
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
1 School of Pharmaceutical Sciences, Jaipur National University, Jaipur, Rajasthan, India.
2 Department of Pharmacology, D.Y. Patil College of Pharmacy, Kadamwadi, Kolhapur 416003, Maharashtra, India.
3 Faculty of Pharmaceutical Sciences, Amrapali University, Haldwani, Uttarakhand, India.
4 Roorkee Business School of Pharmacy, Roorkee, Uttarakhand, India.
5 Om Sai Vindhya College of Pharmacy Mirzapur, India.
6 Surajmal College of Pharmacy, Surajmal University Kichha, Uttarakhand, India.
7 Burman Group of Institutions Roorkee, Haridwar, Uttarakhand, India.
8 Department of Pharmacology, G.S.R.M Memorial College of Pharmacy, Lucknow, India.
Natural products have been a necessary starting point in the discovery of anticancer drugs, providing a array of clinically approved agents and providing structurally diverse molecules with the ability to regulate a variety of hallmarks of cancer. This is a review of natural product-derived compounds with anticancer properties with key examples of alkaloids, flavonoids, terpenoids, polyphenols, saponins, glycosides, and other special phytochemicals. The compounds have strong antiprolliferative action due to their wide range of pharmacological actions, which include the induction of apoptosis by intrinsic and extrinsic apoptotic pathways, the inhibition of cell cycle progression, the inhibition of angiogenesis and the inhibition of metastasis and invasion. Besides, natural agents regulate the crucial oncogenic signaling pathways, including PI3K/Akt/mTOR, MAPK/ERK, Wnt/b-catenin, and NF-kB, and also respond to cancer stem cells, affect epigenetic changes, and oxidative stress reactions. Nevertheless, despite their therapeutic potential, there are a number of concerns around them, such as low bioavailability, lack of uniform standardization, lack of mechanistic understanding, and lack of clinical validation, which hinder their acceptance in the mainstream of oncology. The current limitations are being overcome by advances in nanotechnology, systems pharmacology, AI-based drug discovery, and synthetic biology and can be used to improve the clinical translatability of natural products. It can be concluded that natural product-derived compounds have high potential in developing into safe, effective, and accessible anticancer therapeutics by filling the current gaps and using emerging technologies. Their status, mechanistic understanding, limitations and future opportunities are presented in this review with a great importance to the shaping of next-generation cancer treatment plans.
Cancer is a highly dangerous issue in the global health of the 21st century, which is a combination of uncontrolled growth of cells, lack of cell death, and the onset of metastatic areas. In spite of tremendous scientific breakthroughs, cancer still carries a significant medical, economic and social burden in different countries [1]. The World Health Organization (WHO) reports that cancer contributes to almost 1 out of every six death cases in the world with an estimated 20 million new cancer cases and 10 million deaths being reported every year. It is estimated that the incidence may increase alarmingly in future, due to the ageing of population, environment, lifestyle changes, and exposure to carcinogens [2]. The increasing risk factors and poor access to healthcare services as well as insufficient early detection initiatives subject low- and middle-income countries to disproportional impact. More so, the genetic, epigenetic, and tumor microenvironment differences make cancer heterogeneous, which makes it difficult to design universal therapeutic modalities [3]. Cancer staging is a systematic term to explain the size and the level of cancer in the body. It indicates the extent of primary tumor, the regional lymph nodes involvement and the existence or absence of distant metastasis, thus, giving vital information in diagnosis, treatment planning and prognosis. Stage 0 or carcinoma in situ is a stage where the abnormal or malignant cells are limited to the origninating tissue without extending into the adjacent structures. Stage I is an early stage of cancer where the tumor size is small and localized without any sign of lymph node metastasis or metastasis to other distant areas. Stage II represents a more developed situation where the tumor is bigger and can include the lymph nodes nearby yet the disease is still not diffused [4]. Stage III refers to locally advanced cancer that has a great tumor growth and spread to local lymph nodes or the neighbor tissues. The most developed stage is stage IV, which is characterized by spread of cancer cells to other body parts, including liver, lungs, bones, or the brain, a condition referred to as metastasis Fig.1. Precise staging is also key in the process of choosing the right therapeutic plans and estimating the hospital outcomes of the patient as well as ensuring effective communication between healthcare professionals and researchers [5].
