
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Institute of Pharmacy, Vikram University, Ujjain, Madhya Pradesh.
In recent years, increasing attention has been directed towards lipid – based drug delivery systems to address the limitations of traditional formulations. Nano Structured Lipid Carrier are created using a mixture of various lipid molecules, usually consisting of both solid and liquid lipids as the core matrix Nano Structured Lipid Carrier advancing the drug loading capacity, stability, and controlled release characteristics. In Nano Structured Lipid Carrier have gained more significance as compared to nano-based techniques because of their high biocompatibility and increased bioavailability. Because of their biologically non-toxic, nonimmunogenic, and compatible nature, Nano Structured Lipid Carrier are becoming a widely adopted nano carrier system Nano Structured Lipid Carrier can be utilized to treat a wide range of diseases, including: cancer, infections, neurodegenerative diseases, hypertension, diabetes, and pain management. It is also possible to produce Nano Structured Lipid Carrier on a large scale, making them one of the promising delivery systems likely to emerge in the pharmaceutical market in the near future.
Nano-structural lipid carriers (NLCs), a completely new kind of nanoscale drug delivery technology, with the potential to increase the stability, solubility, and bioavailability of several medications and bioactive compounds. NLCs include a solid lipid core surrounded by a liquid lipid layer, forming a heterogeneous matrix that may accommodate a wide variety of molecules. NLCs perform better than other nanocarriers such as liposomes, solid lipid nanoparticles, and polymeric nanoparticles in a variety of areas, including biocompatibility, scalability, flexibility, and cost-effectiveness. I'll discuss the state of the art for NLCs as of right now, including their characterisation techniques, preparation processes, and applications in a variety of biotechnology and medical fields, as well as the challenges and opportunities for further study and advancement.1
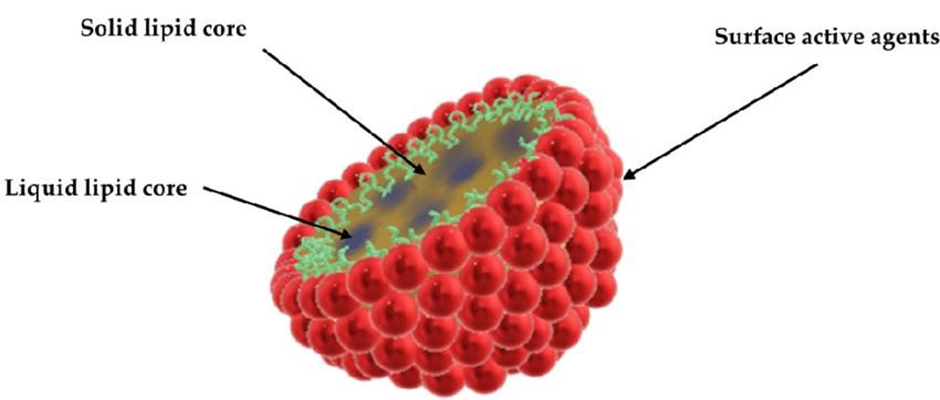
Fig no.1 Nano Structured Lipid Carrier.
NLC types
Depending on the lipids employed, the concentration of the surfactant, the type of medication, the solubility properties, and the synthesis technique, NLCs can be categorized as imperfect, amorphous, or various types.
Type I (Imperfect Type) NLCs
When solid and liquid lipids are combined in an imperfect type, the result is a highly disordered imperfect lipid matrix. High drug entrapment in this created defective lipid matrix is further guaranteed.
NLCs of Type II (Multiple Type)
The double emulsion process, which is just oil in lipids in water, is used to make NLCs of this type. The phase separation technique is used to complete the formulation. This method is used if the drug has more solubility in liquid lipid which further helps to increase drug payload.
Type III (Amorphous) NLCs
Since the lipid matrix of the amorphous variety is amorphous and no crystalline structure forms, no drug can escape from NLCs while they are being stored. Lipids that do not recrystallize when cooled are used to form these kinds of NLCs.2

Fig no:2 Types of Nano Structured Lipid Carrier
Lipid carriers with nanostructures are required.
Bangham identified "swollen phospholipids" as a representation of the cell membrane more than fifty years ago AD; these were subsequently dubbed liposomes.
Since then, research and development into the spontaneous rearrangement of lipids to create nano colloidal particles has continued. In the upcoming years, liposomes will be widely used as drug delivery vehicles due to their exceptional capacity to properly deliver medications while lowering toxicity and boosting bioavailability.
The first FDA approval of liposomal doxorubicin in 1995 marked the beginning of a new era in lipid-based nanomedicine. Liposomes offer a secure and efficient biodegradable medication delivery system. The lipid nanosystem based on vesicles failed not fulfill expected to be a flexible drug carrier because of its physiological stability, the complexity of nanomedicine, and its high cost, which has prompted researchers to look for novel lipid systems. Although nanoemulsion is one of the less expensive options to the vesicle-based technology, it is insufficiently stable and safe for medications.
Owing to the drawbacks of the previous lipid-based nanosystem, a new class of lipid-based particulate systems was created as a substitute by fusing the benefits of polymer nanoparticles, liposomes, and nanoemulsion. One Müller et al. separately created first-generation lipid nanoparticles (SLNs) using several techniques in the early 1990s as an affordable and adaptable drug delivery strategy. The primary purpose of these lipid-based nanosystems is cosmetics. The medicine may be encapsulated in a solid lipid matrix core using SLNs, which are solid lipid cores surrounded by lipidic and surfactant shells.
Because of their many benefits, SLN are good drug carriers for lipophilic medications. They are made of biodegradable materials, have a scalable, quick, and efficient manufacturing process, and are physiologically more stable than other lipid-based nanosystems. Unlike liposomes, they may be kept in an aqueous state for an extended period of time thanks to their solid lipid core.
A significant disadvantage for effective drug delivery is the poor drug loading capacity of solid lipid nanoparticles, which are made up of an ordered solid lipid core. Another drawback of SLN is its initial burst release and stability problems over long-term storage, including drug leakage and the polymeric transition to crystalline form.
To address issues related to the SLN, a novel second-generation lipid nanoparticle surfaced in 1999.
In addition to having a higher drug loading capacity and greater stability than SLN, NLC is a nanocarrier that possesses the benefits of earlier lipidic nanoparticles. Twelve Because of the qualities they provided, NLCs entered the cosmetics industry in 2005, and as of right now, there are about 40 cosmetics items available. Because of NLC's intrinsic properties and effective encapsulation ability, this drug delivery technology has not yet reached the market. As the name implies, NLCs are multiparticulate systems that are nanoscale and range in size from 50 nm to 500 nm. The composition and production technique of the nanoparticles determine the NLC particle size dispersion. This particle is colloidal in nature and has a structure similar to SLN, with the primary difference being in the core of the nanoparticle. In contrast to SLN, which has a solid lipid core with highly ordered lipid arrangement, NLC has both a solid lipid core and lipid liquid, resulting in a disorganized drug matrix. More pharmaceuticals can be placed into the core because of its disorganized nature. Additionally, NLC fixes stability problems caused by crystallization and drug expulsion that arise during long-term storage of SLN. The 70:30 to 99.9:0.1 ratio of solid to liquid lipids that makes up the NLC core alters it. There is more room for drug integration in these nanocarriers because the lipophilic molecule dissolves better in liquid lipid and because of lipid blend imperfections. Since all of the ingredients used to formulate NLC should be non-toxic and biocompatible, they should all be regarded as generally regarded as safe (GRAS).
Drug-lipid compatibility should also be taken into consideration when choosing a lipid for NLC. In addition to lipids, which are utilized in a binary mixture of liquid and solid lipids, one or more surfactants, ranging from 1.5% to 5% (w/v), are used to stabilize the nanosystem. A layer of surfactant surrounds the NLC core. The size of the particles and the physicochemical characteristics of NLC are significantly influenced by the choice of lipid and surfactant. Since combining several surfactants reduces particle size and crystallinity compared to a single surfactant system, the type and quantity of surfactants is an important formulation parameter. A system with two distinct surfactants is more stable.
