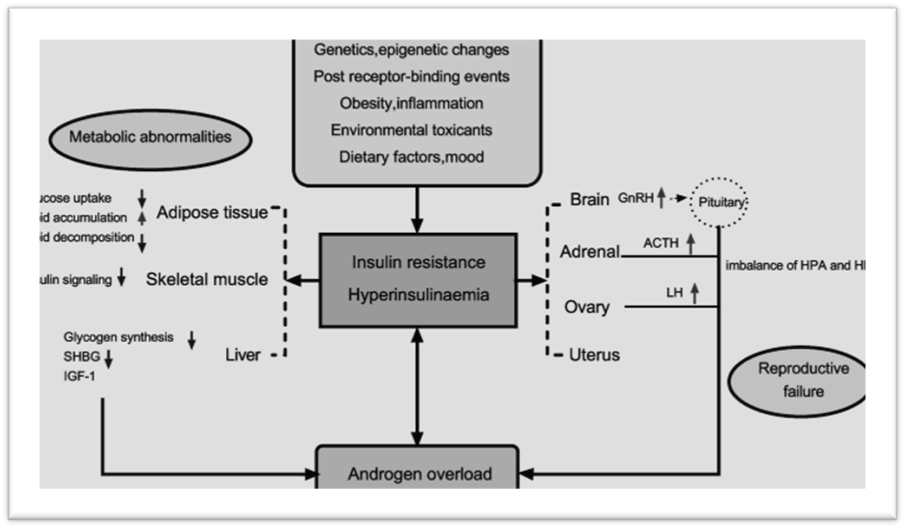
Figue 2. Schematic representation of Insulin resistance & hyperinsulinemia across tissues
4.3 Dyslipidemia and Altered Lipid Metabolism
PCOS is strongly associated with dyslipidemia, independent of BMI. Women typically present with elevated triglycerides, reduced HDL-C, and small dense LDL particles (Wild et al., 2011). Insulin resistance and hyperandrogenism alter hepatic lipoprotein metabolism by increasing VLDL synthesis and impairing lipolysis. These lipid disturbances contribute to long-term cardiovascular risks in PCOS.
4.4 Obesity, Adipokines (Leptin, Adiponectin, Resistin), and Energy Homeostasis
Obesity exacerbates metabolic dysfunction in PCOS, but even lean women can exhibit adipose tissue abnormalities. Adipokines play a crucial role in metabolic regulation:
This dysregulated adipokine profile contributes to a vicious cycle of energy imbalance, inflammation, and hormonal disruption in PCOS.
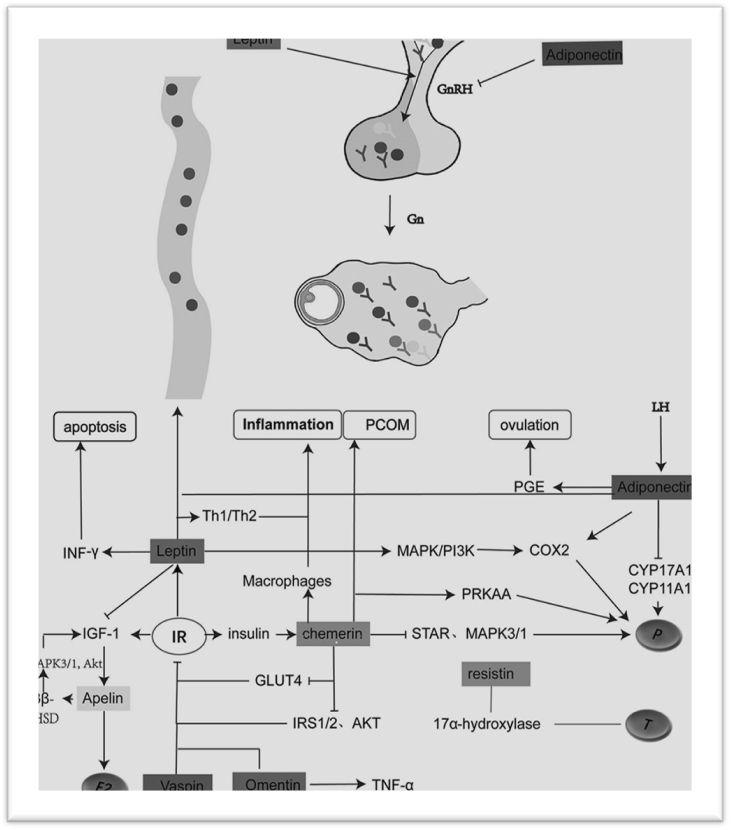
Figue 3: Adipokines (leptin, adiponectin, chemerin, resistin, etc.) in PCOS
Table 4. Metabolic Dysregulation in PCOS
|
Mechanism |
Key Features |
Molecular/Cellular Effect |
Clinical Consequence |
References |
|
Insulin Resistance |
Post-receptor signaling defects (IRS-1/2) |
↓ PI3K-AKT activity, ↓ GLUT4 translocation |
Hyperglycemia, compensatory hyperinsulinemia |
Dunaif, 2012 |
|
Adipose Inflammation |
↑ TNF-α, IL-6, macrophage infiltration |
Impaired insulin signaling |
Worsened IR, metabolic syndrome |
Gambineri et al., 2012 |
|
Mitochondrial Dysfunction |
↑ ROS, ↓ OXPHOS efficiency |
Impaired energy metabolism |
Fatigue, systemic IR |
Victor et al., 2009 |
|
Hyperinsulinemia |
↑ CYP17A1, ↓ SHBG |
↑ Ovarian and adrenal androgens |
Hirsutism, infertility |
Nelson et al., 2019 |
|
Dyslipidemia |
↑ TG, ↓ HDL, small dense LDL |
Altered hepatic lipoprotein metabolism |
Cardiovascular risk |
Wild et al., 2011 |
|
Adipokine Dysregulation |
↑ Leptin, ↓ Adiponectin, ↑ Resistin |
Energy imbalance, inflammation |
Obesity, IR, reproductive dysfunction |