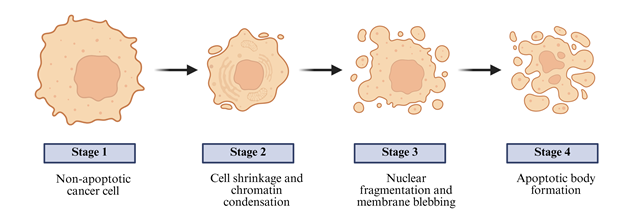
Fig.1: Stages of Cancer
The existing anticancer treatments such as surgery, radiotherapy, and chemotherapy, immunotherapy, and targeted molecular agents, have greatly enhanced the survival of most of the malignancies [6]. Nonetheless, there are significant shortcomings. Traditional chemotherapeutic drugs are not always selective which causes systemic toxicity and adverse effects which seriously impair the quality of life of a patient. Resistance to drugs, whether inherent or acquired, is a major obstacle to effective treatment, which is promoted by genetic mutations, epigenetic re-arrangement, an over expression of efflux transports and is shielded by the tumor microenvironment [7-10]. The various cancers like pancreatic, glioblastoma, triple-negative breast cancer, and metastatic melanoma are still showing poor survival rates despite aggressive treatment. Furthermore, it is expensive and restricts access to modern treatments including monoclonal antibodies, CAR-T cells, and tyrosine kinase inhibitors especially in resource-restricted environment. Multi-drug resistance and tumor relapse is an issue that has highlighted the necessity of new therapeutic molecules with enhanced efficacy and safety and defined mechanisms of action. Moreover, the activation of redundant signaling pathways is also frequently observed in cancer cells, which makes single-target therapies ineffective. It is this complexity that has led to a renewed interest in multi-target natural molecules that could be used to control multiple oncogenic networks at the same time [11].
Overall, natural products have been a foundation of drug discovery in the past. Over 60 percent of the approved anticancer drugs today have a natural origin either directly or indirectly through plants, microbes and marine organisms. Well-known examples are paclitaxel of Taxus brevifolia, vincristine and vinblastine of Catharanthus roseus, camptothein analogs of Camptotheca acuminata, the anthracyclines of Streptomyces species, and trabectedin of marine tunicates. These products have turned the treatment of cancer upside down with their distinct chemical frameworks, their powerful bioactivity and capability to engage various cellular targets [12]. Natural products have their natural benefits over synthetic molecules. Their structural heterogeneity, evolutionary perfection, and biological motivating characteristic allow them to interact with intricate cellular pathways that modulate cell cycle progression, apoptosis, angiogenesis, metastasis and immune reactions [13-15]. A wide variety of natural compounds are multi-target modulators, and have synergistic or pleiotropic actions capable of overcoming resistance-related mechanisms. They are also useful as lead molecules to be semi-synthetically optimized to increase potency, decrease toxicity, increase or decrease pharmacokinetics. The renewed interest in natural compounds has been driven by recent improvements in the methods of their extraction, in metabolomics, in high-throughput screening, and in computational methods, including molecular docking and network pharmacology. The developments have increased the speed with which new bioactive metabolites are identified and allowed greater understanding of how they act in pharmacology and their mechanisms. Natural product-based therapies also contribute to patient preference of therapies that are considered safer or more holistic although they should be scientifically validated [16].
The current review is intended to give a detailed and up to date review of the compounds that are derived out of natural products in cancer therapy with a focus on their pharmacological action and their clinical use [17]. The review features major categories of natural anticancer agents such as alkaloids, flavonoids, terpenoids, polyphenols and other specialized metabolites, their molecular mechanism of actions and role in modern oncology Fig.2. It is an overview of the existing evidence that they play a role in targeting the apoptosis pathways, cell cycle dynamic regulation, metastasis inhibition, and immune response and tumor microenvironment elements modulation. Also, the review examines synergistic advantages of the integration of natural compounds with either conventional chemotherapeutic or immunotherapeutic regimens, which could improve efficacy, decrease toxicity, and eliminate drug resistance. Nanotechnology developments in enhancing the bioavailability, solubility of natural products and targeted delivery are also addressed, as this technology is important in overcoming the conventional formulation problems [18-20].