The fundamental structure and organization of solid and liquid lipids alter as a result of variations in lipid concentration and formulation parameters. There are three possible variations:
Muller et al. divided NLC into three categories.
Lipid content is low in the first kind (imperfect crystal type), which causes distortion in the solid lipid crystalline structure. The structure and imperfections of nanoparticles can be altered by varying the fatty acid triglycerides.
Increases in lipid concentration lead to higher drug incorporation because lipophilic medicines dissolve better in liquid lipid. In the type two (multiple) or without the addition of cosurfactants, which create an aqueous phase in water. After that, the heated aqueous phase and the preheated lipid phase are combined while being constantly stirred.
A microemulsion is produced. A high-pressure homogenizer is used to reduce the size of this heated microemulsion. Different cycles of homogenization might be used according to the size of the particles that are wanted. To transform this nanoemulsion into NLC, it is cooled Longterm use of an intermediate pressure (1000 bar) produces NLC particles that are smaller than 100 nm.24 For medications or materials that break down at high temperatures, this method is not recommended. The methodical procedures to formulate NLC are shown in Figure 3.
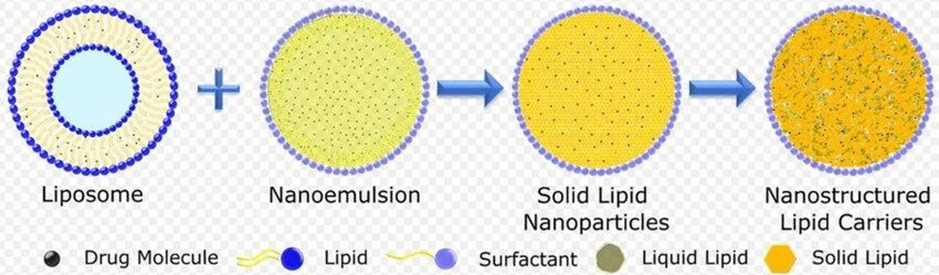
Fig no 3: Evolution of lipid based nanocarriers LIPID:
Lipid is the main ingredient of nanostructure lipid carriers, which control drug loading capacity, prolong activity, and maintain formulation stability. NLC has been formulated using solid lipids such as fatty acids, triglycerides, diglycerides, monoglycerides, steroids, and waxes.
When creating lipid nanoparticles, lipids with physiologically acceptable, biodegradable, nontoxic, and generally recognized as safe (GRAS) status are favored. Selecting the right lipids is crucial before using them to create nanoparticulate carriers. The kind and composition of lipids influence a number of nanocarrier traits. In practice, it has been proposed that the best fitting criterion for selecting an appropriate lipid is the solubility or obvious partition coefficient of the bioactives in the lipid. Because it influences drug loading and encapsulation effectiveness, the solubility of the drug molecules in lipid is essential.
Screening of solid and liquid lipid:
Liquid lipid:
The solubility of drug in various liquid lipids and surfactants was determined by adding excess amounts of drug in 3-5 ml of oils in small vials. The vials were tightly stoppered and were continuously stirred to reach equilibrium for 72 h at 25 0 C in a mechanical shaker. After that, the mixtures were centrifuged using High Speed Centrifuge at 5000 rpm for 30 min at 370 C.The supernatant was separated, dissolved in methanol and solubility was quantified by UV Spectrophotometer .
Solid lipid :
The solubility determination of drug in various solid lipids was performed by adding drug in increments of 1 mg until it failed to dissolve further in the molten solid lipids (which were heated at 5 0 C above their melting point). The amount of solid lipids required to solubilize drug was calculated.
Surfactants:
The kind and concentrations of surfactants affect the effectiveness and quality of NLC. It has been discovered that the choice of surfactant has a significant impact on the toxicity, physical stability, and crystallinity of NLC . Drug permeability and the degree of drug dissolution are also influenced by surfactant systems. The selection of surfactants is based on their effect on particle size, lipid modification, hydrophilic-lipophilic balance (HLB) value, and mode of delivery.
Due to their amphipathic nature, surface active agents (emulsifiers) are adsorbed on the interface where they lessen the tension between the lipid and aqueous phases .A significant factor in choosing the right kind and quantity of surfactant for NLC formulation is required HLB (rHLB). The rHLB of the lipids and lipid matrix is used to determine how many emulsifiers should be added to the formulation. The emulsifier's HLB value, which is necessary for appropriate emulsification—that is, the reduction of interfacial tension between the water and oil phases—is equal to the lipid's rHLB value. This also aids in achieving a small NLC particle size and a stable nanosystem.
OTHER INGREDIENT
Organic salts and ionic polymers can be employed as counter-ions in the development of nanostructure carriers to circumvent the challenge of encapsulating water-soluble pharmaceutical compounds. Surface-modifiers, another class of excipients used in the formulation of NLC, decrease the drug's phagocytic absorption by the reticuloendothelial system's (RES) macrophages. Lipids particles are coated with hydrophilic polymers, such as PEG, poloxamines, or poloxamers, to extend the duration of medication molecules' residence in the bloodstream. Surface alteration may provide additional benefits such improved drug targeting, better transport across epithelium, and improved physical stability and biocompatibility. 3
EXCIPIENTS USED IN NLC 7,9-20
Table no. :1 Surfactant used in nano structured lipid carrier
|
Sr. No. |
Solid lipid |
Liquid lipid |
|
1. |
Glyceryl monostearate |
Capryol® PGMC |
|
2. |
Gelucire® 43/01 |
Medium chain triglycerides |
|
3. |
Lauric acid |
Capryol TM 90 |
|
4. |
Glyceryl tripalmitate |
Capmul PG-8 |
|
5. |
Cetyl palmitate |
Labrafec CC |
|
6. |
Stearic acid |
Labrafil WL 2609 BL |
|
7. |
Palmitic acid |
Labrafil ILM 1944 CS |
|
8. |
Compritol ATO 888 |
Labrafil M 1944 CS |
|
9. |
Dynasan 114 |
Labrafil M 1944 CS |
|
10. |
Dynasan118 |
Castor oil |
|
11. |
Dynasan118 |
OLIEC ACID |
|
12. |
Softisan® 154 |
Capmul MCM C8 |
|
13. |
Trimyristin |
Isopropyl myristate |
|
14. |
Percifac ATC 5(Glyceryl palmitostearate) |
Phosal 53 MCT, Captex 100 |
|
15. |
|
cremophore EL |
|
16. |
|
Solutol HS-15 |
|
17. |
|
Corn oil |
|
18. |
|
Ethyl oleate |
|
19. |
|
Soyabean oil |
|
20. |
|
Miglyol-812 |
|
21. |
|
Cetyl recinoleate |
Table no. :2
|
Surfactant/ solubilizer |
Reference |
|
Pluronic® F- 68 (Poloxomer 188), Pluronic® F-127, (Poloxomer 407), Sodium taurocholate Sodium dodecylsulfate, Tyloxapol, Soy phosphatidyl choline, Polyvinyl alcohol, Egg lecithin (PC-98T), Soy lecithin (S 75), Polysorbate 80 (Tween 80), Polysorbate 60 (Tween 60), Polysorbate 20 (Tween 20), DSPE-PEG (Distearoylphosphatidyl-ethanolamine PEG) |
4, 9, 10, 14, 17, 20, 7, 7, 7, 7, 7, 7, 12, 12, 15, 16, 19, 7, 7. |
Benefits and drawbacks of nanoparticles
Benefits
Drawbacks
Methods of preparation of NLS’s
?ere are several methods are developed for the preparation of NLS’s. ?e most command methods are as follows:6
Methods of preparation lipid screening
?e screening of lipids stepwise, for the preparation of NLC’s are shown stepwise in Figure 4.
