
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacy (Pharmacology), Institute of Technology & Management, Gorakhpur, Uttar Pradesh, India 273209
The flowers of Acacia nilotica (commonly known as Babul) are used in traditionally medicine to treat distress and inflammation. This review collects and carefully evaluates the current literature on the analgesic potential of A. nilotica (Vachellia nilotica) flowers and their pharmacological actions. Plant chemicals have investigated the containing compounds flavonoids, phenolic acids, tannins, terpenoids, and alkaloids, their capacity to regulate like prostaglandins, NSAIDs and cytokines, etc. (pain & inflammation). Vachellia nilotica (Acacia) has impressive analgesic and antipyretic actions through PNS and central processes, perhaps by inhibiting prostaglandin production, free radical scavenging action, and opioid. Its good safety profile, the plant also exhibits antibacterial, neuroprotective, antiplatelet, antihypertensive, antioxidant, and antidiarrheal potential. Even so, most pharmacological research has focused on other plant components, underscoring the need for mechanistic and clinical studies tailored to the flowers.
According to the International Association for the Study of Pain, pain is referred as "an unpleasant sensory and emotional experience associated with actual or potential tissue damage" [1]. Harmful stimuli cause distress, which is channelled to the central nervous system via specialised neural networks. Peripheral nociceptive and central systems in the central nervous system and spinal cord are involved in the perception of pain. The body uses pain as a shielding against potential harm [2]. Globally, pain is becoming an increasingly significant concern. Accordance with survey, 10% of adults worldwide are newly diagnosed with chronic pain each year, but 20% of adults experience pain [3,4]. Over 80% of people visit doctors due to pain, and for many of these independents, pain is mutable and easily forgotten. Unfortunately, for some individuals, pain persists, becoming a constant encumbrance and source of intermittent anguish [5]. Although pain may be more precisely described, it is nearly always linked to medical concern. It is frequently caused by unpleasant stimuli, which can originate from both internal and external origin [6]. Pain-relieving, both central and peripheral, alleviate without impairing awareness. Opioids and non-steroidal anti-inflammatory drugs (NSAIDs) are the two categories of analgesics used. While NSAIDs are recommended for pain related to inflammation and tissue damage, opioids are recommended for deep-seated visceral pain [7]. Opioids and NSAIDs remain the cornerstones of pain management. However, both medication classes are well known for their frequent and serious adverse effects [8]. According to studies, non-steroidal anti-inflammatory drugs typically result in gastrointestinal (GIT) problems, but opiates produce physical dependency, tolerance, and addiction [8, 9]. Therefore, finding alternative ways to alleviate pain is essential [10].
Herbal medicines may be an intriguing choice for the treatment of opioid abuse and withdrawal [11]. Many diseases have been successfully treated with synthetic drugs for a long time [12]. Since ancient times, traditional plant-derived substances have been used as medicines and are crucial to healthcare, particularly in rural areas where access to modern medication is limited [13–16]. It has been demonstrated that phytochemicals substances, found in plants serve as prevent mechanisms against a variety of illnesses [12]. Several studies have documented the effectiveness of medicinal plants in treating various illnesses [17–20].
Plant Profile:
Acacia nilotica, also called Vachellia nilotica, it’s a flowering tree in the Fabaceae family. It is also referred to by the colloquial names Arabic gum tree [21], Babul [22], Egyptian acacia, and Thorny acacia [23]. It is native to the South Asia, the Middle East, and Africa. For hundreds of years, it has been utilised for both prevention and therapy in Unani and other Indian medical systems. It is the second-largest species in the Fabaceae family. Because of the shape of its fruit pods, the genus name "Acacia" was derived from the Greek word "akis," which means a point or a barb. Many parts of the plant, such as the bark, leaves, pods, seeds, and flowers, have been used for ages because of their medicinal and curatives. The pharmacological relevance of A. nilotica is attributed to the abundance of secondary metabolites in its flowers, including alkaloids, flavonoids, tannins, saponins, and glycosides [24]. By interacting with opioid receptors and inhibiting prostaglandin synthesis, these phytochemicals are believed to aid in the regulation of pain [25, 26]. Numerous biological properties, such as antioxidant, antibacterial, analgesic, anti-inflammatory, and anticancer activities, have been reported for the essential oils of these species [27, 28].