Fig.2: Natural anticancer agents
The scope also goes to the assessment of preclinical and clinical trial data to determine the potential of translationality, safety, and pharmacokinetic constraints. The review suggests gaps in research that are present such as challenges relating to standardization, reproducibility, complete toxicological assessment. Lastly, the perspectives on future are provided in terms of natural compounds integrated into personalized cancer therapy, artificial intelligence used in naturally finding drugs, and even designing innovative multi-target therapy. Altogether, this review supports the ongoing role of natural products in cancer therapeutics development and demonstrates that they can be used to overcome unmet clinical needs through novel mechanisms, improved safety, and enhanced therapeutic efficacy [21].
Classification of Natural Product Derived Anticancer Agents
Natural products constitute a huge and chemically diverse pool of bioactive compounds which have played a key role in the discovery and development of cancer drugs. They are essential sources of anticancer agents due to their structural complexity, biological selectivity and multimodal activities. The natural product-derived anticancer compounds may be broadly classified into plant-derived compounds, marine-derived compounds, microbial metabolites, nutraceuticals and dietary phytochemicals, and semi-synthetic derivatives of natural scaffolds. All the classes have different pharmacodynamic and pharmacokinetic properties, which allow them to regulate various molecular pathways in carcinogenesis, tumor progression, angiogenesis, metastasis and immune evasion. The next section gives a detailed summary of these types and their clinical interest in current oncology [22].
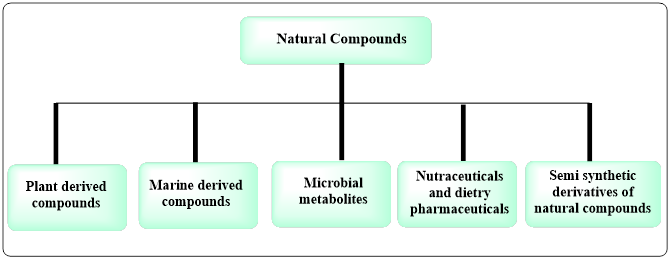
Fig.3: Classification of natural product derived anticancer agents
Plant-derived compounds
The most historically relevant and the most extensively studied natural source of anticancer molecules are plants. They can interact with a variety of cellular targets with impressive specificity because of their ability to synthesize structurally complex secondary metabolites, including alkaloids, terpenoids, flavonoids, phenolics, lignans, tannins and glycosides. A number of clinical anticancer agents of plant origin are still used in the multinational chemotherapeutic regimens. Common examples are the vinca alkaloids (vincristine, vinblastine) of Catharanthus roseus, which prevent the polymerization of micro tubules and causes mitotic arrest; the epipodophyllotoxins (etoposide, teniposide) of Podophyllum peltatum, which inhibit topoisomerase II and causes cell damage; and the taxanes (paclitaxel, docetaxel) of Taxus species, which stabilises micro tubules and blocks cell cycle progression at the G2/M phase In Camptotheca acuminata, camptothecin derived important topoisomerase I inhibitors including irinotecan and topotecan which have been extensively applied in treatment of colorectal and ovarian cancers. In addition to these established agents, many phytochemicals, including curcumin, resveratrol, quercetin, berberine, epigallocatechin gallate, and artemisinin derivatives, have shown strong anticancer activity in various mechanisms, such as induction of apoptosis, regulation of oxidative stress, inhibition of NF-kB, PI3K/Akt/mTOR and MAPK signaling, angiogenesis inhibition and reversal of drug resistance. The compound of plant derivatives usually has poly-pharmacological properties, and thus, multicellular activity allows them to attack cancer cells with minimal harm to normal tissue. The fact that they are relatively cheap and accepted by the culture also promotes continued research in order to exploit their therapeutic effect [23].