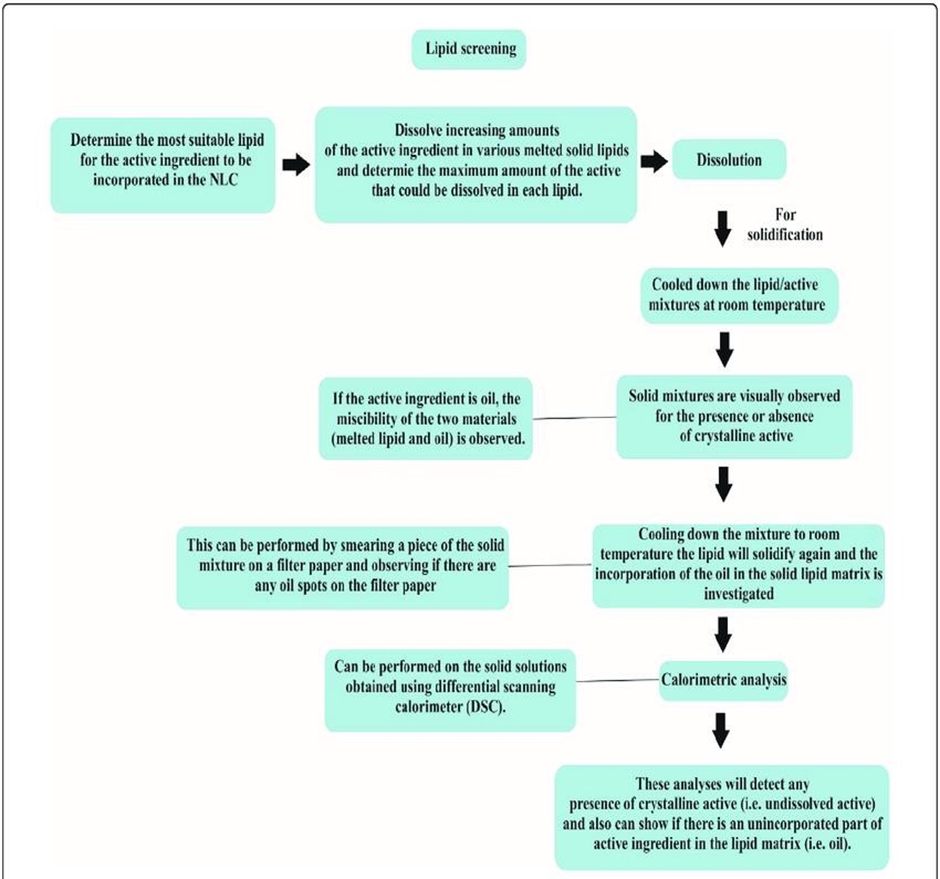
Fig :4 Lipid screening of NLC
High-pressure homogenization
In this process a stable emulsion can be made which involves the subdivision particles into nanosize. In the market, two types of homogenizers are available a) jet-stream homogenizers b) piston-gap homogenizers. ?ere are three methods predominantly used to prepare NLCs by High-pressure homogenization are as follows and shown in Figure.
?e hot homogenization process is executed always at temperature above the melting point of the lipids used in the formulation. While in cold homogenization method the lipid melt is cooled and the solid lipid is ground to lipid micro particles.6
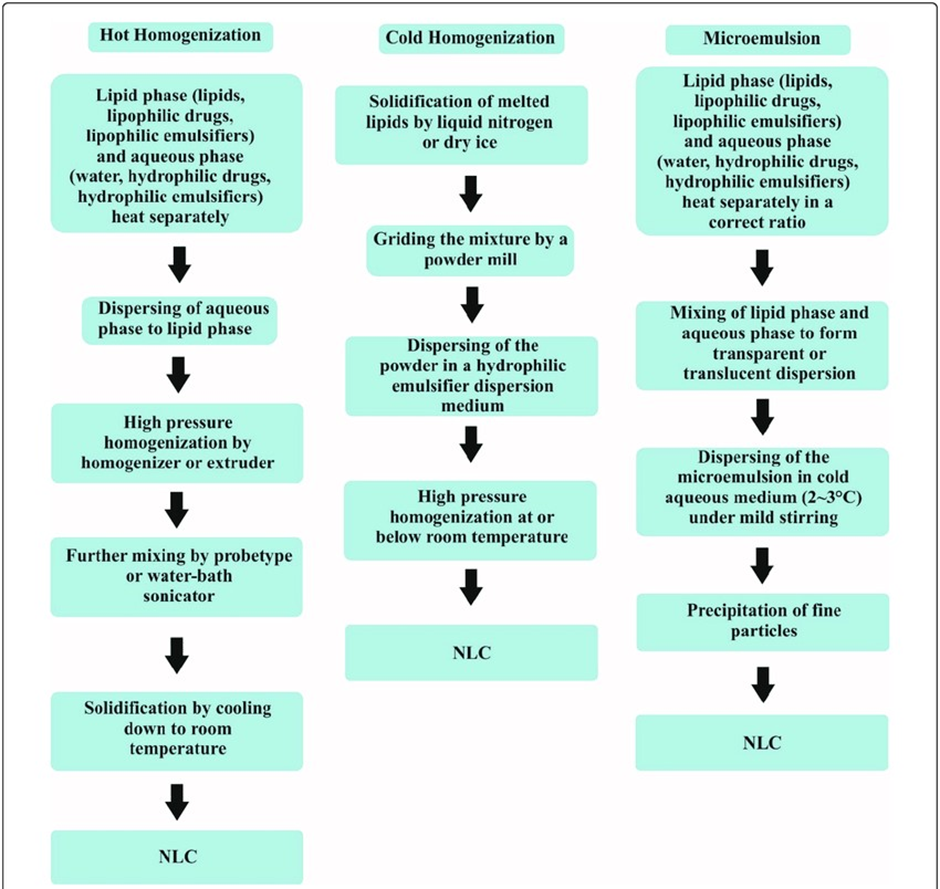
Fig :5 High-pressure homogenization
Method of Melt Emulsification
This method blends and warms solid-liquid lipids. The drugs are introduced to the aforementioned blend in order to frame an organic phase. The addition of the organic phase to the water phase, which contains a surfactant, produced a coarse emulsion. Using HPH, the NLCs are framed. The main features of this technology are the absence of organic solvent residue, the first-ever absence of burst discharge, and scatterings with a high lipid concentration.
The primary drawbacks of this process are the residual organic solvent and the fact that it is not entirely practical for industrial manufacturing. Zhiqiang Tian et al. used this technique with Captex 100 as the liquid lipid and Precirol ATO 5 as the solid lipid to entrap fenofibrate, yielding an encapsulation efficiency of 8.5% and particle size of 227.5 nm. 7
A brief review of the work already done in the field:
1. Patil at el. Due to its significant metabolism in the liver and stomach and its insolubility in water (19 mg/L), felodipine is a BCS class II medication with poor and variable bioavailability. To increase Felodipine's solubility and bioavailability, nanostructured lipid carriers (NLCs) were created for the study. Methods: High shear homogenization by ultrasonication was used to create NLCs loaded with felodipine. Particle size, polydispersity index, entrapment efficiency, drug content, stability, in vitro drug release, and in vivo bioavailability tests were all used to describe the NLCs. Findings: For improved formulation F2, the mean particle size and polydispersity index were 187.0±0.06 and 0.259±0.002, respectively. The drug content attained for formulations F1 through F5 ranged from 51.15± 0.01 to 69.14± 003%. The improved formulation's zeta potential, which demonstrated good stability, was determined to be -38.2 mV. The maximum % entrapment effectiveness, 75.15±0.003, was demonstrated by Formulation F2. Studies on in vitro drug release revealed a sustained release pattern, with the F2 formulation achieving a maximum drug release of 72.82% after 12 hours. Comparing Felodipine NLCs to the marketed medication, the bioavailability trials showed a notable improvement in bioavailability.The NLCs are more stable at 4±2°C, according to stability tests done for optimized formulation F2. In conclusion, felodipine-loaded nanostructured lipid carriers were able to increase the medication's bioavailability by 2.0 times compared to the commercially available product and also showed a longer-lasting drug release pattern.29
Additionally, a dermatokinetic model was fitted to the prepared and characterized ACE-loaded NLC formulation, and its effects on skin integrity were assessed. The study's findings validated the ACE-NLC formulation's stability, spherical shape, and smooth morphology and nanometric size as determined by Zetasizer and scanning and transmission electron microscopy.
