
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
PDEA's Shankarrao Ursal College of Pharmaceutical Sciences & Research Centre, Savitribai Phule Pune University, Pune, Maharashtra, India
Artificial Intelligence (AI) has become a powerful and transformative tool in pharmaceutical sciences, providing advanced approaches to overcome traditional challenges in drug discovery, development, formulation, and clinical decision- making. The pharmaceutical sector is under continuous pressure to shorten development timelines, reduce costs, and improve the success rate of new drug candidates while ensuring safety and therapeutic effectiveness. AI-based technologies such as machine learning (ML), deep learning (DL), natural language processing (NLP), and generative modeling have significantly improved the processes of target identification, molecular interaction prediction, and lead optimization. These computational techniques allow rapid screening of extensive chemical databases, accurate prediction of pharmacokinetic and toxicological (ADMET) profiles, simulation of virtual clinical trials, and implementation of personalized treatment strategies using real-world data. Furthermore, AI contributes to pharmaceutical formulation development by predicting drug–excipient compatibility, optimizing manufacturing parameters, and maintaining product quality through advanced Quality by Design (QbD) frameworks. In pharmacovigilance, AI enhances the monitoring of drug safety by enabling automated adverse event detection, signal generation, and risk evaluation through analysis of electronic health records, clinical databases, and social media sources. Although AI offers significant advantages, its widespread adoption faces challenges including data security issues, lack of algorithm transparency, regulatory uncertainties, and the requirement for robust, high-quality datasets. This review outlines the present applications, recent technological developments, benefits, and limitations of AI in pharmaceutical research, emphasizing its expanding role in driving innovation and improving global healthcare outcomes.
The pharmaceutical sector is currently experiencing a significant digital evolution driven by rapid progress in Artificial Intelligence (AI). Traditionally, drug discovery and development have depended largely on experimental screening methods, iterative trial-and-error approaches, and prolonged preclinical and clinical evaluations. These conventional processes often extend over 10 to 15 years and require substantial financial investment, frequently amounting to billions of dollars. Despite these efforts, only a limited proportion of investigated drug candidates ultimately achieve regulatory approval. The growing complexity of diseases, increasing demand for targeted and personalized therapies, and the urgent need to control research expenditures have intensified the search for more efficient, data-driven methodologies. In this scenario, AI has emerged as a transformative technology capable of accelerating pharmaceutical research through intelligent automation, predictive analytics, and computational modeling.
Artificial Intelligence encompasses computational systems designed to replicate aspects of human cognition, including learning, reasoning, pattern recognition, and decision-making. Within pharmaceutical research, AI tools such as machine learning (ML), deep learning (DL), neural networks, natural language processing (NLP), and generative modeling techniques are extensively applied to interpret and analyze large-scale datasets. These datasets originate from genomics, proteomics, metabolomics, chemical libraries, clinical trials, and real-world clinical evidence. By identifying hidden correlations and predicting molecular and biological behavior, AI assists researchers in designing safer, more effective therapeutic molecules while reducing experimental burden.
Fig 1: Artificial intelligence
One of the most impactful contributions of AI lies in drug discovery. Advanced computational models facilitate virtual screening of extensive compound libraries, prediction of pharmacological activity, molecular docking simulations, de novo molecule design, and identification of novel biological targets. AI-based platforms significantly shorten the timeline for lead identification and structural optimization. In addition, predictive models for ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) properties help identify potential safety concerns early in development, thereby decreasing late-stage clinical failures.
Beyond discovery, AI also plays an important role in pharmaceutical formulation and manufacturing. In formulation development, AI supports rational excipient selection, stability prediction, and optimization of formulation variables under Quality by Design (Qbd) principles. During manufacturing, AI-driven systems enable real-time process monitoring, predictive maintenance, and process optimization, contributing to the implementation of Industry 4.0 concepts in pharmaceutical production.
In clinical research, AI enhances trial efficiency by improving patient recruitment strategies, optimizing study design, predicting treatment outcomes, and enabling adaptive trial management. The ability of AI systems to analyze large electronic health record (EHR) databases and clinical datasets facilitates biomarker identification, patient stratification, and the development of personalized therapeutic strategies. Furthermore, in pharmacovigilance, AI-powered tools— particularly NLP and automated signal detection systems—improve adverse drug reaction (ADR) monitoring. By mining data from scientific publications, medical reports, social media, and global safety databases, AI strengthens post-marketing surveillance and risk assessment.
Despite its considerable advantages, the implementation of AI in pharmaceutical research presents several challenges. Concerns related to data integrity, privacy protection, model interpretability, regulatory approval pathways, and ethical considerations remain significant barriers. Regulatory authorities such as the FDA and EMA are progressively developing frameworks to guide the validation and use of AI-based tools in drug development. Additionally, effective integration of AI requires interdisciplinary collaboration among pharmaceutical scientists, data analysts, clinicians, and regulatory professionals.