Marine-derived compounds
Marine environments are source of one of the most diverse, but least investigated reservoirs of natural bioactive molecules. Algae, sponges, tunicates, mollusks, bacteria, fungi, and soft corals are marine organisms which form unique chemical entities through evolutionary forces like extreme habitat conditions, competition, and symbiosis. Such molecules frequently have infrequent chemical scaffolds that do not exist in Earth-based life, and thus are good drug discovery targets [24]. Anticancer drugs derived in the sea have been met with growing clinical interests. Isolated Trabectedin acts as an anticancer by altering the transcription and the tumor microenvironment since it binds to the minor groove of DNA. Eribulin, a synthetic analog of the marine macrolide halichondrin B of Halichondria okadai, inhibits microtubule dynamics, and is used to treat metastatic breast cancer and liposarcoma [25]. It is an antibody-drug conjugate of a cytotoxic marine peptide known as monomethyl auristatin E that is used in the treatment of Hodgkin lymphoma and anaplastic large cell lymphoma and is named as breastuximab vedotin. A range of marine toxins including bryostatins, apratoxins, dolastatins, and didemnin analogues are highly effective anticancer agents with their activity being mediated by protein kinase c modulation, cyclin-dependent kinase, protein synthesis, cytoskeleton dynamic, and pro-apoptotic actions [26]. Marine natural products have structural novelty which enables them to circumvent resistance by conventional chemotherapeutic agents. The development of marine biotechnology and aquaculture has facilitated sustainable harvesting, elucidation of biosynthetic pathways and production of analogs with enhanced therapeutic indices [27].
Microbial metabolites
Microorganisms such as bacteria, fungi, actinomycetes and soil microbes are abundant sources of bioactive secondary metabolites with potent anticancer properties. This is due to their capacity to assemble chemically heterogeneous molecules because of complex biosynthetic gene clusters including polyketide synthases and nonribosomal peptide synthetases. A large number of anticancer agents used on the frontline have their source in microorganisms. Drugs of the anthracyclines category, including doxorubicin and daunorubicin, which are manufactured by Streptomyces species, show strong cytotoxic properties due to their ability to intercalate DNA, prevent topoisomerase II, and produce reactive oxygen species [28]. Another product of Streptomyces, bleomycin, causes spoilages of DNA strands and is commonly used to treat lymphoma. Mitomycin C is a cross-linking drug which is also used to treat bladder cancer and it has been produced by Streptomyces caespitosus. L-asparaginase is a derogative protein of Escherichia coli and Erwinia chrysanthemi that is specifically utilized up on asparine and is fundamental in managing acute lymphoblastic leukemia [29]. Microbial metabolites comprise also of the very powerful cytotoxins like the epothilones which stabilize the microtubules and prevent inactivation in taxane-resistant cancers. Ixabepilone is a semi-synthetic derivative of epothilone that is approved clinically in metastatic breast cancer. The endophytically produced fungal metabolites include paclitaxel, fusaric acid, gliotoxin, and cladosporin that have newly-emerging anticancer properties. Microbial sources also provide immunomodulatory drugs such as rapamycin and its analogs (everolimus, temsirolimus) which are mTOR-inhibitors and have applications in renal cell carcinoma and other malignancies. Continued identification of microorganisms that have never been identified before due to metagenomics, soil mining and extremophile exploration is still going on, which has led to the identification of new anticancer metabolites with distinct therapeutic properties [30].
Nutraceuticals and dietary phytochemicals
The category of natural anticancer agent based on nutraceuticals and dietary phytochemicals becomes an increasingly significant aspect of treatment and management of cancer because of their availability, safety, cultural acceptability and possibility of a long-term chemoprevention effect. These substances are very common in fruits, vegetables, spices, cereals, nuts, and medicinal herbs and help reduce the risk of cancer by regulating the oxidative stress, inflammation, cell proliferation, and immune reactions [31]. Polyphenols that are prominent examples like flavonoids (quercetin, kaempferol, rutin), stilbenes (resveratrol) and phenolic acid (gallic acid, ferulic acid) have strong antioxidant and anti-inflammatory properties. Some of the best studied dietary agents with proven capacity to suppress oncogenic signaling pathways, induce apoptosis, activate tumor suppressor genes, and inhibit metastasis include curcumin of turmeric, EGCG of green tea, lycopene of tomatoes, sulforaphane of cruciferous vegetables and allicin of garlic. Omega-3 fatty acids are anti-inflammatory modulators that occur in fish oil, and have anti-angiogenic activity [32]. Probiotics and prebiotics also affect the risk of cancer through controlling the gut microbiota and immune homeostasis. Nutraceutical compounds have the ability to interact with conventional therapies to improve their efficacy and minimize adverse effects. Their multi-targeted activity renders them sources of value to integrate cancer therapy and chemoprevention plans. Their variable bioavailability, metabolism and clinical potency, however, require sophisticated formulation approaches, including nanoencapsulation, lipid-based carriers and conjugation technologies to maximize therapeutic effects [33-35].