Carmina et al., 2020; Panidis et al., 2003 |
5. Inflammatory Crosstalk in PCOS
5.1 Chronic Low-Grade Inflammation as a Hallmark of PCOS
Chronic low-grade inflammation is widely recognized as a core feature of PCOS and is observed across phenotypes, including lean and obese patients (Escobar-Morreale, 2018). Inflammatory activation in PCOS is systemic but also local (ovarian and adipose microenvironments), and it contributes to insulin resistance, altered steroidogenesis, and adverse reproductive outcomes (Diamanti-Kandarakis & Dunaif, 2012). Elevated markers of low-grade inflammation correlate with metabolic dysfunction and are thought to participate in the feed-forward cycle that links metabolic and reproductive abnormalities in PCOS.
5.2 Pro-inflammatory Cytokines (TNF-α, IL-6, IL-18, CRP)
Key circulating and tissue cytokines implicated in PCOS include tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), interleukin-18 (IL-18), and C-reactive protein (CRP). These mediators:
Clinical studies and meta-analyses show modest but consistent elevations of CRP, IL-6, and TNF-α in PCOS versus matched controls, independent of BMI in many reports (Escobar-Morreale, 2018).
5.3 Oxidative Stress and ROS Generation
Oxidative stress—marked by increased reactive oxygen species (ROS) production and reduced antioxidant defenses—is prevalent in PCOS and interrelates with inflammation and metabolic dysfunction (Victor et al., 2009). ROS contribute to:
Biomarkers such as increased lipid peroxidation products, lower glutathione levels, and altered antioxidant enzyme activity have been reported in multiple PCOS cohorts (Victor et al., 2009; Escobar-Morreale, 2018).
5.4 Immune Cell Involvement: Macrophages, T-cells, and NK Cells in Ovarian Microenvironment
Innate and adaptive immune cells are active participants in PCOS pathophysiology:
These immune changes create an inflammatory ovarian milieu that disrupts folliculogenesis, steroidogenesis, and endometrial function, thereby linking immune dysregulation with both metabolic and reproductive PCOS manifestations.
