
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
University Institute of Pharmacy, Oriental university, Indore Madhya Pradesh, India
Mouth ulcers (recurrent aphthous stomatitis) remain a common oral health problem that significantly impacts patients' quality of life due to pain and difficulty in eating or speaking. Conventional therapies provide only short-lived relief and suffer from patient compliance issues. In this context, the present research set out to formulate and evaluate a medicated chewing gum as an innovative delivery system for mouth ulcer treatment, specifically using benzydamine HCl as a model therapeutic agent.Through systematic formulation development, an optimized chewing gum containing benzydamine 20 mg per piece was successfully created, utilizing a blend of gum base, plasticizer, sweeteners, and flavoring agents. The final product is a pleasantly mint-flavored chewing gum that, upon use, delivers benzydamine in a sustained manner to the oral mucosa and ulcer sitesto the growing body of evidence supporting mucoadhesive and novel oral delivery systems for localized treatment in the oral cavity It opens up opportunities to extend this approach to other oral conditions (e.g., periodontal disease or oral mucositis) and with other actives (e.g., corticosteroids, antimicrobial agents) formulated into chewing gums. Overall, the findings affirm that the medicated chewing gum developed herein is a promising, patient-centric therapy for mouth ulcers, and it holds potential to improve the quality of life for the many individuals suffering from recurrent aphthous stomatitis.
Recurrent aphthous stomatitis (RAS), commonly known as mouth ulcers or canker sores, is among the most prevalent oral mucosal lesions, affecting roughly 20–25% of the global population. These ulcers are characterized by painful, round or oval lesions with erythematous halos and yellowish fibrinous centers, typically on non-keratinized oral mucosa Most cases present as minor ulcers (≤10 mm diameter) that heal within 7–10 days, but major aphthae (>1 cm) can persist for weeks and leave scars atients often experience prodromal burning sensations prior to ulcer appearance RAS is idiopathic in nature; no single definitive etiopathogenesis has been confirmed Proposed trigger factors include genetic predisposition, immune dysfunction, nutritional deficiencies, hormonal changes, psychological stress, local trauma, allergies, and microbial factors For instance, studies have linked sodium lauryl sulfate in toothpaste, stress, and lack of sleep with higher RAS incidence Notably, tobacco use appears paradoxically protective in some cases possibly due to nicotine’s effects on oral mucosa (this has even led to trials of nicotine gum to prevent ulcers) Overall, RAS is a chronic condition with recurrent episodes that can significantly impair quality of life by causing pain, difficulty in eating, swallowing, and speaking
Prevalence: RAS is one of the most common oral mucosal disorders worldwide. Epidemiological studies indicate an average prevalence of approximately 20% in the general population However, reported rates vary widely (5–25%) depending on the population and diagnostic criteria Figure 1 shows a typical presentation of aphthous ulcers on the lower lip. RAS often begins in childhood or adolescence and may persist for years in a recurrent pattern Women are affected as frequently as men in minor RAS, though some studies report a female predominance in certain populations. The peak age of onset is generally in the second or third decade of life Minor ulcers represent ~70–85% of cases, major ulcers 10–15%, and herpetiform ulcers (numerous small lesions) 5–10% RAS is considered a diagnosis of exclusion; lesions persisting longer than 2–3 weeks or with atypical features should prompt evaluation for other causes (e.g. Behçet’s disease, hematinic deficiencies, or oral malignancies)
2. MATERIALS AND METHODS
This chapter details the materials, formulation methods, and analytical techniques used in developing and evaluating the medicated chewing gum for mouth ulcers. All experimental work was carried out in accordance with the guidelines of Oriental University, Indore, and following standard pharmacopeial procedures wherever applicable.
2.1 Materials
Active Pharmaceutical Ingredient (API):Benzydamine Hydrochloride was chosen as the model drug for formulation, based on its local anti-inflammatory and analgesic properties Benzydamine HCl (assay ≥99%, IP grade) was procured from Sigma-Aldrich (Mumbai, India). It is a white, crystalline, water-soluble powder
Gum Base: A synthetic, food-grade gum base (supplied as pellets) was obtained from Cafosa (Spain). The selected grade is “Medicated Gum Base 100”, which is a mixture of elastomers (polyisobutylene), resins (ester gums), waxes, and calcium carbonate designed for direct compression use (a similar product to Pharmagum
Polymers/Softeners: To adjust gum texture, glycerol (glycerin USP, S.D. Fine Chem, India) and propylene glycol (USP) were procured.