The ACE-NLC-gel formulation demonstrated improved skin dispersion in the dermis and epidermis, as well as good rheological and textural properties. Additionally, ACE-NLC preserved the integrity of the skin by penetrating deeper into the skin layers. In comparison to the traditional Mkt-gel formulation, the NLC-based gel formulation of ACE may be a potential nanoscale lipid carrier for topical delivery.30
2. Garg at el. Aceclofenac (ACE), a cyclooxygenase-2 inhibitor, is the derivative of the diclofenac group that has been in use for the symptomatic treatment of systemic inflammatory autoimmune disease, rheumatoid arthritis (RA). Partial solubility, high lipophilic nature, and stability challenge its application in producing topical formulations. created and described an ACE hydrogel based on nanostructured lipid carriers (NLC) for effective transdermal administration. Several lipids were used to create the NLC microemulsion, which was then evaluated for drug encapsulation efficiency, surface morphology, zeta potential, and particle size. In order to evaluate in vitro drug release, rheology, textural profile, in vivo skin retention and penetration, and stability, the optimized NLC formulation was added to Carbopol® 940 gel. This configuration was then described and contrasted with the currently available commercial gel (Mkt-gel)
3. Jacinto at el. The development and assessment of a dual hydrogel system based on Carbopol® and nanostructured lipid carriers (NLC) as a possible transporter for the topical administration of curcumin to the skin is the main objective of this work. Using dynamic light scattering, two populations of negatively charged NLCs of varying sizes (P1, 70–90 nm and P2, 300–350 nm) were created and described. Transmission electron microscopy methods verified that NLCs have an oval platelet form. Using spectrophotometric techniques and high pressure liquid chromatography (HPLC), the entrapment efficiency and release patterns of curcumin NLC were evaluated. Curcumin (CUR) antioxidant activity in NLCs was preserved and increased by up to seven times, and tests of cell viability on fibroblasts and keratinocytes showed that CUR-NLCs had a moderate anti-migration/proliferation effect and were noncytotoxic at concentrations up to 10 µM. After that, CUR-NLCs were incorporated into a hydrogel based on Carbopol® without affecting the gel's mechanical characteristics. Franz diffusion cell penetration tests in CUR-NLCs and CUR-NLCs/gels over a 24-hour period revealed a 22% and 5% buildup of CUR in Strat-M® membranes, respectively. The utility of this novel dual CUR-NLC/hydrogel combination as an adjuvant treatment in topical dermal applications is supported by all of the data that has been given.31
4. Ortiz at el. The physicochemical properties and behavior of medications that are poorly soluble in water, such as their oral bioavailability and aqueous dispersibility, could be greatly enhanced by lipid nanocarriers.
Based on a combination of solid lipid glycerides, fatty acid esters of PEG 1500 (Gelucire® 44/14), and an oil mix made up of capric and caprylic triglycerides (Miglyol® 812), this study introduces a unique nanostructured lipid carrier (NLC). These NLCs were created using a straightforward low-energy technique based on melt emulsification, which produced narrowly distributed, strongly encapsulating nanoparticles (~100 nm, PdI = 0.1, and zeta potential = ~10 mV). Because of its spectroscopic characteristics and poor water solubility, rhodamine 123 was chosen as a pharmacological model. Dynamic light scattering (DLS), zeta potential, and nanoparticle tracking analysis (NTA) were used to characterize the new NLCs. colloidal stability, differential scanning calorimetry (DSC), and transmission electron microscopy (TEM). A dialysis bag and vertical Franzs' cells were used to measure the drug release in order to shed light on the applicability of the techniques, showing comparable results despite their disparate fluid dynamics. According to Korsmeyers-Peppas kinetic modeling, rhodamine 123 has a distinctive biphasic release profile because of the hydrophilic polymer coating's swelling and the diffusion process from the lipid core. Moreover, a number of molecular dynamics simulations were carried out to clarify the synthesis and integration of Rhodamine 123 into the NLC core. It has been demonstrated that temperature has a significant role in enhancing nanoparticle production.
Furthermore, the inclusion of liquid lipid to the formulation results in nanoparticles with defective centers, as opposed to nanoparticles in its absence. Additionally, Miglyol® 812 increases the solubility of hydrophobic molecules. These findings point to the possibility of using new NLC as a drug delivery method for medications that are not particularly soluble in water.32
5. Gorle at el. The purpose of the Nanostructured Lipid Carriers (NLCs) was to increase Axitinib's water solubility and, eventually, oral bioavailability. Using the High-Pressure Homogenization process, axitinib-loaded NLCs were created with Compritol ATO 888 as the solid lipid, Oleic acid as the liquid lipid, and Tween 80 as the surfactant. The impact of the independent parameters, surfactant and the solid-lipid to liquid-lipid ratio (6:4), on the dependent variables, including Mean Particle Size (MPS) and Entrapment Efficiency (%EE), was investigated using a full 23 factorial design. The optimized formulation provided good stability for the produced NLCs with 202.2 nm MPS, -21.5 mV zeta-potential, 0.44 PDI, and 88% EE. By analyzing the findings of a study using Fourier transform infrared spectroscopy and differential scanning calorimetry, the physicochemical characterization of AXT loaded NLCs was completed. The improved formulation's stability investigation revealed no appreciable changes in drug content or particle size. The in vitro release profiles of NLCs showed that AXT had a prolonged-release profile until roughly more than 10 hours after a burst release for the first two hours. Better bioavailability, dose reduction, frequency of administration, dose-related side effects, and disease control may all be offered with Axitinib NLCs. The aforementioned findings demonstrated the potential of NLCs to significantly increase the oral bioavailability of poorly soluble Axitinib in the treatment of cancer.33
6. Sherwin at el. In the industrialized world, herpes keratitis is the most prevalent infectious cause of blindness. Acyclovir (ACV) can be used to treat it, but it requires high and frequent dosages due to its poor solubility and low ocular absorption. By increasing corneal penetration and extending the time that infected cells are exposed to the antiviral agent, nanostructured lipid carriers (NLCs) have been studied as a means of increasing the ocular bioavailability of ACV. The hot microemulsion method was used to create NLCs, which were then coated with 0.5% w/v chitosan. Investigations were conducted into the new lipid-based formulations' in vivo corneal penetration, ex vivo tolerance, and cell uptake effectiveness. . High performance liquid chromatography (HPLC) and fluorescence microscopy, respectively, demonstrated that NLCs could enhance the cell absorption of ACV and encapsulated fluorescein. After 24 hours of exposure, the antiviral effectiveness of ACV entrapped in NLCs rose by 3.5 times. When compared to a commercially available ACV ophthalmic ointment, the formulation's in vivo corneal penetration was examined in Albino rabbits with NLCs that could increase the corneal bioavailability by 4.5 times. In summary, NLCs improved ACV's antiviral and ocular bioavailability through greater corneal penetration, prolonged release, and cell internalization.34
7. Abo El-Enin at el. The purpose of this study was to develop, optimize, and assess berberineloaded nanostructured lipid carriers with chitosan overlay (BER-CTS-NLCs) for effective intranasal brain delivery. By using a hot homogenization and ultrasonication technique, the nanostructured lipid carriers containing berberine (BER-NLCs) were created. They were then optimized for the effects of several causative factors, such as the concentration of glycerol monostearate (solid lipid), poloxamer 407 (surfactant), and oleic acid (liquid lipid), on particle size, entrapment, and the overall drug release after 24 hours. Chitosan was then applied to the ideal BER-NLCs formulation. Their properties were estimated, including diameter, in vitro release, surface charge, morphology, ex vivo permeability, pH, histology, and in vivo (pharmacokinetics and brain uptake). BER-CTS-NLCs were 180.9 ± 4.3 nm in size, exhibited sustained-release characteristics, 36.8 mV positive surface charge and increased ex-vivo penetration through the nasal mucosa. The BER-CTSNLCs system is safe for nasal delivery, according to the histopathological evaluation. Experiments on pharmacokinetics and brain accumulation revealed that mice given BER-CTS-NLCs intranasally had significantly higher drug levels in their brains. In comparison to BER solution (IN), the ratios of BER brain/blood levels at 30 minutes, AUCbrain/AUCblood, drug transport percentage, and drug targeting efficiency for BER-CTS-NLCs (IN) were greater, indicating improved brain targeting. Through intranasal therapy, the optimized nanoparticulate system is thought to be a successful strategy for increasing the effect of BER in treating CNS illnesses, including Alzheimer's disease.35
8. Gilani at el. The current study used the melt emulsification ultrasonication approach to create luteolin (LTN)-encapsulated chitosan (CS) coated nanostructured lipid carriers (NLCs). The 33-QbD technique was used to optimize NLCs for increased in vitro efficacy against breast cancer cell lines. Several in vitro and ex vivo tests were used to successfully characterize the improved LTN-CS-NLCs, and their cytotoxicity in MDA-MB-231 and MCF-7 cell lines was assessed. The produced LTN-CS-NLCs displayed entrapment efficiency (%EE), polydispersity index (PDI), and particle size (PS) in the range of 0.11 and 0.20, 65.55% and 95.37%, and 101.25 nm to 158.04 nm, respectively.
