
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacy Practice, Sri Adichunchangiri College of Pharmacy, Mandya 571448
In paediatrics, inflammatory bowel disease (IBD) is a chronic, recurrent illness that has a significant effect on development, growth, and quality of life. A fully human monoclonal antibody that inhibits TNF-?, adalimumab, has been a successful therapy for moderate to severe Crohn's disease in children. This study reviews the pharmacological profile, safety, effectiveness, and clinical results of adalimumab in children. Additionally, growth-related outcomes and adverse event profiles in the paediatric population are discussed, along with a comparison of adalimumab to other biologics.
The incidence of inflammatory bowel disease has been steadily increasing globally. (1) IBD can be patchy and affect any part of the gastrointestinal system, although it primarily affects the terminal part of the ileum or perianal region, resulting in transmural inflammation. Patients' quality of life may be significantly impacted by the strictures, fistulas, and abscesses that can develop as a result of inflammatory bowel disease. Controlling symptoms and lowering the risk of serious IBD consequences requires prompt diagnosis and the best possible care. IBD can be patchy and affect any part of the gastrointestinal system, although it primarily affects the terminal part of the ileum or perianal region, resulting in transmural inflammation. Patients' quality of life may be significantly impacted by the strictures, fistulas, and abscesses that can develop as a result of inflammatory bowel disease. Controlling symptoms and lowering the risk of serious IBD consequences requires prompt diagnosis and the best possible care. (1) IBD represents two types of chronic, relapsing inflammatory bowel diseases, Ulcerative colitis and Crohn's disease. (3) Compared to UC, CD is more commonly associated with strictures, fistulas, and abscesses, while colon cancer is commonly associated with inflammation of the mucosa. (1) Children with inflammatory bowel disease (IBD) experienced lower bone mineral density (BMD) than children in good overall health (5, 2). Decreased height velocity was noted in up to 88% of a group of 50 children at the time of diagnosis with CD—a number of these children had impaired linear growth prior to the onset of gut symptoms (20). Growth retardation in childhood and adolescence impacts 13% to 58% of individuals with inflammatory bowel disease, while higher prevalence rates may have been seen (3). ADL has been in use for more than nine years, having received its initial license for the paediatrics CD in 2012(8).
Inflammatory circumstances related to the gastrointestinal tract tend to worsen over time. A teenager's development may be severely affected by this chronic condition's numerous implications, which could involve delays in puberty and nutritional inadequacies. To lessen the effects and promote healthy growth and development during these critical years, early detection and intervention are essential. The number of children and adolescents diagnosed with inflammatory bowel disease (IBD) is predicted to continue to rise globally, with 15% to 25% of IBD diagnoses occurring in children and adolescents (4) .
In 2012, the completely monoclonal antibody ADL was licensed for the treatment of Paediatric CD. Since then, it has been in use for over 9 years, during which time a significant quantity of empirical data has been gathered. The medication is authorized for use in treating moderately to severely active Crohn's disease (CD) in children aged 6 to 17 years in the United States and Europe, as well as highly active CD in teenagers weighing 40 kg or more in Canada (4,21).
Adalimumab is currently being studied in a 52-week, multicentre, randomized, double-blind phase 3 trial. Participants on adalimumab experienced clinical remission rates that were statistically significant when compared to placebo in the trial. These findings offer confirmation in Favor of adalimumab as a possible therapeutic option for children with Crohn's disease. Adalimumab was demonstrated to be an effective medication in the induction and maintenance of clinical remission and response in the paediatric population, which is comparable to the safety record in adult Crohn's studies (4). was first administered to a study patient in 1997(17) and ADL had first been approved for paediatric CD in 2012(18)
They involve the complete and long-lasting healing of the intestinal mucosa, quick and long-lasting clinical remission, decreased steroid use, fewer hospital stays and surgeries, prevention of disease complications (such as fistulas and stenoses), and an improvement in the patient's general quality of life (6). Children diagnosed between 2016 and 2020 had a higher prevalence of positive family history compared to those diagnosed between 2010 and 2015 (7). A study on adults showed that the remission rate of ADL and IMM combination therapy was slightly higher than monotherapy immediately after introductory treatment, but the remission rate or dose escalation at 1 year was similar in both groups (22). The TNF molecules are homotrimers that typically exist on cell surfaces as membrane TNF (mTNF) or as free molecules in solution (soluble TNF) when the TNF-α-converting enzyme (TACE) cleaves mTNF. Reports also suggest the potential for inhibition of mTNF or soluble TNF binding to TNF-R (8).