5.5 Gut Microbiota Dysbiosis and Systemic Inflammation
Emerging evidence implicates gut microbiota alterations (dysbiosis) in PCOS pathogenesis. Changes in microbial composition and reduced diversity have been associated with systemic inflammation, increased intestinal permeability, and metabolic dysregulation (Lindheim et al., 2017). Proposed mechanisms include:
Although causality remains under investigation, microbiome modulation (probiotics, prebiotics, dietary interventions) is an active area for therapeutic exploration.
Table 5. Key Inflammatory Mediators in PCOS and Their Effects
|
Mediator / Cell |
Source (Systemic / Local) |
Mechanistic Effects |
Clinical Associations |
Representative References |
|
TNF-α |
Adipose, ovarian stroma |
Impairs insulin signaling (IRS serine phosphorylation); promotes lipolysis and inflammation |
Insulin resistance, hyperandrogenism |
Diamanti-Kandarakis & Dunaif, 2012; Escobar-Morreale, 2018 |
|
IL-6 |
Adipose, immune cells |
Stimulates hepatic CRP, influences hepatic glucose production |
Metabolic syndrome markers, inflammation |
Escobar-Morreale, 2018 |
|
IL-18 |
Immune cells, adipose |
Pro-inflammatory; linked to metabolic risk |
Cardiometabolic risk |
Escobar-Morreale, 2018 |
|
CRP |
Hepatic (IL-6 driven) |
Systemic inflammation marker; prognostic for CV risk |
Elevated in PCOS; correlates with IR |
Escobar-Morreale, 2018 |
|
ROS / Oxidative stress |
Mitochondria, inflamed tissues |
Damages signaling proteins; impairs insulin action and follicular function |
Linked to infertility, metabolic dysfunction |
Victor et al., 2009 |
|
Macrophages (M1) |
Adipose, ovary |
Secrete TNF-α, IL-1β; promote local insulin resistance |
Adipose inflammation, ovarian dysfunction |
Gambineri et al., 2012 |
|
T-cells (Th17/Treg imbalance) |
Ovarian, systemic |
Promote auto-inflammatory signaling; alter tolerance |
Possible contribution to reproductive pathology |
Escobar-Morreale, 2018 |
|
Gut microbiota dysbiosis |
Intestinal microbiome |
↑ LPS translocation, altered metabolites → systemic inflammation |
Metabolic and reproductive phenotype modulation |
Lindheim et al., 2017 |
Table 6. Therapeutic Strategies Targeting Inflammation and Oxidative Stress
|
Strategy |
Mechanism of Action |
Evidence / Rationale |
|
Lifestyle / weight loss |
Reduces adipose inflammation and systemic cytokines |
Weight loss lowers CRP, improves IR and ovulation (general clinical evidence) |
|
Omega-3 fatty acids & antioxidants (vitamin E, C, N-acetylcysteine) |
Anti-inflammatory, ROS scavenging |
Small RCTs show metabolic and ovulatory benefits in PCOS subgroups |
|
Metformin |
Lowers insulin & indirectly reduces inflammatory mediators |
Improves insulin sensitivity and may decrease CRP/IL-6 |
|
Probiotics / prebiotics |
Modulate gut microbiome, reduce LPS translocation |
Pilot studies suggest metabolic benefit; more RCTs needed |
|
Targeted anti-cytokine therapy (experimental) |
Direct inhibition of pro-inflammatory cytokines |
Theoretical; limited clinical data in PCOS specifically |
6. Cellular and Molecular Interactions
6.1 Theca and Granulosa Cell Dysfunction
PCOS ovaries exhibit intrinsic cellular abnormalities, particularly in theca and granulosa cells, which underpin altered folliculogenesis and steroidogenesis.
6.2 Insulin Receptor and Downstream Signaling Defects
Insulin resistance in PCOS extends to post-receptor signaling defects, particularly at the level of IRS-1/IRS-2 phosphorylation.
6.3 Mitochondrial Dysfunction and Impaired Cellular Energy Metabolism
Mitochondrial defects are increasingly recognized in PCOS. Altered mitochondrial morphology, reduced mtDNA copy number, and impaired oxidative phosphorylation have been reported in ovarian, adipose, and skeletal muscle tissues (Victor et al., 2009). These abnormalities:
6.4 Crosstalk Between Adipose Tissue, Liver, and Ovary
The multi-organ nature of PCOS involves bidirectional communication:
This adipose–liver–ovary axis underscores PCOS as a systemic metabolic–reproductive disorder rather than an isolated ovarian condition.