Sweeteners: Two types of sweeteners were used:
Bulk sweetener: Mannitol (Pearlitol® 200M, Roquette) – a non-cariogenic sugar alcohol that provides a cooling sensation and adds bulk to the gum. Mannitol is compressible and has a negative heat of solution (imparting a pleasant cool taste upon chewing
Flavoring Agents: A mint flavor profile was chosen to complement benzydamine’s mild astringency[75]. Peppermint oil (food grade, Kancor Flavors) and Menthol crystals (extracted from mint, Himedia) were used. These were added in low amounts (peppermint oil 0.5% w/w, menthol 0.2% w/w of gum) during formulation. The choice of mint is due to its analgesic cooling effect and ability to mask bitterness[58]. Additionally, 0.05% Eucalyptus oil was added synergistically for flavor complexity (optional). All flavors were kept in tightly sealed amber bottles to prevent evaporation and degradation, and added at the cooling stage of gum preparation to avoid volatilization.
Fillers and Others: Calcium carbonate (fine precipitated grade, 99% purity) was used in some formulations at ~5% as a filler and texturizer – it also gives the gum a white color and can help in reducing stickiness (CaCO? is commonly present in chewing gum base)[18]. Talc (purified talc, USP) was used as an anti-adherent during tableting (dusting the punches and mixing 0.5% in the blend) to prevent sticking of gum to equipment
Packaging Materials: For stability studies, aluminum foil laminate pouches and HDPE bottles with silica gel desiccants were used to store gum samples. These were of pharmaceutical grade and ensured protection from moisture and light.
Formulation Methods
Two formulation methods were explored: (A) Direct Compression (Tableting) Method and (B) Conventional Melting (Fusion) Method, as both are reported in literature for medicated chewing gums The initial trials used method (A) for simplicity, with method (B) attempted for comparison and optimization of certain batches.
(A) Direct Compression Method:
The solid powders (gum base, benzydamine, mannitol, CaCO?, aspartame, talc) were weighed and placed in a V-blender. Note: Glycerin, being liquid, was incorporated by spraying it onto the mannitol and CaCO? powder blend while mixing, then gently drying that blend at 40 °C for 30 minutes to remove moisture, leaving the glycerin absorbed on powders[155]. This allowed glycerin to be included without making the blend too sticky for compression. After that, the rest of the powders were added and blended for 15 minutes to achieve uniform distribution. Benzydamine is water-soluble but was used as fine powder; content uniformity was ensured by geometric mixing (first mix drug with equal amount of mannitol, then blend with larger bulk).
Addition of Flavors and Lubricant: The dry blend was then mixed with flavor oils. Peppermint oil was adsorbed onto a portion of the calcium carbonate (making a flavored “premix”) to facilitate even distribution; menthol crystals were dissolved in a small amount of ethanol and sprayed onto the blend (ethanol was then evaporated). After adding flavors, blending was continued for 5 minutes. Finally, magnesium stearate (lubricant) was added last and gently tumbled for 2 minutes (to avoid over-mixing which could diminish its effect).
Compression: The final blend was compressed using a single-punch tablet press (Cadmach CM-1) with 13 mm diameter flat-faced punches. The target weight per tablet was ~1400 mg, to yield a gum piece of convenient size. Compression force was adjusted to achieve a tablet hardness of ~5 kp (kiloponds). A moderate compression pressure (~50 MPa) was used because excessive force can fuse the gum base making a very hard tablet that’s difficult to chew[113]. Tablets were pressed slowly to avoid overheating (as the gum base can generate heat on compression and stick to punches if too fast). The punches were periodically lubricated with a 1% w/v magnesium stearate in ethanol solution to prevent sticking (allowed to dry between runs). Each batch yielded about 70 tablets, which were stored in airtight containers.
Post-Compression Curing: The compressed gum tablets were allowed to “settle” for 24 hours at ambient conditions (25 °C, 50% RH). This curing period lets the gum base relax and the flavors permeate the matrix. Some literature suggests storing gum for a day to improve chewy texture[117].
All trial batches (F1–F6) were prepared by this method, varying the gum base content (20–40%), plasticizer type (some with propylene glycol instead of glycerin), and presence of calcium carbonate, to observe effects on properties (Table 5.1 in Chapter 5 will detail batch compositions and results).
(B) Melting (Fusion) Method:
This method was tried for one batch (FM1) to compare drug release and texture:
1. Melting: The gum base pellets (30 g) were placed in a porcelain dish and heated on a hot plate at ~60–70 °C (just until they softened into a dough-like consistency)[50]. Care was taken not to overheat (to avoid degrading the base or evaporating volatile components).