The incidence of IBD varies depending on the country's geographical location and level of industrial development. They occur primarily in Europe, especially in Scandinavia and the UK, North America, China, South Korea, Japan, India, and Australia. (23)
PHARMACOLOGY OF ADALIMUMAB IN CHILDREN
Numerous mucosal cells, primarily T cells and macrophages, generate TNF from the plasma membrane as a precursor. The converting enzyme breaks down TNF into its soluble form (sTNF) from its transmembrane precursor form (mTNF). The TNF receptor exists in two variants, 55-kDa TNFR1 and 75-kDa TNFR2, both of which are physiologically active. Although sTNF primarily signals through TNFR1, signal transduction of the membrane-bound version of TNF can be conveyed through both TNFR1 and TNFR2. Cell division, the release of cytokines, or apoptosis are all part of the TNF receptor's downstream signalling cascade. (10)
Figure 1: Chemical Structure of adalimumab
PHARMACOLOGICAL PROFILE OF ADALIMUMAB
|
Chemical Name: |
Adalimumab(D2E7) (12) |
|
Molecular Formula: |
C6428H9912N1694O1987S46 |
|
Molecular Weight: |
148 kilodaltons (12) |
|
Storage temp. |
Store at -80°C |
|
Volume of distribution |
5-6 Litres
|
|
Biological half-life |
14 days |
|
bioavailability |
A single subcutaneous injection is 64% |
Anti-TNF drugs decrease inflammation by effectively neutralizing the proinflammatory cytokine TNF, rendering it ineffective. Many studies demonstrate that anti-TNF autoantibodies perform a greater complexity of roles than basic inhibition due to the complex nature of TNF signalling. (11)
MECHANISM OF ACTION OF ADALIMUMAB
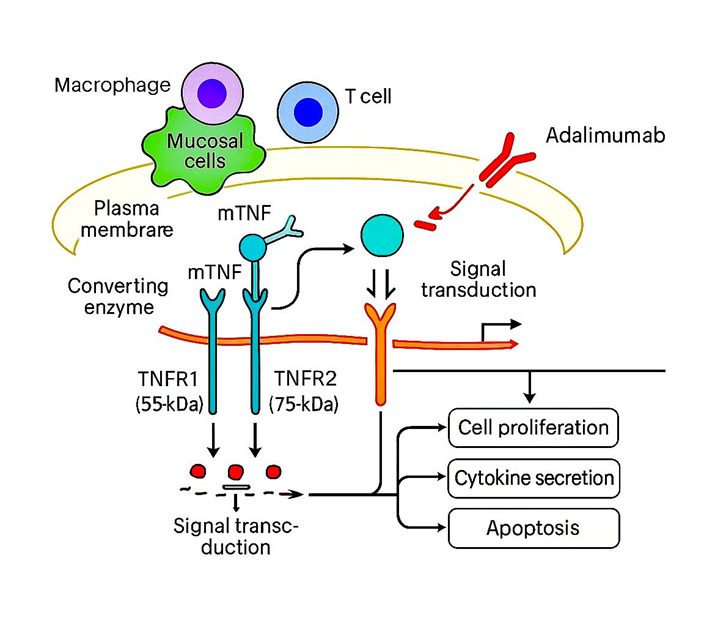
Figure 2: Mechanism action of adalimumab in IBD
Antigen-presenting cells, including activated macrophages, dendritic cells, and effector T cells, produce the pro-inflammatory cytokine TNF α. [4] TNF-α initiates and drives inflammatory cascades that contribute to the onset and progression of autoimmune disorders. It regulates various cellular and molecular signals, leading to both local and systemic effects. TNF-α activates T cells, promotes immune cell infiltration, stimulates angiogenesis, and increases skin cell proliferation, all of which intensify inflammation. Adalimumab binds directly to TNF-α and blocks it from interacting with its receptors, thereby reducing inflammatory signalling and helping control autoimmune disease activity.[12]
DOSAGE
ADL is a monoclonal anti-TNF antibody derived entirely from humans. Children who weigh less than or more than 40 kg are often administered a subcutaneous injection of 20 or 40 mg every two weeks after the induction phase. ADL can be used to treat moderately to severely active CD in children (ages 6 and up) who do not respond well to or are intolerant of traditional treatments like immunomodulators, corticosteroids, and primary nutritional therapy, or who are not candidates for these treatments. [9,18]
Adalimumab is injected subcutaneously at a dose of 160 mg at the start of treatment and 80 mg two weeks later. Adalimumab is subsequently given at a dose of 40 mg every two weeks to maintain remission.[11]
SAFETY AND EFFICACY
In paediatric CD, biological treatments, such as anti-TNF-α drugs, have been widely employed since they have been shown to improve the natural course of IBD and promote mucosal repair. Numerous studies have also shown the effectiveness of ADA in helping children with CD achieve clinical remission. ADA helps children with CD maintain a long-term response and remission.[8]