Table 7. Cellular and Molecular Abnormalities in PCOS
|
Site / Cell Type |
Molecular Alteration |
Functional Consequence |
Clinical Outcome |
|
Theca cells |
↑ CYP17A1, ↑ CYP11A1 |
Excess androgen biosynthesis |
Hyperandrogenism |
|
Granulosa cells |
↓ CYP19A1 aromatase; ↑ AMH |
Impaired estradiol production, follicular arrest |
Anovulation |
|
Insulin receptor/IRS |
Serine > tyrosine phosphorylation |
Defective PI3K–AKT signaling |
Insulin resistance |
|
Mitochondria |
↓ mtDNA, impaired oxidative phosphorylation |
↑ ROS, ↓ energy metabolism |
Infertility, metabolic dysfunction |
|
Adipose tissue |
↑ TNF-α, IL-6; ↓ adiponectin |
Inflammation, insulin resistance |
Obesity-linked PCOS |
|
Liver |
↑ gluconeogenesis, dyslipidemia |
Hyperinsulinemia, lipid accumulation |
Metabolic syndrome |
7. Clinical Manifestations Linked to Multi-System Crosstalk
7.1 Reproductive Outcomes: Anovulation, Infertility, Miscarriage Risk
PCOS is the most common cause of anovulatory infertility. Follicular arrest due to granulosa cell dysfunction, excess AMH, and disrupted gonadotropin secretion leads to chronic anovulation (Franks et al., 2008). Infertility is compounded by impaired endometrial receptivity, and PCOS women have a higher risk of miscarriage, partly attributable to hyperinsulinemia, hyperandrogenism, and chronic inflammation (Palomba et al., 2015).
7.2 Metabolic Syndrome and Type 2 Diabetes Mellitus
PCOS is strongly associated with metabolic syndrome, defined by abdominal obesity, insulin resistance, dyslipidemia, and hypertension (Ehrmann, 2005). Longitudinal studies confirm a 3–5 fold increased risk of type 2 diabetes mellitus (T2DM) in PCOS women, independent of BMI (Legro et al., 2013).
7.3 Cardiovascular Complications: Hypertension, Atherosclerosis
Chronic low-grade inflammation, endothelial dysfunction, and dyslipidemia predispose PCOS women to hypertension and premature atherosclerosis. Although absolute cardiovascular event risk remains debated due to age-related confounding, subclinical markers (carotid intima-media thickness, coronary artery calcification) are consistently elevated in PCOS cohorts (Wild et al., 2010).
7.4 Endometrial Dysfunction and Cancer Risk
Unopposed estrogen exposure due to chronic anovulation, along with hyperinsulinemia, increases the risk of endometrial hyperplasia and carcinoma in PCOS women (Chittenden et al., 2009). Defects in progesterone signaling, chronic inflammation, and altered immune surveillance exacerbate this vulnerability.
7.5 Psychological and Neurological Aspects: Anxiety, Depression, Cognitive Changes
PCOS is associated with significantly higher prevalence of anxiety, depression, and reduced quality of life (Dokras et al., 2011). Emerging data also suggest cognitive alterations, potentially linked to chronic inflammation, insulin resistance, and hyperandrogenemia, although findings are still preliminary.