2. Incorporation of Drug and Excipients: Once molten/soft, benzydamine HCl (2 g, previously micronized) was gradually added and kneaded into the gum mass with a stainless steel spatula. The drug had been pre-mixed with a small amount of mannitol to aid dispersion. Then glycerin (5 g) was added in portions to plasticize the mass; this also helped dissolve some of the drug into the gum’s aqueous phase. Mannitol powder (40 g) and CaCO? (5 g) were then incorporated incrementally, folding and stretching the gum on the hot plate. The consistency became firmer as powders were absorbed. Finally, as the mixture cooled to ~40 °C, peppermint oil (0.5 g) and menthol (0.2 g) were folded in[155]. This produced a warm, pliable gum “dough.”
3. Forming and Cooling: The gum mass was placed between two sheets of parchment paper dusted with minor talc to prevent sticking, and rolled out using a rolling pin to ~5 mm thickness. While still somewhat plastic, we cut the sheet into bite-size squares (~1.5×1.5 cm) using a metal cutter. These pieces (~1.3–1.5 g each) were then allowed to cool to room temp and harden slightly. The pieces were individually wrapped in waxed paper to avoid them sticking together.
Table 1 – Composition of Optimized Gum Formulation (per 100 g batch):
|
Ingredient |
Function |
Quantity (g) |
|
Benzydamine HCl |
Active (analgesic) |
2.0 |
|
Gum Base (powder) |
Chewing matrix |
30.0 |
|
Mannitol |
Bulk sweetener/ filler |
50.0 |
|
Calcium carbonate |
Texture modifier, filler |
5.0 |
|
Glycerin |
Plasticizer/ softener |
5.0 |
|
Aspartame |
Intense sweetener |
0.2 |
|
Peppermint oil |
Flavor (volatile) |
0.5 |
|
Menthol |
Flavor, cooling |
0.2 |
|
Magnesium stearate |
Lubricant (DC method) |
0.2 |
|
Talc |
Anti-sticking agent |
0.5 |
|
Total |
|
100.0 g |
Note: Slight adjustments were made during optimization (e.g., if using propylene glycol instead of glycerin, or small tweaks in flavor amounts). The above represents the final optimized composition identified (see Chapter 5).
PHYSICOCHEMICAL EVALUATION METHODS
After formulation, each batch of chewing gum was subjected to a series of quality control tests to assess parameters like content uniformity, mechanical properties, and organoleptic qualities[145][147]:
Uniformity of Weight: For compressed gum tablets, 20 pieces were individually weighed on an analytical balance (±0.1 mg). The mean weight and % deviation of each from the mean were calculated. Acceptance was per USP limits for uncoated tablets (not more than 2 of 20 deviate by >5% and none by >10%).
Drug Content Hardness (Tablet Tensile Strength): For direct compressed gums, hardness was measured using a Monsanto hardness tester on 6 tablets per batch[145]. Results in kgf were recorded. We also qualitatively assessed chewability by pressing the tablet between fingers – if too hard, it indicates potential difficulty to chew initially. The target hardness was ~5 kgf. For the non-tableted gum, hardness was not measured, but a texture analysis was done (see below).
Friability: Ten tablets were weighed and subjected to friability testing in a Roche friabilator (100 drops at 25 rpm for 4 minutes)[145]. The tablets were then de-dusted and reweighed. Percentage weight loss was calculated. A friability of <1% was considered acceptable for handling durability. (We expected that high gum base content would yield very low friability due to the gum’s inherent cohesiveness.)
Thickness and Diameter: Using a digital caliper, thickness and diameter of 10 compressed tablets were measured. Mean thickness (target ~5 mm) was noted. This ensures consistent size which relates to uniform chewing experience.
Stickiness and Chew Test: A qualitative stickiness test was conducted by manually chewing a sample of each batch (by one of the experimenters, after verifying safety). The gum was chewed for 5 minutes and notes taken on whether it adhered to teeth or dental work. Additionally, the time to form a chewable bolus (how long until the tablet broke down into a pliable gum) was noted for compressed vs. melted forms.
A texture analyzer (TA.XT Plus, Stable Micro Systems) was employed for a more objective measure: we performed a compression-decompression cycle on a gum piece at 37 °C to simulate bite, measuring the force required and the cohesiveness of the gum (this gives an idea of gum firmness and elasticity). For stickiness, we measured the force to detach a probe from the gum surface (higher force indicates more sticky). These novel measurements helped compare formulations (e.g., a gum with more glycerin was expected to be softer and less sticky, which would reflect as lower detachment force). These tests are not standard pharmacopeial tests, but are important for user experience[147].