Overall, the incorporation of Artificial Intelligence into pharmaceutical research represents a paradigm shift toward faster, more precise, and cost-effective drug development. As AI technologies continue to mature, they are expected to play a central role in advancing personalized medicine, disease modeling, safety evaluation, and therapeutic innovation. This review therefore examines current developments, applications, limitations, and future perspectives of AI in the pharmaceutical field, emphasizing its transformative impact on global healthcare systems.
CORE AI METHODOLOGIES IN THE PHARMACEUTICAL FIELD
Artificial Intelligence in the pharmaceutical sector is supported by multiple computational methodologies that enable efficient drug discovery, development, and healthcare innovation. The major AI techniques applied in pharmaceutical research are described below:
1. Machine Learning (ML):
Machine Learning serves as the foundation of AI applications in pharmaceuticals. It enables computer systems to learn patterns from experimental, chemical, and clinical datasets without explicit rule-based programming. ML models continuously improve their predictive performance as more data becomes available.
Applications:
Common ML Algorithms:
2. Deep Learning (DL)
Deep Learning is an advanced subset of machine learning that employs multi-layered artificial neural networks to analyze highly complex and high-dimensional datasets. It is particularly effective in handling molecular representations, imaging data, and biological sequences.
Applications:
Common DL Architectures:
3. Natural Language Processing (NLP)
Natural Language Processing enables AI systems to interpret and analyze unstructured textual data such as scientific publications, electronic health records (EHRs), regulatory documents, and clinical trial reports.
Applications:
4. Generative Artificial Intelligence (GenAI)
Generative AI models are designed to create new molecular entities with specific biological or physicochemical characteristics. These models explore chemical space efficiently and accelerate innovative drug design.
Types of Generative Models:
Applications:
5. Predictive Modeling and QSAR
Quantitative Structure–Activity Relationship (QSAR) modeling utilizes AI algorithms to establish correlations between chemical structure and biological activity or toxicity. AI-enhanced QSAR models provide improved predictive accuracy.
Applications:
6. Graph-Based AI Models
Many pharmaceutical datasets, including molecular structures, proteins, and biological pathways, possess inherent graph structures. Graph-based AI models learn from these interconnected data representations.
Applications:
Common Models:
7. Reinforcement Learning (RL)
Reinforcement Learning enables AI systems to make decisions through iterative trial-and-error processes guided by reward-based feedback mechanisms.
Applications:
8. Computer Vision
Computer Vision allows AI systems to analyze visual biomedical and pharmaceutical data using image processing and pattern recognition techniques.
Applications:
APPLICATIONS OF ARTIFICIAL INTELLIGENCE (AI) IN PHARMACEUTICAL RESEARCH
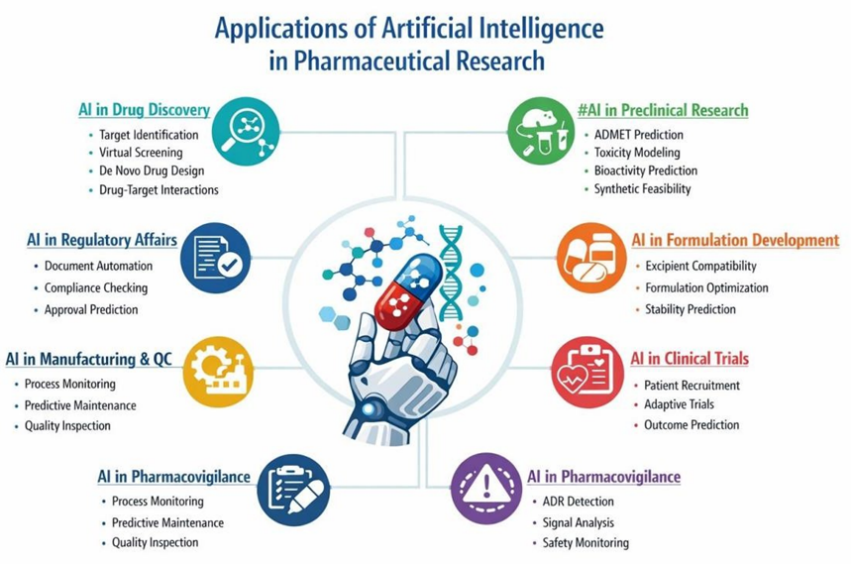
Fig no 2: Application of artificial intelligence in pharmaceutical research
1. AI in Drug Discovery
AI helps identify potential drug molecules faster and more accurately.