Figure 1: Flower of Acacia nilotica
Taxonomy & Classification [29]
Table 1: Taxonomy & Classification of Acacia nilotica.
|
Taxonomy |
Classification |
|
Kingdom |
Plantae |
|
Division |
Magnoliophyta |
|
Class |
Magnoliopsida |
|
Order |
Fabales |
|
Family |
Fabaceae |
|
Genus |
Acacia (Vachellia) |
|
Species |
Nilotica |
Common Names of Acacia nilotica:
Table 2: Common Names of A. nilotica
|
Language |
Names |
|
English |
Egyptian mimosa, Thorn Acacia, Gum Arabic |
|
Hindi |
Babul, Kikar |
|
Sanskrit |
Babhula |
|
Kannada |
Gobbli, Karijali |
|
Gujarati |
Babaria, baval, Kaloabaval |
|
Malyalam |
Karivelan, Karuvelum |
|
Bengali |
Babla, Babul |
|
Punjabi |
Sak |
|
Telugu |
Nallatumma, Tuma |
|
Tamil |
Kaluvelamaram, Karuvelam |
|
Orisa |
Bambuda, Baubra |
|
Marathi |
Babhul, Vedibabul |
The flowers of A. nilotica are described botanically as follows:
The blossoms of Acacia nilotica, often called Babul or Kikar, are easily identifiable because of their unique morphological characteristics.
One of the most distinctive characteristics of A. nilotica is its golden puffball-like blossoms, which add to the plant's aesthetic and ecological importance. V. nilotica is a single-stemmed plant that can reach heights of 15–18 meters and diameters of 2–3 meters [30–31]. The leaves are 1.3–3.8 cm long, bipinnate, pinnate in 3–10 pairs and have 10–20 pairs of leaflets that are 2–5 mm long [31]. Typically, stems have a deep longitudinal fissure, a green-pinkish slash, a dark to black colour, and reddish gum. Younger trees have orange or green bark, whereas older trees have rough, dark bark and often lose their thorns [30–31]. In older areas, the root was brown, whereas in younger areas, it was pale. Pods are 7–15 cm long, indehiscent, green when immature or greenish-black when mature, and severely constricted between the seeds, giving the impression of a necklace. Each pod contains eight to twelve compressed ovoid seeds [30]. In young trees, thorns are thin, straight, light grey, and 5–7.5 cm in length. They are found in axillary pairs, often 3–12 [31]. The colour of the gum can range from extremely pale yellowish-brown to dark reddish-brown, depending on the tannin content in the sample [32].
Table 3: Phytochemical Composition and biological activity with their structure [33-34]:
|
Phytochemicals |
Structure |
Biological Activity |
|
Phenolic Compounds: Gallic acid, Methyl gallate, Ethyl gallate |
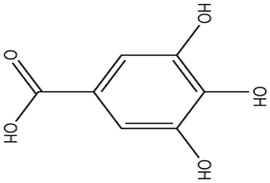
|
Antioxidant activity, anti-inflammatory activity, anticancer activity, and antimicrobial properties |
|
Catechins (Catechin, Gallocatechin, Catechin-7-O-gallate) |
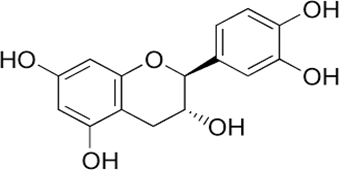
|
Antioxidant, Anti-diabetic, and Cardioprotective effects |
|
Flavonoids: Quercetin, Kaempferol, Iso-quercetin, Naringenin |
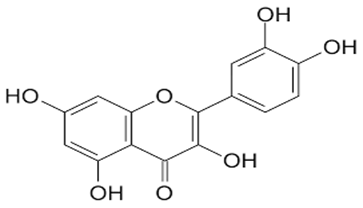
|
Anti-inflammatory, anti-mutagenic, anti-obesity, and chemopreventive |
|
Tannins: Tannic acid, Polygalloyltannins |