Particle size, encapsulation efficiency, and zeta potential changes were all favorably enhanced by coating NLCs with CS. Furthermore, during the 24-hour testing period for LTN-CS-NLCs, a slow-release rate of LTN was attained. Furthermore, as compared to LTN-NLCs that were not coated and LTN suspension, optimized LTN-CS-NLCs demonstrated noticeably greater intestinal penetration, gastrointestinal stability, and mucoadhesion. In addition, compared to uncoated LTN-NLCs and pure LTN, LTN-CS-NLCs demonstrated dose- and time-dependent cytotoxicity against MDA-MB-231 and MCF-7 cells together with statistically increased antioxidant capability. Based on the aforementioned results, it can be concluded that LTNNLCs coated with chitosan offer significant promise for the treatment of breast cancer.36
9. Ajiboye at el. Understanding how high shear homogenization (HSH) and ultrasonication (US) affect the physicochemical characteristics of blank and olanzapine-loaded nanostructured lipid carriers (NLCs), as well as their potential for drug loading and drug release profiles from formulated particles, was the goal of this study. Under varying HSH and US durations ranging from 0 to 15 minutes, NLCs were made using various ratios of Compritol and Miglyol as the solid and liquid lipids, respectively.
In every formulation, the medication content and the surfactants (Poloxamer 188 (P188) and tween 80) remained consistent. Particle size, polydispersity index, zeta potential, drug crystallinity, and chemical interactions between lipids and OLZ were assessed for the produced NLCs. The dialysis tube method was used to carry out the in-vitro drug release in phosphate buffer solution (PBS) at a pH of 7.4. The produced NLCs had a spherical shape, were monodisperse, negatively charged, and had particle diameters between 112 and 191 nm. When compared to HSH, US time had a substantial impact on the preparation of NLCs, as seen by the notable decrease in mean particle diameter observed following 5 minutes of sonication. Particle size increased as the amount of miglyol in NLCs increased. The in-vitro drug release was conducted in phosphate buffer solution (PBS) at a pH of 7.4 using the dialysis tube method. The resulting NLCs were monodisperse, spherical, negatively charged, and had particle sizes ranging from 112 to 191 nm. The significant reduction in mean particle diameter after 5 minutes of sonication indicates that US duration significantly influenced the production of NLCs in comparison to HSH. As the amount of miglyol in NLCs grew, so did the particle size.37
10. Pawbake at el. To treat inflammation and problems related to acceleration and prolonged action, Diacerein Nanostructured Lipid Carrier (NLC) topical gel was developed. The nanoparticle dispersion was accurately gelled and described in terms of pharmacology, pH, proliferation, rheology, and in vitro releases. Primary skin irritation tests were used to evaluate the safety of NLC-based gels, and pharmacodynamics research validated their effectiveness. The most popular medications for managing osteoarthritis (OA) pain are non-steroidal antiinflammatory drugs (NSAIDs), despite their severe gastrointestinal and vascular side effects that do not address the underlying systemic cartilage damage. A lot of doctors are older. Diacerein is therefore the recommended medication for severe OA discomfort due to its potent analgesic effect. Diacerein is economical as well as dose-efficient. The gel formulation makes it simple to apply in any amount to a painful area. The NLC-based gel demonstrated quicker start and extended habits up to 24 hours.38
Route of administration Topical administration method
Worldwide, skin-related illnesses are highly prevalent. Low drug efficacy due to inadequate skin penetration or skin permeation of medications from the most traditional formulations is one of the main therapy constraints for these conditions. The main skin barrier, the stratum corneum, should be avoided by switching the penetration pathway from transcellular to paracellular or follicles. NLCs and SLNs are examples of lipid nanoparticles that have been created to improve skin penetration.
SLNs or NLCs are combined with traditional formulations to create these particle formulations. They might be made immediately in a single step, creating NLCs or SLNs filled with drugs. For topical drug delivery, lipid nanoparticles offer numerous benefits, including biocompatibility and biodegradability, a regulated and prolonged drug release profile, strong skin adhesion and close contact, skin hydration, and the production of films to improve dermal and skin penetration.
Oral route
Due to the highest patient compliance, oral drug administration is the most often used drug delivery method.
In oral medication administration, the most significant obstacles that need to be addressed are low oral bioavailability brought on by restricted drug solubility and/or strong hepatic first pass effect.
Drug delivery methods based on nanoparticles were thought to be an appropriate way to boost oral bioavailability. One benefit of lipid nanoparticles, like SLNs and NLCs, is their capacity to deliver drugs continuously while maintaining steady plasma levels. Furthermore, the commencement of a drug's effect can be accelerated by the faster dissolution rate of nanoparticles with higher specific surface area and saturation solubility. Chemical or enzymatic degradation and pglycoprotein efflux pumps are additional significant obstacles in oral medication administration. According to recent studies, certain lipids or surfactants that are employed in lipid nanoparticles have the ability to block pglycoprotein efflux pumps. Lipid nanoparticles loaded with pharmaceuticals may lessen the enzymatic or chemical breakdown of medications incorporated in lipid matrices. Bypassing the liver and avoiding the hepatic first pass effect, lipid nanoparticles may facilitate lymphatic transport .