Adalimumab can be used alone or in combination with MTX or other DMARDs, of adalimumab has been extensively studied in randomised controlled trials (RCTs). [14,15] A 2012 study led to the approval of ADL in children and represented the first prospective RCT in the management of paediatric ADL. [19]
Adalimumab helped 33.5% of children with medium to severe Crohn's disease in this research achieve remission at 26 weeks. Infliximab-naïve patients significantly surpassed the high-dose group. Of those in remission by week 52 of treatment, 28.4% reported so. Reduced inflammation, improved growth, and steroid-sparing effects were seen in both dosing groups. Adalimumab was well-received and was not associated with any new safety issues.[19]
Comparison With Other Biologics
The first TNF-α inhibitor that was approved for the treatment of CD in adults and children was infliximab (IFX), which is a monoclonal IgG1 antibody with a partially murine anti-TNF Fab region. However, recent data indicate that approximately 30% of patients lose response within 3 years after starting the treatment, while half withdraw from the treatment due to a lack of the drug. In randomized clinical trials, adalimumab (ADA), an IgG1 antibody containing a humanized Fab region, has shown effectiveness in the treatment of paediatric patients with CD and has been approved by the United States Food and Drug Administration for treating moderate and severe CD in children. It is also recommended by the European consensus guidelines for the treatment of long-term active intestinal diseases in children.[19]
|
Adalimumab |
Etanercept |
Infliximab |
|
Fully human IgG1 monoclonal antibody against TNF?α |
Fusion protein of soluble TNF receptor p75 and Fc fragment of human IgG |
Chimeric human?mouse anti-TNF alpha antagonist |
|
Self-injectable subcutaneously or with the help of parents |
Self-injectable subcutaneously or with the help of parents |
Intravenous injection, inconvenient as it requires more clinic visits |
|
Dosing: 80 mg subcutaneous, then 40 mg after one week, followed by 40 mg every other week |
25?50 mg subcutaneous twice weekly for 3 months, then 50 mg once weekly |
5 mg/kg intravenous at 0, 2, and 6 weeks, then every 8 weeks |
|
Must be refrigerated (2?8°C) and protected from sunlight |
Must be refrigerated (2?8°C) and protected from sunlight |
Must be refrigerated (2?8°C) and protected from sunlight |
|
Relatively early onset as compared to etanercept |
Slower onset and moderate efficacy as compared to infliximab, fewer chances of developing |
Highly effective with early onset |
|
Fewer chances of developing antibodies |
Lower chances of developing antibodies in comparison to infliximab |
Increased chances of developing antibodies against infliximab as compared to adalimumab or etanercept |
ADVERSE EFFECTS OF ADALIMUMAB
Various clinical trials have shown that adalimumab is usually well?tolerated. The adverse events noted were mild and did not require discontinuation of the therapy. Development of serious adverse reactions was rare.[12]
Local injection site reactions: The most common adverse events after adalimumab injections were injection site reactions. They are usually mild and transient, being characterized by pruritus, pain, erythema, and urticarial plaques. Hypersensitivity reactions, Anaphylaxis, and angioedema have been reported following treatment with adalimumab.