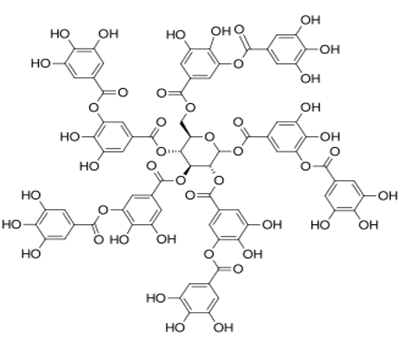
|
Astringent Properties and Antimicrobial Activity |
|
Terpenoids: Lupeol, Lupenone, Niloticane |
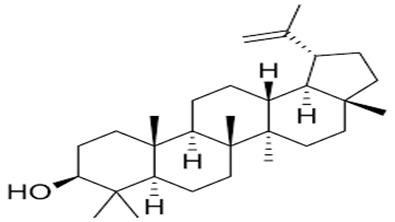
|
Anti-inflammatory, Analgesic, Anticancer, Anti-arthritic, and hepatoprotective |
|
Alkaloids: Dimethyltryptamine, N-methyltryptamine, Tryptamine |
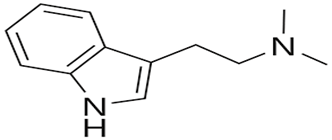
|
Mild analgesic activity and CNS modulation (analgesic) |
Pharmacological Activity:
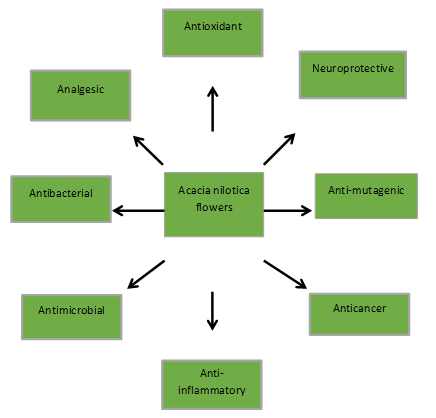
Neuroprotective Activity:
Niloticane exhibits in-vitro cholinesterase inhibitory effect with an IC50 value of 4 µM, whereas galantamine, a positive (+ve) control, has a value of 2.0 µm. AChEIs (Acetylcholinesterase inhibition) improves neuronal transmission and may be useful in treating neurocognitive conditions, such as Alzheimer's disease. Additionally, Vachellia nilotica pod polyphenolics exhibit antioxidant activity and acetylcholinesterase inhibition against arsenic-induced neurotoxicity in rats. In addition to its documented anti-inflammatory and antioxidant properties, the authors postulated that Vachellia nilotica may have potential actions in the treatment of Alzheimer's disease symptoms that are connected to its cholinergic route [35].
Analgesic and Antipyretic Activity:
The hot plate test on albino Swiss mice was used to investigate the analgesic effects of Vachellia pod aqueous extract (500 mg/kg) in comparison to aspirin (100 mg/kg) as a positive (+ve) control. The results showed a significant increase in reaction time compared to aspirin, with the maximum action occurring 90 minutes after administration. Additionally, although it was less effective than aspirin, it had antipyretic efficacy as measured by yeast-induced pyrexia in Albino Wistar rats. Swiss albino mice were used in the formalin-induced writhing assay to investigate the acute and chronic analgesic effects of Vachellia bark aqueous extract. Although no mechanistic studies have been reported, the observations showed both direct analgesic actions on nociceptor blocking and suppression of the synthesis and release of inflammatory pain mediators [36].
Antiplatelet Activity:
The in-vitro human platelet aggregation induced by the platelet-activating agents’ collagen, ARA (arachidonic acid), and adenosine diphosphate was suppressed by the methanolic extract of Vachellia (Acacia) fruits. It was proposed that the protein kinase C effect and Ca2+ channel blockage were responsible for this effect. Furthermore, it inhibited platelet aggregation mediated by the calcium ionophore A-23187, indicating the potential for this action through Ca2+ influx blocking and providing an explanation for its antihypertensive effect. In-vivo, STZ-induced diabetic rats treated with 50 mg/Kg methanolic leaf extract for three weeks showed a significant decrease in platelet aggregation (4.35%) compared to normoglycemic and untreated diabetic rats (2.11–8.76%) [37].