Administration through the eyes
Due to certain physiological and anatomical characteristics of the eyes, ocular medication administration has numerous restrictions and continues to be difficult. The eyes are an extremely intelligent and complicated organ, and accessing particular ocular tissue requires overcoming a number of obstacles. To get around these obstacles and increase the bioavailability of ocular tissue, novel medication delivery techniques as lipid nanoparticles were taken into consideration. The most popular method of administering medication to the front portion of the eyes is topical administration. For superficial ocular disorders, this injection method is the preferred option due to its numerous benefits. The blood ocular barrier, corneal epithelium, conjunctival blood flow, and tear drainage are the main obstacles in this pathway. When utilized as ocular drug delivery systems, lipid nanoparticles can cross the blood-ocular barrier, provide controlled and prolonged drug release, shield medications from lacrimal enzymes, and extend the duration of drug deposition and residence in the eyes. It is particularly challenging to treat ocular illnesses that affect the posterior portion of the eyes. The posterior portion of the eyes can be targeted in a variety of ways. Targeting intraocular tissues with a topical approach is not appropriate; instead, transscleral administration (subconjunctival and retrobulbar injection), intravitreal route, subretinal injection, etc. are utilized for this purpose. Since the majority of these methods are intrusive, innovative drug delivery techniques like lipid nanoparticles might be a suitable substitute. Gene delivery via nonviral vectors, such as SLNs and NLCs, was also explored for the aim of retinal targeting in retinal disorders.
Parenteral administration
Enhancing parenteral drug distribution is a major function of nanomedicine and nanotechnology. The main benefits of using lipid nanoparticles for this purpose are their ease of production scaling up, the biocompatibility and biodegradability of the formulation's ingredients, their controlled and adjustable drug release pattern, their ability to stop drug degradation, and their ability to maintain more consistent drug serum levels. Lipid nanoparticles laden with drugs can be administered intravenously, subcutaneously, intramuscularly, or straight to the intended organs. Lipid nanoparticles can release drugs either by diffusion, which may promote a prolonged drug release, or by erosion, such as enzymatic destruction. Lipid nanoparticles' ability to incorporate proteins and peptides has been validated by recent studies. Because of their restricted drug loading capacity, SLNs are not a good carrier in this situation; however, NLCs are a suitable substitute. Peptides and proteins can be shielded from adverse environmental conditions with this technique.
Delivery of pulmonary
A relatively novel strategy with several benefits is pulmonary medication delivery. It is a noninvasive method of administering drugs locally and systemically. This direct administration method may lower the amount of the medication, which would lessen its side effects. The beginning of effect can potentially be accelerated by direct drug inhalation.
An additional benefit of this mode of administration is the high drug accumulation in the target site. High drug permeability may be ensured by the pulmonary system's large surface area and thin alveolar epithelium. Lipid microparticles were employed as lung-targeting delivery methods. Comparing these particle systems to traditional formulations, positive outcomes were seen, including an increase in medication bioavailability. For pulmonary delivery, lipid nanoparticles, such as SLNs and NLCs, have been investigated. Compared to previously developed particle systems, they offer the advantages of sustained drug release, biocompatibility and biodegradability, reduced toxicity, and improved stability. Drug-loaded nanoparticles delivered through the lungs would produce a high local concentration and lessen systemic side effects. For systemic distribution, nanoparticles can also attain greater bioavailability.
Like other modes of administration, lipid nanoparticles utilized in lung medication delivery offer the benefit of prolonged drug delivery.
Delivery of the brain
Because of the blood-brain barrier (BBB), one of the most significant problems in pharmaceutical science is getting drugs into the brain. For precise targeting of brain tissues, nanoparticles with the benefits of small particle size and high drug encapsulation efficiency have been explored. Nanoparticles are appropriate as brain drug delivery devices because they can avoid the reticuloendothelial system (RES). Limited drug penetration across the bloodbrain barrier and drug efflux from the brain into the bloodstream are two significant barriers to brain medication delivery. To get over these obstacles, colloidal drug delivery methods have been used, including lipid nanoparticles like SLNs and NLCs. The benefits of lipid nanoparticles include extending the duration of drug retention in brain capillary blood, causing a drug gradient from blood to brain tissues, opening tight junctions to make it easier for drugs to pass through the blood-brain barrier, and facilitating the transcytosis of drug-loaded lipid nanoparticles through the endothelium layer. Both lipophilic and hydrophilic medications that can be delivered through various methods can be incorporated into lipid nanoparticles. The importance of surfactant compatibility for brain medication delivery was highlighted in earlier studies. It is possible to select suitable surfactants based on their packing characteristic and HLB. Polysorbates, particularly polysorbate 80, have demonstrated the best outcomes for sitespecific drug delivery in the brain. Furthermore, findings demonstrated that positively charged lipid nanoparticles improve medication accumulation in the brain.8
OTHER APPLICATION
Cosmetics
NLCs have recently been created using a controlled nanostructuring of the particle matrix, which offers significant benefits in terms of long-term stability and loading capacity. NLC dispersions can be administered as gel, cream, lotion, or ointment, among other forms. Enhancing skin bioavailability of active ingredients, film formation and controlled occlusion, UV protection, penetration enhancement and epidermal targeting, improving physical and chemical stability, and in vivo skin hydration are just a few of the many advantageous features linked to these NLCs in cosmeceuticals.With fewer adverse effects, NLCs significantly enhanced oxybenzone's in vitro SPF and erythemal UVA protection factor by more than six and eight times, respectively. According to studies, NLC with Cutanova Cream NanoRepair Q10 (Dr. Rimpler, Wedemark, Germany), outperformed a traditional o/w cream with the same composition in terms of skin hydration. Another partially completed cosmetic product based on lipid nanoparticles is NanoLipid Restore CLR (Chemisches Laboratory, Dr. Kurt Richter, Berlin, Germany). In order to prevent oxidation and improve the stability of the finished product, the readily oxidized black currant seed oil is added to NLCs.
An additional product called Surmer (Dr. Rimpler, GmbH, Wedemark, Germany) improves a day cream's occlusion without altering its light nature; in other words, it achieves greater occlusive qualities without giving the appearance of shiny skin. By adding fragrances and insect repellents to NLCs, a longer release profile can also be achieved.
Chemotherapy
According to recent research, NLCs improved the stability and effectiveness of numerous cytotoxic medications while also lowering their adverse effects. Various nanosystems have been created using anti-cancer medications. For instance, etoposide NLCs were discovered to be cytotoxic against human epithelial-like lung carcinoma cells; topotecan NLCs were stabilized and had a prolonged release in order to treat small-cell lung cancer and refractory ovarian cancer; and albumin-paclitaxel NPs were approved in early 2005 for use in chemotherapy for metastatic breast cancer. High drug loading efficiency, a delayed release profile, improved chemical stabilization, and higher cytotoxicity are benefits of using anticancer medications in NLCs. Because these NLCs circumvent several possible SLN issues, namely drug leakage during storage and reduced loading capacity. To further boost the therapeutic effect, they work by extending the time that tumor cells are exposed to anti-tumor drugs and improving their permeability and retention effect. Additionally, it has been noted that hyaluronic acid coated NLC may increase the amount of paclitaxel (PTX) that accumulates in the tumor and lengthen its stay in circulation in the blood. The experiment's findings demonstrated that compared to Kunming mice harboring Taxolin B16, hyaluronic acid-coated, paclitaxel-loaded NLCs (HA-NLCs) exhibited greater anti-tumor activity and fewer side effects.
HA-NLC's total tumor targeting efficiency was 14.46%, or around 1.4 times that of Taxol.
Nutraceuticals
Bioactive substances known as nutraceuticals offer medical or health advantages, such as illness prevention and treatment. Because of their various roles, structural diversity, and extensive distribution in plant tissues, carotenoids rank among the most significant classes of natural pigments. Natural oils and a flexible high-shear homogenization method were used to successfully create carotene-LNC, which has strong antibacterial and antioxidant properties. Another flavanone that is helpful in chemically induced mammary tumorigenesis, colon carcinogenesis, heart attacks, and blood pressure is hesperetin (5,7,3′-trihydroxy-4′-methoxy flavanone). It was also successfully encapsulated in NLCs, which demonstrated good acceptance, homogeneity, improved taste, and enhanced therapeutic effects.