Cutaneous adverse events: Skin infections, allergic rash, urticaria, drug eruptions, eczematous and psoriasiform eruptions, vasculitis, ulcers, actinic keratoses, tumours, treatment-related plaque psoriasis, and pustulosis have all been described.[13]
Recurrent episodes of amaurosis fugax, psoriasis, tremor, depressive syndrome, and mild reactions at the injection site.[16]
CONTRAINDICATION
It is important to highlight that adalimumab has not been well examined in individuals who are 4 years of age or younger or who weigh less than 15 kg, even though the manufacturers have not listed any contraindications. Clinicians are advised to avoid administering adalimumab to this specific age group as a precaution. [3,8] In patients with underlying infection foci, congestive heart failure, or hepatic dysfunction, as well as in cases of hypersensitivity, clinicians should refrain from giving adalimumab. Adalimumab has been linked to hepatitis B reactivation, especially in inactive HBsAg carriers who have low amounts of hepatitis B virus (HBV) DNA and normal serum aminotransferase levels. Adalimumab's immunomodulatory action raises serum HBV DNA levels and accelerates HBV replication.[25]
Patients with comorbidities, those using concurrent immunosuppressants such as corticosteroids and methotrexate, and those 65 years of age or older are at a higher risk of getting serious infections, which can result in hospitalization and/or death. It is advised that a patient stop taking adalimumab if they get sepsis or an infection that is severe.[24]
GROWTH AND DEVELOPMENT OUTCOMES
Adalimumab normalized height velocity in children who had growth impairment at baseline (height velocity z?score improved from?−2.8 to?0 by week?192), indicating sustained growth catch?up during long?term therapy (3). Health?related quality of life also improved significantly and was maintained through 240?weeks.[3]
Among 62 children (mean age?13?years), 81% achieved clinical remission at?12?weeks and 95% at?52?weeks. Height, growth velocity, and BMI z?scores increased significantly over 52?weeks, especially in those with baseline growth failure.[16]
CONCLUSION
The management of moderate to severe paediatric Crohn's disease has improved significantly with the introduction of adalimumab. It is a fully human monoclonal anti-TNF-α antibody with a favourable safety and efficacy profile. It can induce and sustain clinical remission, encourage mucosal healing, and enhance quality of life about health. In addition to improving patient compliance through subcutaneous injection, its decreased immunogenicity compared to other biologics offers a benefit for prolonged therapy. The benefits of adalimumab for encouraging healthy growth and development have been important, particularly for kids who exhibit growth failure. Although being typically well tolerated, careful patient selection and close observation are nevertheless essential to reduce possible hazards, especially those related to infections and hypersensitivity responses.
REFERENCES


Sanketh Gowda Hannasoge Manja, Sushmitha S M, Adalimumab in Pediatric Inflammatory Bowel Disease: A Comprehensive Review of Efficacy, Safety, and Clinical Outcomes, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 7, 2831-2838. https://doi.org/10.5281/zenodo.16268281
 10.5281/zenodo.16268281
10.5281/zenodo.16268281