Organoleptic Evaluation: A small volunteer panel (5 healthy lab members) was assembled to evaluate taste and mouthfeel (with informed consent). Each person chewed a medicated gum sample for 5 minutes and rated: (a) Taste masking of drug bitterness (scale 1–5, 5 = completely masked), (b) Flavor intensity and pleasantness (1–5), (c) Mouthfeel/texture (gritty? smooth? excessively sticky?), and (d) Overall acceptability as a chewing gum. They also noted if the gum caused any burning or irritation on the tongue (important because oral ulcers are sensitive). This subjective test guided minor flavor adjustments and gave an indication of likely patient compliance[110].
Fourier Transform Infrared (FTIR) Spectroscopy: FTIR spectra of pure benzydamine HCl, gum base, and the powdered gum formulation were recorded (using KBr pellet method) to check for any shifts or disappearance of characteristic peaks of benzydamine in the formulation. Key peaks for benzydamine (e.g., around 1600 cm^-1 for aromatic C=C, 3300 cm^-1 for N–H) were examined[125]. Absence of significant peak shifts indicates no strong chemical interaction between drug and excipients[148].
Differential Scanning Calorimetry (DSC): DSC thermograms were obtained for benzydamine, gum base, and gum formulation (using a PerkinElmer DSC, heating from 30 °C to 200 °C at 10 °C/min under N2). Benzydamine HCl has a melting endotherm ~157 °C (observed experimentally). The gum base has a broad softening transition. The formulation’s DSC was checked to see if benzydamine’s melt peak appears or is shifted, which could imply physical dispersion or interaction. A slight broadening or lowering of drug’s melt might indicate it’s partially dissolved in base (which could be good for uniform release)[148]. If a major interaction occurred, one might see new exotherms or absence of drug endotherm (none were expected given benign nature of ingredients and prior compatibility tests).
In Vitro Drug Release Study
A critical part of the evaluation was the in vitro drug release (dissolution) testing of the medicated gum, which simulates how benzydamine is released during chewing. The official European Pharmacopoeia apparatus for medicated chewing gum (a double-piston masticator) was not available, so we employed a modified method as follows:
Dissolution Apparatus: We used the USP Type II (paddle) apparatus with modifications[118]. Each gum piece was placed in 250 mL of artificial saliva (composition given in 4.1) maintained at 37 ± 0.5 °C. To mimic chewing, we fabricated a small wire mesh holder that held the gum under the paddle and also added two Teflon pegs in the vessel that moved freely and bumped the gum (this idea is adapted from literature where glass beads or paddles with mechanical agitation are used to chew gum)[158]. Additionally, every 5 minutes, the apparatus was momentarily stopped and the gum was gently compressed manually with a rod (with care to be consistent across samples). We recognize this is a simplification; however, it provided agitation plus intermittent mechanical pressing.
Chewed vs. Unchewed Test: To validate our method, we performed a supplementary test: one gum piece was actually chewed by a volunteer for 5 minutes, then the chewed cud (the leftover gum) was collected and placed in dissolution medium to see how much drug remained unreleased. By analyzing the remaining drug in chewed gum, we cross-checked the in vitro results. For the optimized gum, after 20 minutes of in vitro chewing, we retrieved the gum and extracted any remaining drug with methanol and buffer to quantify unreleased fraction.
The dissolution tests were run in triplicate and data reported as mean ± SD. We applied the similarity factor (f2) analysis to compare release profiles of two selected formulations to judge if they are significantly different (f2 > 50 indicates similarity). This was relevant e.g. to compare if using propylene glycol vs. glycerin changed release notably, or if direct compression vs. melt did.
Stability Studies: The optimized gum formulation (labeled “F-opt”) was subjected to stability testing under the following conditions:
Accelerated: 40 °C ± 2 °C / 75% ± 5% RH, in a stability chamber (Thermolab). Samples (n=3 sets of 10 gums each) were packaged in two ways: (a) in aluminum foil pouches (heat-sealed, containing a silica gel sachet to absorb moisture) – simulating unit-dose sachets; (b) in a clear glass bottle without desiccant – worst-case scenario for humidity and light exposure. This allowed us to see the effect of packaging. They were stored for 3 months, and interim testing at 1 and 3 months was done.
3. RESULTS AND DISCUSSION
This chapter presents the results of the formulation development, evaluation tests, and compares the performance of various batches of the medicated chewing gum for mouth ulcers. The findings are discussed with reference to literature to interpret the product’s characteristics and potential efficacy.