In the food industry
Because of its great loading capacity and outstanding stability, NLCs are used extensively in the pharmaceutical industry. The use of NLC as a nutritional supplement carrier in the food industry for the manufacturing of capsules and beverages was almost ever documented. The food business still faces significant risks when investing in this sector, nonetheless, because of particular challenges with the supply, availability, and environmental aspects of raw materials. For use in food applications, coenzyme Q10-loaded NLCs were created to improve the physicochemical stability and bioavailability.21
DELIVERY OF GENE
The utilization of novel gene delivery systems for the treatment of human diseases has increased as a result of the development of gene therapy. Since nonviral vectors are rather safe, they are being studied in great detail. Since lipid nanocarriers, such as liposomes and SLNs, are superior delivery vehicles for proteins, enzymes, nucleic acids, and DNA, they can increase therapeutic efficacy. By adding cetylated polyethylenimine (PEI) to NLCs, a unique nonviral vector is created. After effectively escaping the endocytic pathway through the proton-sponge effect, PEI may transport DNA to the nucleus by forming a compound with anionic DNA. The human lung adenocarcinoma cell line and Chinese hamster ovary cells are used to assess the in vitro gene transfer capabilities of NLCs. After triolein is added to the nanocarriers, NLC transfection efficiency is increased. Triolein is a liquid lipid that can disrupt phospholipid planar bilayers and partition in a favored orientation in phosphatidylcholine lipids. The endosomal membrane may become unstable as a result. While the standard Lipofectamine® 2000 considerably reduces transfection in both cell lines, the optimized NLCs do not significantly alter transfection in either cell line when 10% serum is present. NLCs are therefore efficient vectors for gene transfer following triolein insertion.
There is great promise for cancer treatment when small-interfering RNA (siRNA) is used to silence oncogenic targets like survivin. However, their brief action represents a clinical constraint. NLCs are used to address this issue from the standpoint of intracellular siRNA kinetics. Xue and Wong show that intracellular siRNA kinetics can be tailored by adjusting the way NLCs degrade in response to lysomal enzymes. The survivin knockdown phase can be prolonged to nine days with customized NLCs that distribute survivin-siRNA. The anticancer medication docetaxel is given to mice in the in vivo tumor growth test either with or without siRNA-loaded NLCs. In comparison to the negative-siRNA group, the siRNAloaded NLCs group exhibits persistent tumor growth suppression following docetaxel treatment. When docetaxel and siRNA NLCs are combined, the tumor volume is 65% and 55% less than that of blank control and negative-siRNA control, respectively. The new nanocarriers offer a way to establish a weekly or longer siRNA treatment administration cycle that is clinically more acceptable.22
Various NLC methods for increased bioavailability
Direct absorption: by the gastrointestinal tract, or intestinal lymphatic transfer. The NLC may promote the development of chylomicrons, which adhere to the transcellular route of absorption, because of its lipophilic nature and use of long chain triglycerides. The intestinal lymphatic system transports highly lipophilic substances, avoiding hepatic first-pass metabolism. Lipids are necessary for the development of lipoproteins because the components are delivered in conjunction with the intestinal lipoproteins' triglyceride (TG) lipid core.Lingual and gastric lipases in the GIT initiate TG hydrolysis to create a crude TG emulsion, which is then expelled into the duodenum. The synthesis of pancreatic juice, bile salts, and biliary lipids is stimulated by this crude emulsion. e. Biliary lipids adhere to the surface of the TG emulsion, stabilizing it. At the surface of emulsified TG droplets, pancreatic lipase produces fatty acids (FA) and monoglycerides (MG). The enterocyte absorbs the long-chain FA and MG.
The mono-acylglycerol route re-esterifies the ingested FA.These are further digested by several organelles and organized to form the chylomicron's lipid core. Phospholipids and apolipoproteins are added to stabilize the chylomicrons that are produced. Following stabilization, these lipoproteins are released into the mesenteric lymphatic and lamina propria. released into the mesenteric lymphatic and lamina propria.
Muco-adhesion:
The lipid nanoparticles stick to the mucus, lengthening its residence period and causing the medicine to be released from the carrier more readily .
Mixed micelle formation:
The lipids employed in NLC cause bile production in the small intestine because they resemble dietary lipids. Lipids are broken down by enzymes to produce lipid digestion products, which are then combined with bile to create a mixed micelle. This phenomenon increases the drug's solubility, which makes it easier for it to pass through the membrane.
Increased permeability:
Through a variety of processes, surfactants change intestinal permeability. For instance, it is known that poloximer can disrupt the tight connection of intestinal epithelial cells and distort their cell membrane, allowing NLCs to move paracellularly. Additionally, it promotes NLC transport through the intestinal mucosa and suppresses P-gp efflux. Additionally, these offer the necessary steric stability.
Prevents the deterioration of drugs: In the hostile GI environment, several medications become unstable. NLC has the benefit of shielding the medication from enzymatic and chemical breakdown by the lipids, which delays in vivo metabolism.24
Breaking through physicochemical obstacles
Its inherent lipid nature has been the main barrier to the creation of lipid-based nanoparticles, impeding the routing approaches for the nanoparticles' physicochemical characterisation.12 Advanced methods, like as transmission electron microscopy (TEM), which are forceful and jeopardize nanoparticle structure, can be used to visualize LPs. The collected photographs do a terrible job of capturing the appearance of LP. Their stability while being stored is another crucial concern. The only way to retain these nanoparticles is by using the aid of a cryoprotectant and/or preservative, already jeopardizing the LPs' original physicochemical characteristics (namely their size and surface charge). Lastly, it has been questioned whether lipid nanoparticles can be seen in vivo because the lipophilic dyes (like Nile Red) that are used to track LPs are said to be released from the nanoparticles, marking the surrounding tissues where the nanoparticles are thought to have accumulated and halting the nanoparticles' own track. Thankfully, there has been a positive development on these important issues lately. sincerity. The appearance of LP is terribly captured in the gathered photos. Another important consideration is their stability during storage. These nanoparticles can only be retained by employing the aid of a preservative and/or cryoprotectant, already endangering the LPs' initial physicochemical properties (namely, their surface charge and size).
Finally, because the lipophilic dyes (such as Nile Red) used to track LPs are said to be released from the nanoparticles, marking the surrounding tissues where the nanoparticles are believed to have accumulated and stopping the nanoparticles' own track, it has been questioned whether lipid nanoparticles can be seen in vivo. Fortunately, there has recently been progress on these crucial topics.
However, in the absence of a cryoprotectant, lyophilization of LPs typically indicates aggregation. As cryoprotectants for NLC formulations, sucrose and sorbitol at varying concentrations (5, 10, and 15% (w/v)) are used. Following their resuspension, the freeze-dried samples were examined for appearance, rehydration rate, mean particle size, and zeta potential. The only substance that prevented or avoided NLC aggregation at all tested concentrations (5, 10, and 15% (w/v)) was trehalose. Remarkably, trehalose was also reported to support the stability of the shell structure in other lipid nanocapsules It should be remembered that applying a cryoprotectant frequently results in changes to the initial characteristics of the nanoparticle surface and, consequently, an other preservation method. approach might be wise.
Morphology of NLC
Electron microscopic techniques and dynamic light scattering (DLS) are used to characterize drug delivery systems at the nanoscale. and atomic force microscopy (AFM) are frequently used to get trustworthy data regarding the shape and structural characteristics of nanocarriers. They have also been used to study NLCs.
Fluorescent dye labeling of LPs
When using fluorescent dye labeling to examine the biodistribution or cellular trafficking of nanocarriers, a crucial consideration is the labeling's stability in the absence of grafting into the lipid-based nanocarriers. 25
NLCs' Limitations in Drug Delivery.
Although NLCs have several benefits for medication delivery, it's crucial to be aware of any potential disadvantages and restrictions to make sure the creation of improved formulations.