Semi-Synthetic Derivatives of Natural Compounds
Semi synthetic derivatives are very important in linking the natural molecules and modern medicinal chemistry. Despite their strong biological activity, most natural compounds have weaknesses including low solubility, unfavorable pharmacokinetics, toxicity or instability. Semi-synthesis allows chemical functional group modification of natural scaffolds to enhance therapeutic properties and maintain biological functionality. There are a number of semi-synthetic derivatives of clinically important anticancer drugs [36]. The camptothein analogs irinotecan and topotecan also aimed to solve solubility and stability problems of the parent molecule, and are currently commonly used in colorectal and cervical cancers. Paclitaxel analogs like cabazitaxel and docetaxel exhibit superior activity, solubility and efficacy in tumor resistance. To overcome multidrug resistance, the epothilone analogue ixabepilone was synthesized to maintain the microtubule-stabilizing activity [37]. The semi-synthetic anthracyclines include epirubicin and idarubicin, which are safer than their natural counterparts, especially with regard to cardiotoxicity. Artificial alteration of podophyllotoxin resulted in the creation of etoposide and teniposide, which are the key elements of combination chemotherapy. Synthetic biology, combinatorial chemistry, and structure-activity relationship (SAR) studies have made possible the creation of derivative compounds with increased target selectivity, desirability pharmacokinetics, enhanced drug-target interaction and low toxicity. Semi-synthesis is also sensitive to the issue of sustainability in the sense that, it reduces the extensive use of natural resources to provide them [38-40].
Phytochemicals in Cancer Therapy
The multitude of bioactive compounds known as phytochemicals generated by plants are the focus of natural product-based therapies against cancer cancers because of their complex structures and ability to regulate numerous cellular pathways that can lead to cancer. The compounds interact with signaling pathways that regulate cell cycle progression, apoptosis, angiogenesis, inflammation, metastasis, oxidative stress and epigenetic regulation [41]. They represent indispensable leads to anticancer drug discovery due to their multimodal activity, which is frequently minimally systemic toxic. Other classes of phytochemicals that have demonstrated anticancer activity include alkaloid, flavonoid, terpenoid, polyphenol, saponin, glycoside, and other structurally distinct molecules, e.g. coumarin, lignan and xanthone. Plant secondary metabolites have given rise to many clinically approved chemotherapeutics, including paclitaxel, etoposide, vincristine, and topotecan, which makes them highly relevant to the field of oncology [42-45].
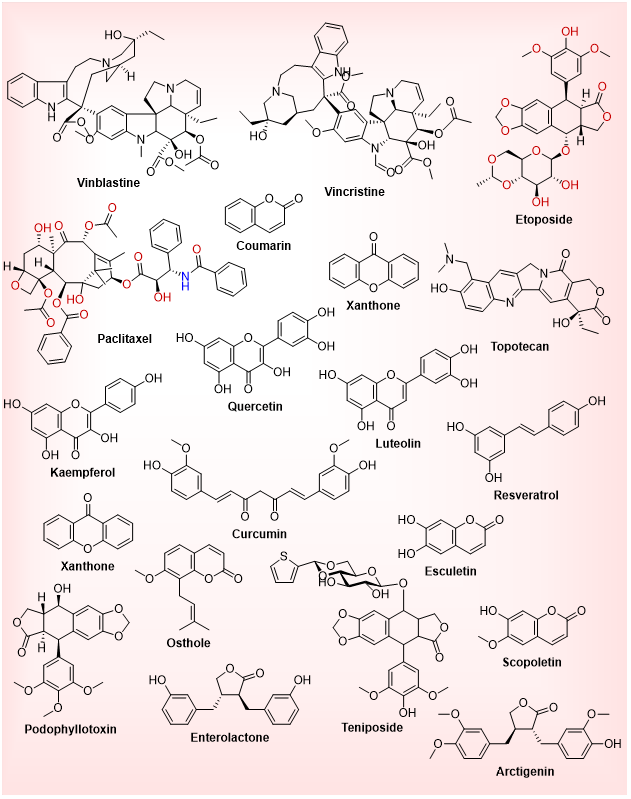
Fig.4: Phytochemicals in cancer therapy
Alkaloids
Alkaloids are heterocyclic compounds that are formed out of nitrogen and they are characterized by strong biological activity as well as wide clinical applications in the treatment of cancer. The vic part of Catharanthus roseus are used in the production of vincristine, vinblastine, vinorelbine, and vindesine which are all vinca alkaloids. These chemicals interfere with the formation of microtubules by binding to tubulin leading to mitotic arrest and apoptosis. Vinblastine and Vinristine are still important agents of chemotherapy in leukemias, lymphomas, breast cancer and sarcomas [46]. The discovery of Camptotheca acuminata as a source of Camptothein as a topoisomerase I inhibitor ushered in a novel generation of topoisomerase I inhibitors. Its semi-syntic analogs, irinotecan and