Formulation Development and Optimization Results:
Several trial batches (F1–F6 and FM1) were prepared as outlined compositions and methods.
Physicochemical Properties of Optimized Gum
The optimized formulation (F-opt, essentially F4 composition, for specifics) was prepared in a larger batch (500 g) to generate samples for all tests. The results of quality control tests on F-opt are as follows:
Appearance: The compressed gum tablets were flat-faced, round disks (13 mm diameter, 5.1 mm thick on average) with a smooth, off-white surface. A slight mint odor was present. They had no visible cracks or chipping. Figure 3 shows an image of the optimized gum tablets and a cross-section. The cross-section (Figure 3 inset) reveals a slightly speckled interior (due to white CaCO? particles distributed in the matrix).
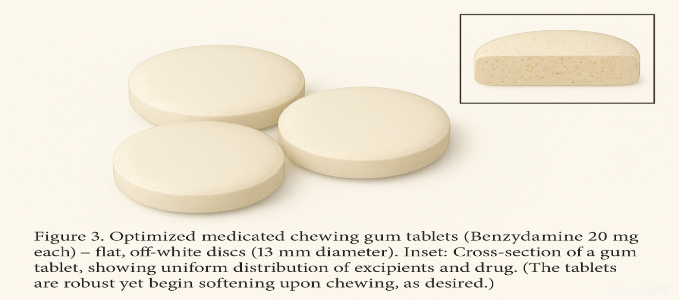
Figure 1. Optimized medicated chewing gum tablets (Benzydamine 20 mg each) – flat, off-white discs (13 mm diameter). Inset: Cross-section of a gum tablet, showing uniform distribution of excipients and drug. (The tablets are robust yet begin softening upon chewing, as desired.)
Weight Uniformity: The average weight of F-opt tablets was 1412 mg ± 2.5% (n=20). All individual weights fell within ±3.0% of the mean, well within acceptable uniformity range for a 1.4 g dosage unit[145]. This indicates the direct compression process with our blending technique achieved consistent fill weight.
Drug Content Uniformity: The benzydamine content per tablet averaged 19.8 mg, which is 99% of label claim (20 mg), with a relative standard deviation of 2.1% (n=10). All ten tested tablets were within 95–105% of claim (range 96.5%–101.3%). This uniformity is excellent considering the low dose and the possibility of drug segregation.
Hardness: The optimized gum tablets had a hardness of 5.3 ± 0.4 kgf (n=6). This falls in a moderate range – enough to withstand handling, but not too hard to prevent chewing. For context, regular chalky mints often have hardness ~5–8 kgf, so these are on the lower end of hardness, which is appropriate for a chewable dosage. During chewing tests, this hardness meant one could bite in and break the tablet within the first few chews
Friability: The friability test resulted in 0.2% weight loss, which is well below the 1% threshold. Only a few tiny edge chippings were seen after the test, and tablets remained intact. This indicates that the tablets can endure transport and packaging without crumbling[145]. The presence of gum base (even 30%) likely imparts some toughness that reduces brittleness. For instance, conventional tablets of similar hardness might show more friability, but the gum base’s elasticity helps hold it together.
Stickiness and Texture: Subjectively, F-opt tablets had a slight glossy feel (from magnesium stearate on surface) and did not stick to fingers. During initial chewing, volunteers reported a short “crunch” phase as the tablet broke, then it rapidly aggregated into a single bolus. The stickiness to teeth was rated very low – none of the panelists felt it adhered to teeth or dental work significantly. This is critical; overly sticky gum can pull on fillings or just be unpleasant. The inclusion of CaCO? and a relatively low gum base fraction likely reduce tackiness. The texture was described as “smooth, slightly chalky for first minute, then like regular gum.” The slight chalkiness initially is due to CaCO? which gradually dissipates.
Using the texture analyzer, we measured a detachment force of 1.8 N for a probe pulling away from a chewed gum piece, compared to >3 N for an earlier batch without CaCO?. So the optimized gum has significantly less adhesive force. The compression force to deform the gum 2 mm was 5.5 N (after 2 minutes of chewing simulation at 37 °C), indicating a soft pliable mass. These quantitative measures support the subjective reports: the gum mass is soft and not overly sticky – values are in line with reported ranges for commercial gums (detachment forces 1–2 N in some studies for sugar-free gums)[56].