The following restrictions need to be considered when creating NLCs:
(i) Complex formulation:
It can take a lot of time and effort to develop NLCs.
(ii) Polymorphism:
According to this theory, the instability of the Matrix lipid recrystallization causes the suspension to become unstable, which results in the conversion of round particles into needle-shaped ones. The presence of a well-ordered β-structure within the lipid matrix and thermodynamic stability are responsible for the change to a needle-like morphology. It's also important to remember that stabilizers, contaminants, and surfactants can all have an impact on this occurrence. Rapid solvent evaporation during the spray drying process causes unstable polymorphic shapes to arise. Similar results have been reported with the spray congealing process, where the triglyceride's α-form can change into the more stable β-form, which has a higher melting point. This change results in the creation of crystalline aggregates and makes it easier for medications to be released because of a reduction in the number of amorphous zones within the carrier matrix.
(iii) Phase separation:
Particle aggregation may lead to reversible processes like sedimentation and coalescence or irreversible processes like Phase separation results from focculation. Additionally, there is a chance of formation during storage. Surfactants are used to stabilize NLC suspensions and lessen aggregation and gelling problems. Depending on their traits, In this stabilizing process, surfactants play different roles. While nonionic surfactants serve as steric stabilizers, cationic or anionic surfactants can raise the zeta potential value. Solid versions of lipids can be used to solve the storage stability issues related to liquid lipids. This change can be accomplished by methods like spray drying the suspension or lyophilizing it.
(iv) Modification of lipids during storage:
NLCs are dynamic systems where lipid molecules show indications of thermodynamic instability. Their encapsulating ability is improved by this structural feature. However, improved drug mobility is sacrificed for greater drug integration efficiency in these unstable configurations. Drug molecules may eventually be expelled as a result of the crystal lattice's gradual rearrangement during storage, which could produce a thermodynamically stable configuration.
(v) Storage requirements:
4°C is the optimal temperature for NLC storage. This study examined the quercetin-loaded NLCs' physical stability at different temperatures.
Zeta potential (ZP), size, and the polydispersity index were used as stability markers. The findings showed that after 28 days of storage at a low temperature of 4°C, quercetin-loaded NLCs showed stability. However, particle aggregation and a decrease in surface charge were caused by exposure to higher temperatures, specifically 22°C for 10 days and 37°C for 24 hours.
As a result of the rising temperature, the hydrogen bonds between surfactant molecules at the lipid/water interface were broken, causing this phenomenon.
(vi) manufacture challenges:
Maintaining consistent quality during large-scale manufacture may be difficult, and scaling up the production of NLCs from a laboratory to a commercial scale can be difficult and expensive.
(vii) Potential toxicity:
If given in large quantities or if they build up in the body over time, certain lipid components employed in NLCs may have harmful effects. Conducting comprehensive toxicity tests and carefully choosing biocompatible lipids are crucial.
(viii) Limited drug payload:
The quantity of medication that can be put into the lipid matrix using NLCs may be restricted.
This restriction may be a barrier for medications that need high dosages. 26
NLCs' Zeta Potential Characterization
Zeta potential is the net charge on the distributed particles found in the medium of dispersion. Determining the zeta potential provides insight into the system's stability as well as the existence of aggregates that may compromise the formulation's stability. Surface charges make particles more stable because they electrostatically reject one another, thus preventing particle aggregation. Zeta potential measurement can be used to assess the stability of colloidal dispersions since it provides insight into the presence of aggregation as well as electrostatic repulsion between the particles. For physically stable preparations, a zeta potential value of 30 mv is taken into consideration.
The shape of particles
Transmission and Scanning Electron Microscopy (SEM) can be used to determine the morphology and form of nanoparticles.
TEM stands for electron microscopy. The particle size distribution is also established using these methods. Original morphology cannot be acquired in SEM and TEM for a variety of reasons, including the shrinking of nanocarriers brought on by dehydration, which alters the nanocarriers' structural makeup. To get rid of the restrictions related to SEM and TEM, a novel method that uses atomic force morphogenesis to provide three-dimensional nanocarrier formations.
Particle size
The size of the particles and their distribution are crucial factors in determining the formulation's physical stability. Laser diffraction and photon correlation spectroscopy are used to measure particle size. Zeta sizer can be used to detect particle size at the nanoscale. The kind and quantity of lipids and emulsifiers employed in NLCs might have an impact on particle size. In the case of NLCs as well, particle size reduces with increasing emulsifier content.
Efficiency of drug encapsulation
The ratio of the drug to the total weight of the particles is known as the entrapment efficiency. Efficiency of entrapment is sometimes referred to as medication payload capacity or encapsulation efficiency.
Determining the effectiveness of medication encapsulation is highly helpful since it provides insight into how well NLCs work as carriers.78 For the medicine to have high entrapment efficiency, it must be well soluble in lipids.
Crystallinity and polymorphism Both crystallinity and polymorphism can be determined by differential scanning calorimetry and X-ray diffractometry. These techniques give an idea about the melting and recrystallization pattern of solid lipids present in NLCs. The polymorphic transition of lipids also can be determined by using these methods. The crystal lattice structure of special crystalline compounds can be identified using wide angle X-ray diffractomatry.
Tension at the surface
Surface tension can be measured using a torsion balance, a Kibron device, or Wilhelmy plates. Torsion balance is the most common tool used to measure the surface tension of NLCs. Contact angle can also be used to calculate surface tension.2
Prospects for the Future The majority of studies on the utilization of NLCs as chemotherapeutic drug delivery systems have been on low molecular weight medications. As a result, their range of uses must be broadened to encompass high molecular weight medicines like proteins, peptides, and nucleic acids that are utilized to treat cancer. This might present a better chance to treat a larger range of malignancies.
Indeed, NLCs have been shown to be a new vector for lung cancer gene therapy . Another significant worry is the dearth of critical evaluation about the safety of NLCs as medication carriers. Actually, very few research discuss how safety concerns impact the use of NLCs. More in vivo research is therefore desperately needed to precisely identify the safety margins and characteristics that should be incorporated as design guidelines for NLCs. Lastly, additional research on NLCs' absorption, distribution, metabolism, and excretion as well as strategies to scale up their production and use in clinical trials in the near future could further explore their potential. The expected outcomes of these studies could offer a safer and more effective way to deliver chemotherapeutic agents.28
CONCLUSION
Lipids as a structural material combined with nanotechnology allows for NLCs. NLC is a flexible platform that can be used for a variety of drug delivery methods. Researchers from all around the world are interested in these nanocarriers. Compared to liposomes, nanoemulsions, and SLNs, partially crystallized lipidic nanocarriers exhibit superior drug loading and stability. These nanocarriers have a high drug loading capacity and are safe, efficient, biodegradable, and biocompatible. These nanocarriers can also be used to target drugs, either actively or passively, to certain organs or malignant cells. NLC has demonstrated its value in the cosmetics sector, but its potential in the pharmaceutical sector has not yet been thoroughly investigated. Diseases that are challenging to treat, like cancer, infections, neurological conditions, localized medication delivery, and genetic disorders, can benefit from NLC in ways that traditional carrier systems cannot. To bring these intriguing nanocarriers from the lab to the market, more study is needed in this field. The toxicity profile of these nanocarriers must be assessed, and issues with nanomedicines including formulation and characterisation complexity must be resolved. These nanocarriers evolve slowly as a result of insufficient clinical research and data.
REFERENCES




Shivani Rathor, Dr. Tanu Bhargava, Dr. Praveen Khirwadkar, Dr. Kamlesh Dashora, Nanostructured Lipid Carriers: A Versatile Platform for Drug Delivery Across Therapeutic areas, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 12, 2733-2757. https://doi.org/10.5281/zenodo.17966504
 10.5281/zenodo.17966504
10.5281/zenodo.17966504