Applications:
2. AI in Preclinical Research
AI reduces unnecessary animal testing and predicts drug behavior before laboratory experiments.
Applications:
3. AI in Formulation and Product Development
Formulation scientists use AI to optimize dosage forms efficiently.
Applications:
4. AI in Clinical Trials
AI significantly improves the efficiency, cost-effectiveness, and accuracy of clinical trials.
Applications:
5. AI in Pharmacovigilance
AI automates the detection and monitoring of adverse drug reactions (ADRs).
Applications:
6. AI in Manufacturing and Quality Control
AI enhances manufacturing efficiency and ensures consistent product quality.
Applications:
7. AI in Regulatory Work and Documentation
AI simplifies regulatory submission processes and documentation management
Applications:
ADVANTAGES OF AI IN PHARMACEUTICAL FIELD
1. Accelerated Drug Discovery and Development: Artificial Intelligence processes vast biological and chemical datasets rapidly, significantly shortening the time needed to identify drug targets and promising molecules. Techniques such as virtual screening and computational modeling reduce dependence on time-consuming laboratory experiments.
2. Lower Research and Development Expenses: AI reduces repetitive trial- and-error experimentation and helps identify unsuitable drug candidates early by predicting toxicity, safety, and effectiveness, thereby decreasing overall development costs.
3. Greater Accuracy and Predictive Capability: Machine learning algorithms uncover hidden patterns within complex datasets that may not be detectable manually. They provide reliable predictions regarding drug–target binding, ADMET characteristics, and potential toxic effects.
4. Advanced Formulation Optimization: AI assists in selecting optimal polymer ratios, ensuring excipient compatibility, and improving dissolution behavior. It also forecasts product stability and shelf life with greater efficiency.
5. Higher Clinical Trial Success Rates: AI enhances study design and streamlines patient recruitment by identifying suitable patient populations. It can also forecast treatment responses, increasing the probability of successful clinical outcomes.
6. Automated Pharmacovigilance Systems: AI monitors adverse drug reactions using real-world evidence such as electronic health records and social media data, offering faster and more precise safety surveillance compared to traditional manual methods.
7. Support for Personalized and Precision Medicine: By analyzing genomic and clinical data, AI enables customized therapy plans, optimized dosing strategies, and prediction of individual patient responses to medications.
8. Improved Manufacturing and Quality Assurance: AI facilitates continuous monitoring of production processes and applies predictive maintenance strategies to avoid equipment breakdowns, ensuring consistent product quality and minimizing defects.
9. Faster and Informed Decision-Making: AI delivers real-time analytical insights to researchers, formulation scientists, and clinicians, supporting quicker and more evidence-based decisions during development and regulatory evaluation.
10. Efficient Data Handling and Analysis: AI effectively manages large-scale biomedical, chemical, and clinical information and extracts meaningful insights from unstructured sources such as research articles and technical reports.
ROLES OF AI IN PHARMACEUTICAL FIELD
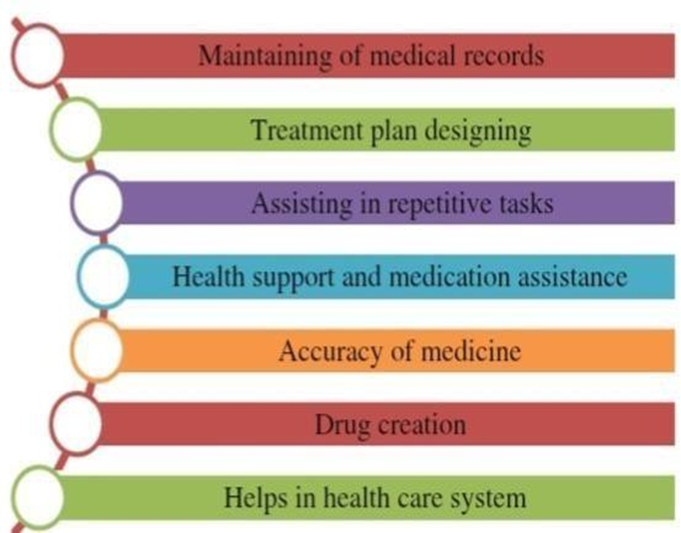
Fig 3: Role of AI in Pharmaceutical Field
1. Management of Medical Records: Artificial Intelligence systematically stores, arranges, and evaluates patient information and clinical histories, enabling researchers and healthcare professionals to retrieve precise and well-structured data to support drug research and development.
2. Development of Personalized Treatment Strategies: AI examines individual patient data to design customized therapy plans. By analyzing genetic makeup and disease characteristics, it predicts the most suitable and effective treatment for each patient.