topotecan, circumvent the issues of solubility and toxicity of the parent molecule and are in common use in colorectal, ovarian and cervical cancers. The benzylisoquinoline alkaloids, which were noscapine, berberine, are another important alkaloid group, which show anti-proliferative activity by inducing apoptosis, inhibiting NF-kB, altering microtubule dynamics, and blocking angiogenesis. Besides, the homoharringtonine produced by Cephalotaxus species is approved as omacetaxine and is a protein synthesis inhibitor with some activity against tyrosine kinase-resistant chronic myeloid leukemia. Alkaloids are highly cytotoxic with distinct molecular actions, upon which contemporary therapeutic approaches to cancer are based, and against which they serve to direct the next generation of drugs [47-50].
Flavonoids
Flavonoids are polyphenolic compounds that are highly abundant in fruits, vegetables, tea, and medicinal plants and they have a multimodal anti-cancer effect associated with their antioxidant, anti-inflammatory, and signaling-modulatory properties. One of the most investigated flavonoids is quercetin, which performs apoptosis through mitochondrial pathways, prevents PI3K/Akt and MAPK signaling, suppresses anti-apoptotic proteins, and sensitizes tumor cells to chemotherapy and radiotherapy [51]. It has anti-metastatic action, which is explained by the inhibition of epithelial-mesenchymal transition and matrix metalloproteinases. Kaempferol has a strong antiprolliferative effect by disrupting cell cycle regulators (cyclin D1 and CDK4), altering p53, angiogenesis, and oxidative stress. It also disrupts the growth of tumors by altering EGFR, NF-kB and Wnt/b-catenin signaling. The flavone luteolin (celery, parsley and chamomile) exhibits effective antiprolliferative effects on tumor cells by inhibiting JAK/STAT and Akt signaling, causing ROS-mediated apoptosis, and inhibiting metastasis by regulating integrin and chemokine signaling [52]. Flavonoids mostly serve as multi-target molecules with the ability to modulate redox homeostasis, DNA repair, inflammation, and tumor-microenvironment relationships. Their low toxicity and dietary high concentration favors their use as chemopreventive agents and adjuvants to increase the effectiveness of conventional anticancer agents [53].
Terpenoids
Terpenoids (based on the units of isoprene) form one of the most extensive families of plant metabolites which has significant potential of anticancer. Taxol (paclitaxel), which is a diterpenoid extracted in Taxus brevifolia has transformed the world of cancer treatment by stabilizing microtubules, inhibiting depolymerization and arresting G2/M cell cycles [54]. It cannot be done without in managing breast, ovarian, lung, and pancreatic cancers. Its semi-synthetic counterpart docetaxel but with better solubility and efficacy is acceptable. Artemisinin and its analogs, which were initially used as antimalarial compounds obtained out of Artemisia annua have also shown important anticancer properties [55]. They mediate their activity with endoperoxide bridge cleavage in the presence of intracellular iron to produce cytotoxic free radicals that cause apoptosis, autophagy, ferroptosis, and DNA damage. They also prevent angiogenesis and metastasis and sensitize tumors to chemotherapy. Other attractive terpenoids are limonene, ursolic acid, betulinic acid and andrographolide and each has distinct anticancer mechanisms of PI3K/Akt modulation, STAT3 inhibitor, VEGF signaling, and apoptotic pathways. Terpenoids have good pharmacological properties and structural diversity, thus they are good leads in the development of new anticancer drugs and nanotechnology systems of drug delivery [56-60].