Table 8. Major Clinical Manifestations of PCOS and Their Pathophysiological Drivers
|
Clinical Manifestation |
Underlying Pathophysiology |
Key Risk Amplifiers |
|
Anovulation, infertility |
Follicular arrest, high AMH, endometrial dysfunction |
Hyperinsulinemia, obesity |
|
Miscarriage risk |
Poor oocyte/embryo quality, endometrial inflammation |
IR, hyperandrogenism |
|
Type 2 diabetes mellitus |
Insulin resistance, β-cell dysfunction |
Obesity, family history |
|
Hypertension, atherosclerosis |
Dyslipidemia, endothelial dysfunction, chronic inflammation |
Obesity, IR |
|
Endometrial carcinoma |
Unopposed estrogen, progesterone resistance |
Obesity, chronic anovulation |
|
Anxiety, depression |
Chronic disease burden, inflammation, neuroendocrine dysregulation |
Young age at diagnosis, obesity |
8. Therapeutic Implications
8.1 Current Management Strategies
Lifestyle modification, including dietary interventions, weight reduction, and physical activity, remains the cornerstone of PCOS management, with evidence showing improvements in insulin sensitivity, ovulation, and menstrual regularity (Moran et al., 2017). Pharmacological therapies such as oral contraceptives (OCPs) are used to regulate menstrual cycles and reduce hyperandrogenic symptoms, while metformin improves insulin resistance and metabolic outcomes (Teede et al., 2018). Anti-androgens such as spironolactone and flutamide may be prescribed for hirsutism, while GLP-1 receptor agonists have emerged as novel agents for weight management and metabolic regulation in PCOS (Jensterle et al., 2020).
8.2 Targeting Inflammatory and Metabolic Pathways
Given the role of chronic inflammation and metabolic dysregulation in PCOS, anti-inflammatory interventions such as omega-3 fatty acids, vitamin D, and antioxidants have been shown to reduce circulating cytokines and improve insulin sensitivity (Asemi et al., 2014; González et al., 2012). Insulin sensitizers, beyond metformin, such as thiazolidinediones (TZDs), also demonstrate beneficial effects on glucose metabolism and ovarian function but may be limited by adverse effects (Ehrmann, 2016).
8.3 Emerging Therapies
Recent advances focus on molecular targets including AMPK activators and mTOR inhibitors, which aim to restore metabolic balance and regulate follicular development (Diamanti-Kandarakis & Dunaif, 2012). Modulation of the gut microbiota through probiotics and prebiotics has gained interest, given emerging evidence linking dysbiosis with systemic inflammation and insulin resistance in PCOS (Qi et al., 2019). Personalized and precision medicine approaches, integrating genetic, metabolic, and inflammatory profiling, may help tailor therapies to individual PCOS phenotypes.
9. Future Directions and Research Gaps
9.1 Integrative Omics in PCOS Research
The application of genomics, proteomics, and metabolomics is essential to unravel complex PCOS pathophysiology and identify novel biomarkers (Goodarzi et al., 2015).
9.2 Systems Biology Approaches
PCOS reflects a network of interactions across endocrine, metabolic, and immune systems. Systems biology and computational modeling approaches are needed to map these interactions and predict treatment responses (McCartney & Marshall, 2016).
9.3 Patient-Specific Therapeutic Strategies
Current treatments are often symptom-driven rather than pathophysiology-targeted. Future therapies must be tailored to the unique clinical and molecular phenotype of each patient, accounting for genetic predisposition, metabolic risk, and inflammatory profile.
9.4 Longitudinal Studies
There is a lack of long-term studies assessing the progression of metabolic, cardiovascular, and inflammatory risks in PCOS. Addressing this gap will guide preventive and therapeutic strategies across the lifespan.
10. CONCLUSION
PCOS is increasingly recognized as a multi-system disorder involving a complex interplay of hormonal, metabolic, and inflammatory pathways. At the cellular and molecular level, ovarian dysfunction, insulin resistance, adipose tissue inflammation, and immune activation form a self-perpetuating cycle that underpins both reproductive and systemic manifestations. A deeper understanding of these crosstalk mechanisms will pave the way for innovative therapeutic targets and personalized medicine approaches. Ultimately, a holistic management strategy addressing reproductive, metabolic, cardiovascular, and psychological aspects is essential to improve long-term outcomes for women with PCOS.
REFERENCES









Sumit Ghosh, Yash Srivastav, Shonu Jain, Nilanjan Pahari, Soni Singh, Johny Lakra, Sujit Kumar Panda, Jayalekshmi M., Srinivas Venkatraman*, Polycystic Ovary Syndrome as a Multi-System Disorder: A Comprehensive Review of Hormonal, Metabolic, and Inflammatory Crosstalk at the Cellular and Molecular Level, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 9, 365-383 https://doi.org/10.5281/zenodo.17047472
 10.5281/zenodo.17047472
10.5281/zenodo.17047472