Antimicrobial Activity:
A disc diffusion assay was used to examine antibacterial activity. In which nutrient agar plates were streaked with reference microorganisms from the stock, and the inoculated plates were incubated overnight at 37°C. A small amount of the subculture was placed into a test tube with nutrient broth using a sterile loop and cultured for two–four hours at 37°C until the growth reached the log phase. Petri dishes were filled with nutrient agar medium seeded with a standard inoculum suspension and allowed to settle. To achieve the required concentrations in an aseptic setting, a measured quantity of each test sample was dissolved in a predetermined volume of solvent (methanol). Under a laminar hood, sterile metric filter paper discs were placed in a Petri dish. The discs were then dried after being dipped in the test sample solutions. Using sterile forceps, discs impregnated with the extract, standard antibiotic discs (ciprofloxacin 30 μg/disc), and blank discs (solvent methanol) were placed on the Petri plates and gently pressed to contact the infected agar surface. The zone of inhibition was measured in millimetres (mm) after the inoculated plates were incubated for 24 h at 37 °C [38].
Antifungal activity and Antiviral activity:
The highly active nature of the Acacia (Vachellia) nilotica species makes them an attractive source of antibacterial medicines. Methanolic and aqueous extracts of A. nilotica demonstrated antifungal activity, with percentage (%) inhibition ranging from 34.27±1.45 to 93.35±1.99. Acacia (Vachellia) nilotica dried fruits are used to treat oral candidiasis because they are effective against Candida albicans. Newcastle disease and fowl pox viruses are two animal viruses that can be effectively combated by the methanolic extract of plants [39].
Anti-mutagenic activity:
The plant's acetone extract contains gallic acid and polyphenols, which are what give it its antimutagenic properties. Vachellia nilotica acetone extract showed antimutagenic efficacy against both the S9-dependent mutagen 2-aminofluorene (2-AF) and direct-acting mutagens (NPD, sodium azide). Using various strains of Salmonella typhimurium, the activity was calculated using the plate incorporation Ames Salmonella histidine reversion assay [40].
Antihypertensive activity & Vasoconstriction activity:
Vachellia (Acacia) nilotica pod methanol extract lowers arterial blood pressure at doses of 3–30 mg/kg. In guinea pig paired atria, it has an inhibitory effect on contraction force and rate. Vachellia (Acacia) nilotica aqueous extract causes vasoconstriction in the ileum of guinea pigs by increasing calcium ion inflow. The extract exhibits prolonged contractile action that is dose-dependent [41].
Antibacterial Activity:
Methanolic extracts of Vachellia (Acacia) nilotica's Staphylococcus demonstrated the strongest antibacterial potential against Bacillus subtilis and aureus with an inhibition zone of 15±0.66 mm (millimetres), while leaf extract demonstrated the strongest activity against Bacillus subtilis with an inhibition zone of 20±1.20 mm. Vachellia (Acacia) nilotica showed the strongest efficacy against two fungal strains (Candida albicans and Aspergillus niger) and three bacterial strains (Escherichia coli, Staphylococcus aureus, and Salmonella typhi) in comparative antimicrobial tests of Vachellia species [39].
Anti-Diarrhoea Activity:
Human cough and diarrhoea have been effectively treated with Acacia nilotica. Acute diarrhoea is treated with powdered bark of the plant and a small amount of salt. Using the agar-well diffusion assay technique, methanol and chloroform extracts showed action against Escherichia coli with a Minimum Inhibitory Concentration of 50µg/ml. A. nilotica's ethyl acetate fraction has been shown to have antidiarrheal properties in a castor oil-induced animal. It lowers the intestinal transit of charcoal and the quantity of unformed faeces [42].
Antioxidant Activity:
The decrease in free radical DPPH (2,2-diphenyl-1-picrylhydrazyl) assay is the foundation of this technique. This results in decolourisation. At a concentration of 60 micrometer (µm), the antioxidant caused the methanol to lose its colour. The solution instantly obtained an absorbance value of approximately 0.7 at 517 nm at this concentration. When 20 microlitre (µL) of the sample and 980 microlitre (µL) of 2,2-diphenyl-1-picrylhydrazyl assay (DPPH) (60 µm) were combined, a reaction occurred. The duration required to reach a steady reading of the reaction with the sample was determined to be 2 hours at room temperature. The dose-response curve of this substance was used to express the results in millimolar Trolox [43].
Spasmogenic activity & Antispasmodial activity:
The isolated guinea pig ileum exhibited spasmogenic action when exposed to an aqueous extract of Vachellia nilotica seeds. Muscle spasms may be caused by an increase in calcium influx. The spontaneous contraction of rabbit jejunum was decreased by Vachellia nilotica (methanolic extract) in a dose-dependent manner (0.1–3.0 mg/mL). Additionally, it prevents K+ induced contraction. The underlying mechanism is calcium channel blockage, which lowers blood pressure [41].