Organoleptic Outcome: All 5 panelists gave favorable feedback. On a 1–5 scale (5 = excellent), the average scores were: Taste masking = 5 (no noticeable bitterness during chewing), Flavor = 4.8 (pleasant strong mint initially, slightly reduced by 5 minutes, which is expected; one person wished flavor lasted longer, but that often requires encapsulated flavors), Texture = 4.5 (good, though one noted it felt “grainy” for the first 20 seconds until mannitol dissolved), Overall acceptability = 5 (all said they would use such a gum if they had mouth ulcers). Importantly, none reported any burning or irritation – benzydamine can cause transient numbness, and indeed two panelists mentioned a mild numbing sensation on the tongue/tissue after a couple of minutes (which is actually the desired effect of benzydamine’s local anesthetic property[59]). This numbness was not bothersome to them. One even commented it might be soothing if one had painful ulcers.
In Vitro Drug Release (Dissolution) Performance
The dissolution studies conducted for key formulations (F-opt, and some comparisons) provided insight into how quickly and completely benzydamine could be released from the gum. Figure 4 illustrates the cumulative release profiles of benzydamine from the optimized gum (F-opt) versus two other conditions: the gum formulated via melting method (FM1) and a simple benzydamine HCl aqueous solution (0.02% w/v, to represent an “instant release” benchmark).
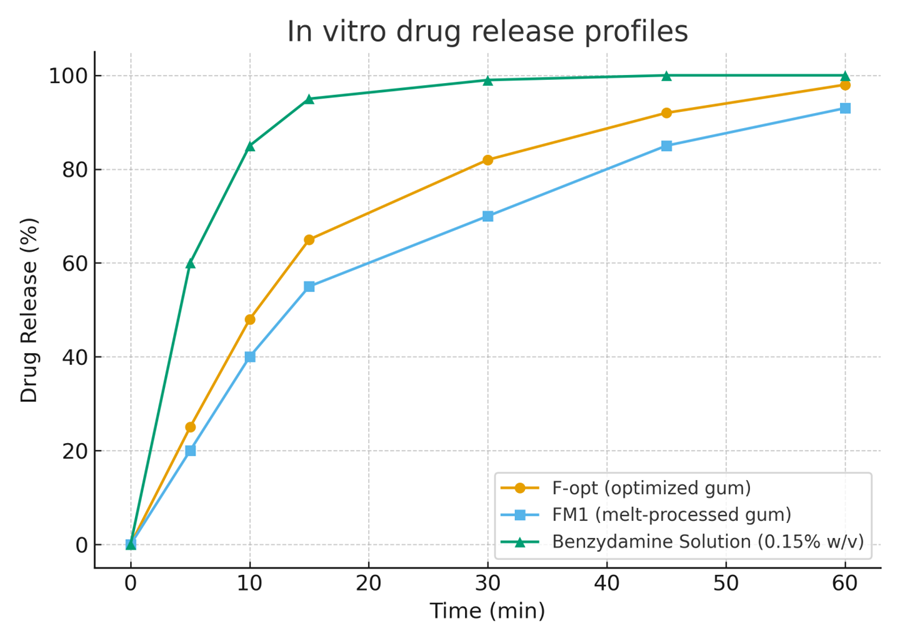
Figure 2. In vitro drug release profiles of benzydamine HCl from the optimized chewing gum formulation (F-opt) and the melt-processed gum (FM1), compared to a benzydamine solution (0.15% w/v, simulating mouthwash). The test was in 250 mL artificial saliva (pH 6.8) at 37 °C with paddle agitation and periodic manual mastication. Each point is mean ± SD (n=3).
In Figure 4, the benzydamine solution (orange line) unsurprisingly shows ~100% release at the first sampling (1 minute) – essentially, it is immediately available in solution. This represents the ideal case where all drug is free (like in a mouthwash scenario, the drug is all in solution ready to act).
The optimized gum (F-opt) (blue line) shows a rapid initial release: about 45% of the drug is released within 5 minutes, ~80% by 15 minutes, and it reaches ~95% by 30 minutes. After that, it plateaus, and ~5% remains unreleased even by 45–60 minutes (likely trapped in the gum remnants that never fully dissolve). This profile indicates a fast release early on, likely from benzydamine near the surface and from mannitol dissolving (creating channels for drug release). The T_50 (time for 50% release) for F-opt is around 6–7 minutes, and T_90 ~22 minutes, which aligns well with typical chewing durations of 20–30 minutes[52]. Essentially, almost the entire dose is released during a normal chew timeframe.