3. Automation of Routine Activities: AI streamlines repetitive responsibilities such as documentation, report preparation, and laboratory operations. This automation reduces the likelihood of human error and enhances overall productivity in pharmaceutical research environments.
4. Patient Health Monitoring and Medication Support: AI-based systems assist patients by monitoring medication compliance, sending reminders, and tracking health parameters. These tools also facilitate clinical trials through the collection of real-time patient information.
5. Enhancement of Drug Accuracy and Quality: AI improves precision in drug formulation by forecasting stability, excipient compatibility, and appropriate dosage levels, thereby reducing formulation errors and ensuring high-quality pharmaceutical products.
6. Drug Discovery and Molecular Development: AI supports the identification of novel drug candidates, predicts interactions between drugs and biological targets, and accelerates lead optimization processes, ultimately decreasing the time and expense required to develop safe and effective medications.
7. Advancement of Healthcare Infrastructure: AI strengthens healthcare systems by improving diagnostic accuracy, supporting clinical decision- making, and predicting disease trends, while promoting better collaboration among pharmaceutical industries, hospitals, and public health organizations.
REGULATORY, ETHICAL, AND PRACTICAL CONSIDERATIONS
1. Regulatory Considerations:
Regulatory bodies like FDA, EMA, and ICH are creating frameworks for AI use in pharmaceuticals.
Important Regulatory Documents
2. Ethical Considerations:
Ethics play a major role in AI adoption due to patient data and automated decisions.
3. Practical Considerations:
While AI provides major benefits, practical challenges exist in real-world implementation.
CHALLENGES AND LIMITATIONS OF AI IN PHARMACEUTICS
FUTURE SCOPE OF AI IN PHARMACEUTICAL FIELD :
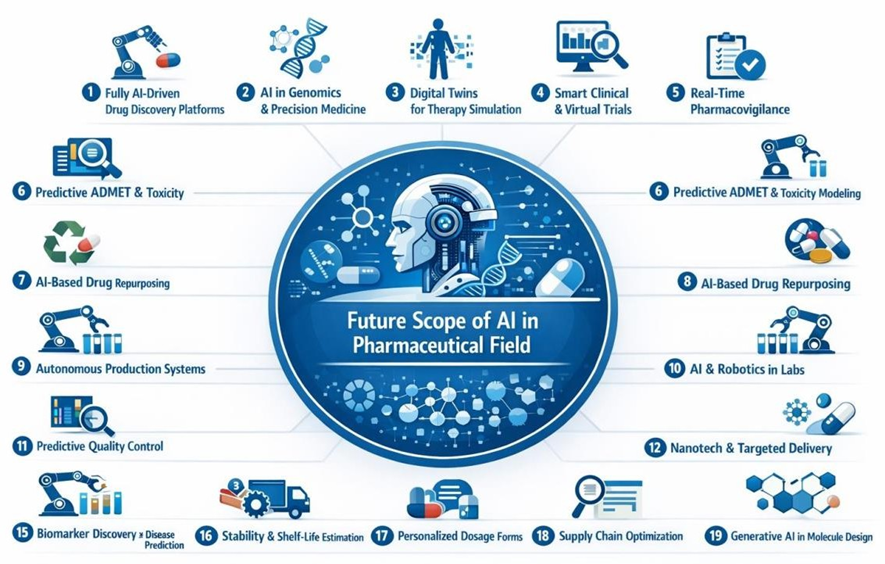
Fig 4: Future Scope Of AI In Pharmaceutical Field
CONCLUSION
Artificial intelligence is transforming pharmaceutical research by enabling faster, smarter, and more accurate scientific decision-making across the entire drug development pipeline. From target identification and virtual screening to formulation design, clinical trials, and pharmacovigilance, AI significantly reduces time, cost, and failure rates. Its ability to analyze large datasets, predict outcomes, and automate complex processes has made it an essential tool for modern drug discovery and development.
Although challenges such as data privacy, algorithmic bias, and regulatory limitations still exist, ongoing advancements in machine learning, deep learning, and big data analytics continue to strengthen AI’s role. With continuous improvements, AI will pave the way for personalized medicine, autonomous laboratories, and highly efficient pharmaceutical systems. Overall, AI holds immense potential to revolutionize the pharmaceutical industry, making drug development more precise, faster, and more cost-effective than ever before.
REFERENCES




Suraj Zunzulde, Tanuja Tandale, Dr. Sujit Kakade, Dr. Ashok Bhosale, Review on Artificial Intelligence (AI) in Pharmaceutical Field, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 3629-3640. https://doi.org/10.5281/zenodo.18730246
 10.5281/zenodo.18730246
10.5281/zenodo.18730246