Anthelmintic, Analgesic & Anti-inflammatory activity:
According to the adult motility, egg hatch, and larval development assays, the in-vitro methanolic extract of Acacia (Vachellia) nilotica fruit has anthelmintic action against Haemonchus contortus at LC50 = 512.86 and 194.98 microgram per milliliter (μg/ml) concentrations. Rats with acetic acid-induced pain were used to test the analgesic efficacy of A. (Vachellia) nilotica. The plant exhibited strong activity. At the two plant extract dosages (150 and 300 mg/kg bw), large percentages of analgesia were observed. Rats with egg albumin-induced paw oedema were used to assess the anti-inflammatory properties of A. (Vachellia) nilotica extract. This indicates that there was no suppression of paw oedema and, thus, no anti-inflammatory action. However, paw oedema was not suppressed by the extracts [44].
MECHANISTIC STUDIES:
The flowers of Acacia nilotica resistance to pain may be mediated via interactions with opioid receptors, COX and LOX enzymes, and other pain signalling pathways. This study clarifies the mechanisms of action of these extracts and aids in the development of more specialised pain treatments. To advance the use of these substances from research to practical use, it is important to consider safety and any negative consequences. This will determine whether their offer of greater benefits than harm will result in effective pain management.
TOXICITY:
When Acacia nilotica is used at therapeutic levels, toxicological assessments often show a broad margin of safety. Acute oral toxicity studies of aqueous, methanolic, and ethanolic extracts of various parts of A. nilotica (such as flowers, bark, pods, and leaves) in experimental rodents, up to doses of 2000–5000 mg/kg body weight, have shown no mortality or notable behavioural, neurological, or autonomic changes, indicating a high LD50 value. At routinely used doses, subacute and subchronic toxicity studies showed no discernible changes in body weight, food intake, haematological parameters, or significant biochemical markers of liver and kidney function. However, because of their astringent properties, extracts rich in tannins and polyphenols may induce minor gastrointestinal irritation and decrease feed intake at very high doses or with extended administration. Vital organs, such as the liver, kidney, heart, and spleen, generally exhibit normal architecture in histopathological investigations, which supports the relative safety of A. nilotica. Although more chronic toxicity and clinical safety studies are necessary to validate long-term use in humans, the evidence currently available indicates that Acacia nilotica flower extracts are non-toxic and safe for pharmacological use within the suggested dose limits.
SUMMARY OF CURRENT RESEARCH STATUS:
Although the flowers contain potentially beneficial compounds with general anti-inflammatory and antioxidant properties, most published research on analgesic properties has focused on the plant's more commonly used and commercially relevant seeds, bark, leaves, pods, roots, gums, and fruits. Researchers have suggested that more comprehensive studies are required to fully understand the distinct pharmacological pathways and potential therapeutic applications of floral extracts.
CONCLUSION
In conclusion, the current Acacia nilotica (Babul) blossoms are a promising natural source of analgesic and multi-pharmacological compounds, supported by increasing scientific evidence and widespread ethnomedical use. Flavonoids, phenolic acids, tannins, terpenoids, and alkaloids are among the many bioactive phytoconstituents found in flowers, which together have analgesic, antipyretic, antioxidant, antibacterial, neuroprotective, and anti-inflammatory properties. According to experimental research, both central and peripheral mechanisms, including nociceptor modulation, prostaglandin synthesis inhibition, antioxidant activity, and potential interactions with opioid and calcium-dependent pathways, may influence the analgesic effects. The justification for their medical use is strengthened by toxicological data, which also show a large margin of safety at therapeutic levels. Nevertheless, most research has focused on other plant parts, with few mechanistic and clinical studies exclusive to flower extracts, despite promising preclinical results. To confirm efficacy and safety, future research should focus on standardising flower extracts, isolating active ingredients, conducting in-depth mechanistic studies, and conducting clinical evaluations. All things considered, the flowers of Acacia nilotica have a lot of promise as a safer, plant-based treatment for pain and related conditions, which calls for more research and pharmaceutical development.
REFERENCES


Adarsh Singh, Arshad Ayub, Analgesic Activity of Acacia nilotica (Babul): An Ethnopharmacological Review, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 2445-2455. https://doi.org/10.5281/zenodo.19142933
 10.5281/zenodo.19142933
10.5281/zenodo.19142933