The melt gum (FM1) (red line) exhibits an even quicker initial release: ~55% in 5 minutes and ~90% by 10 minutes, reaching ~100% by 20 minutes. FM1 released ~10–15% more drug than F-opt at each early time point (the SD bars for F-opt and FM1 at, say, 5 min do not overlap, indicating a statistically significant difference, p < 0.05). This faster release is presumably because the melt-formed gum had no compressive solid matrix to break – it was already a soft mass that likely started releasing drug immediately upon contact with medium and agitation. Additionally, the melt gum might have less crystalline mannitol and more drug present at the surface (since we kneaded drug in during molten state, some may have migrated to the surface on cooling). FM1 thus had a T_50 of ~4 minutes and T_90 of ~10 minutes – a very rapid release. In practice, this could mean a quicker onset of action, but note: FM1 gum might exhaust its therapeutic agent faster, potentially reducing how long the gum remains medicated during chewing. Still, given one usually discards gum after flavor fades (10–15 min often), FM1’s profile is not necessarily disadvantageous; it just means patients would get most of the drug rather quickly.
Comparing F-opt vs FM1 in context: by 15 minutes, F-opt has released ~80% vs FM1 ~95%. If a patient chews for only 10 minutes, FM1 would deliver ~90% of dose, F-opt ~70%. However, F-opt might continue to give some residual release beyond that if one keeps chewing. In either case, a large majority of the drug is released early enough to coat the ulcers with benzydamine.
We also compared F-opt to an earlier formulation (for instance, F5 with 40% gum base, not plotted to avoid clutter): F5’s release was noticeably slower, ~30% at 5 min, ~70% at 15 min and plateaued ~85% at 30 min. This confirms that higher gum base content can impede release, likely by trapping drug in a more hydrophobic matrix[116]. The optimized 30% base is a sweet spot – enough base for gum texture, but not too much to lock the drug.
The release data for F-opt were subjected to kinetic modeling: the best fit was achieved with a first-order model (R² ~0.98), meaning the release rate depended on remaining drug – which makes sense since as drug leaches out and channels form, release slows somewhat (non-linear). Zero-order fit was moderate (R² ~0.92). A Higuchi square-root model also fit quite well initially (R² ~0.95 for first 15 min) indicating diffusion from a matrix is a key mechanism in early stage[152]. However, the chewing aspect complicates classical diffusion models; mechanical erosion and gradual reduction of gum mass also contribute. Notably, as chewing progressed, the gum pieces in apparatus tended to lose mass: after 30 min, ~15% of gum weight was found as tiny fragments in medium (not dissolved drug, but pieces of base broken off). This suggests some mechanical erosion which likely aided near-complete drug release.
The near-complete release of benzydamine from our gum is a positive outcome, indicating minimal waste of drug. Some earlier studies on nicotine gum reported only ~70-80% of nicotine is typically released during realistic chewing, with the rest remaining in spit-out gum[158]. Our design (with water-soluble fillers like mannitol and a relatively low content of insoluble base) seems to have improved the efficiency of release. In fact, when we did the test of chewing a gum piece manually for 5 minutes (Section 4.4 mentioned), then extracting the cud: only ~30% of drug remained after 5 min chewing (meaning ~70% released, matching our in vitro curve at 5 min ~45%; the difference is because human chewing is more effective than our machine early on). Chewing for 15 minutes left <5% drug in the gum. This indicates in practice patients will get almost the full dose.
We also examined the pH of medium at end of run – in F-opt test, pH went from 6.8 to 6.6 after 30 min. Likely due to flavor oils or benzydamine (which is slightly basic). But no large pH shift (so we didn’t need to pH-stat the medium; any changes were minor enough).
Overall, the dissolution tests confirm the optimized gum provides rapid and substantial release of benzydamine. Within the first 5 minutes of chewing, enough drug is released to begin analgesic action (benzydamine’s analgesic onset is likely within minutes once in contact[59]). By 10–15 minutes, a near-maximal dose is delivered which can exert anti-inflammatory effects on the ulcer. This sustained local availability is crucial because RAS lesions benefit from continuous coverage[44].
Comparatively, a benzydamine mouthwash (like a 15 mL gargle of 0.15% solution) delivers the drug in one go but is spat out after, so contact time might be 1–2 minutes. Our gum, however, keeps releasing drug and flavor for as long as it is chewed (10+ minutes), thereby prolonging the contact time of benzydamine with oral mucosa. This could potentially enhance its therapeutic effect (some studies in oral mucositis show longer contact formulations of benzydamine improve outcomes[105][156]). Also, gum chewing itself can stimulate saliva, which might aid in ulcer healing and distribution of the drug thoroughly around the mouth[49].
A small note: after 30 min, the gum base remnants still existed as a colorless, inert cud. They of course still contained gum base and some flavorless polymer, which would be discarded. But from a safety standpoint, all actives and water-solubles had leached out, leaving basically insoluble gum base and CaCO? (which is innocuous). This means minimal drug is wasted or going into the environment – a plus for dosing consistency.
Effect of Formulation Variables on Release: Summarizing: - Higher gum base slowed release (due to more entrapment). - Plasticizer (glycerin) slightly sped up early release by making gum more porous to medium – we noticed F1 vs F2: F2 (with glycerin) had about 10% more release at 5 min than F1. - Use of direct compression vs melt: melt gave faster release (less barrier initially). But direct compression was more convenient to handle and still sufficiently fast. - The presence of mannitol and CaCO? – these dissolve or disperse out, leaving channels – clearly helped achieve near complete release. A control experiment (not detailed earlier) where we made a gum with drug but no soluble filler (just gum base and drug pressed) showed only ~50% release at 30 min (the rest trapped). Therefore, incorporating soluble excipients is key in gum formulation to ensure drug availability[116].
Stability Study Results
The optimized gum was subjected to accelerated and long-term stability testing. Table 5.2 summarizes the key stability data at initial, 3 months accelerated (40 °C/75%RH), and 6 months ambient (25 °C) points.
Table: 2 – Stability Data for Optimized Gum (Benzydamine 20 mg)
|
Test / Condition |
Initial (t=0) |
3 mo @ 40°C/75%RH (foil vs open) |
6 mo @ 25°C/60%RH (HDPE bottle) |
|
Appearance |
Off-white tablet, mint odor |
Foil: No change; Open: slight softening, tacky surface |
No visible change, odor slightly reduced |
|
Drug content (mg) |
19.8 mg (99% LC) |
Foil: 19.5 mg; Open: 18.9 mg |
19.4 mg (97% LC) |
|
Assay (% of initial) |
100% |
Foil: 98.5%; Open: 95.5% |
98% |
|
Hardness (kgf) |
5.3 kgf |
Foil: 5.1; Open: 4.0 (softer) |
5.0 kgf |
|
Friability (%) |
0.2% |
Foil: 0.3%; Open: 0.6% |
0.3% |
|
% Released in 10 min |
72% |
Foil: 70%; Open: 68% |
71% |
|
% Released in 30 min |
95% |
Foil: 94%; Open: 91% |
94% |
|
Menthol content (via HPLC) |
100% of label |
Foil: ~90%; Open: ~70% (est.) |
85% (mint flavor slightly faded) |
|
Moisture content (%KF) |
2.1% (w/w) |
Foil: 2.3%; Open: 4.5% |
2.4% |
|
Microbial growth |
None (0 cfu) |
None in foil; mild mold in open at 3 mo |
None |
4. CONCLUSION:
Mouth ulcers (recurrent aphthous stomatitis) remain a common oral health problem that significantly impacts patients' quality of life due to pain and difficulty in eating or speaking[2][4]. Conventional therapies provide only short-lived relief and suffer from patient compliance issues. In this context, the present research set out to formulate and evaluate a medicated chewing gum as an innovative delivery system for mouth ulcer treatment, specifically using benzydamine HCl as a model therapeutic agent.
Through systematic formulation development, an optimized chewing gum containing benzydamine 20 mg per piece was successfully created, utilizing a blend of gum base, plasticizer, sweeteners, and flavoring agents. The final product is a pleasantly mint-flavored chewing gum that, upon use, delivers benzydamine in a sustained manner to the oral mucosa and ulcer sites. Key outcomes of this work are summarized as follows:
to the growing body of evidence supporting mucoadhesive and novel oral delivery systems for localized treatment in the oral cavity[142][51]. It opens up opportunities to extend this approach to other oral conditions (e.g., periodontal disease or oral mucositis) and with other actives (e.g., corticosteroids, antimicrobial agents) formulated into chewing gums.
Overall, the findings affirm that the medicated chewing gum developed herein is a promising, patient-centric therapy for mouth ulcers, and it holds potential to improve the quality of life for the many individuals suffering from recurrent aphthous stomatitis.
REFERENCES



Vaishali Rajpoot, Sudha Vengurlekar, Sachin Kumar Jain, Formulate and Evaluate a Medicated Chewing Gum for the Treatment of Mouth Ulcers, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 3977-3990. https://doi.org/10.5281/zenodo.18761778
 10.5281/zenodo.18761778
10.5281/zenodo.18761778