Polyphenols
Polyphenols are a general group of antioxidants which are present in spices, fruits, and pharmaceutical plants and have been known to have strong anticancer, anti-inflammatory, and chemopreventive properties. Curcumin as the major curcuminoid of Curcuma longa is an anticancer agent that can alter the activity of various cellular pathways such as NF-kB, Akt, STAT3, COX-2 and mTOR [61]. It triggers apoptosis through caspases activation, lowers Bcl-2 protein, and produces mitochondrial impairment. Curcumin also prevents angiogenesis, metastasis and cancerous stem cell growth and increases sensibility to chemotherapy agents and radiations. Resveratrol, the stilbene present in grapes, berries, and peanuts, controls the process of apoptosis by activating p53, regulating SIRT1 and blocking the development of mitochondrial dysfunction. It also disrupts the important oncogenic signaling pathways like Wnt/b-catenin, PI3K/Akt, and NF-kB and inhibits the metastatic events by lowering EMT and activity of matrix metalloproteinases. The two compounds also have some issues with bioavailability, which have led to the establishment of nanoformulations, phospholipid complex, and synthetic analogs to enhance clinical applicability. The capacity of polyphenols to act on a broad variety of molecular pathways simultaneously makes them useful in integrative chemotherapy and prevention of cancer [62-65].
Saponins
Saponins are steroidal or glycosylated triterpenoids that occur in the broadest variety of medicinal and edible herbs. They show high anticancer effects due to their membrane-permeabilizing effects, and capability to cause ROS production and apoptotic pathways modulation. Saponins including ginsenosides, dioscin, and saikosaponins have been shown to have the ability to inhibit proliferation, induce mitochondrial depolarization, activate caspase cascades, and arrest the cell cycle [66]. Most saponins inhibit tumor angiogenesis by suppressing VEGF and inhibiting metastasis by suppressing EMT and matrix remodelling enzymes. Others, are also immunomodulatory, boosting the action of NK cells and raising the production of cytokines, thus, adding to anti-tumor immunity. Moreover, saponins may be used to sensitize cancer cells to chemotherapy, inhibit efflux pumps, diminish drug resistance mechanisms, and enhance intracellular drug accumulation. Their amphiphilic nature enables them to be incorporated in new drug delivery systems to enable targeted cancer therapy with enhanced bioavailability [67-70].
Glycosides
Glycosides are made of a sugar molecule that is conjugated to a bioactive aglycone which contributes to a set of diverse anticancer effects that involve cytotoxicity, cellular metabolism, and signal transduction pathway interference. The cardiac glycosids like digoxin, ouabain, digitoxin, and the like have attracted interests due to their anticancer effect in areas outside the heart. Their actions cause downstream inhibition of NF-kB signaling, induction of apoptosis, and impairment of calcium homeostasis by inhibiting Na+/K- ATPase [71]. PI3K/Akt signaling inhibitors Anthraquinone glycosides such as emodin and aloe-emodin cause ROS-dependent apoptosis and prevent tumor invasion through EMT inhibition. Iridoid glycosides including catalpol and aucubin have anticancer effects through the regulation of oxidative stress, the suppression of inflammatory cytokines and metastasis. Flavonoid glycoside, such as rutin and hesperidin, have antioxidant and cytoprotective properties, as well as proliferation and angiogenesis inhibitor activities. Glycosylation is also relevant in drug delivery since glycosylation may be used to increase solubility, targeting, and pharmacokinetics, and glycosylation-modified compounds are useful in future anticancer therapies [72].
Others
Other classes of phytochemicals such as xanthones, coumarins and lignans have also shown a lot of anticancer effects. Xanthonesa-mangostin and gambogic acid are highly cytotoxic, and their action is based on the mitochondrial dysfunction, activation of caspase, and inactivation of NF-kB and STAT3. They also block angiogenesis and metastasis through the regulation of VEGF, MMPs, and integrins. Coumarins are anti-proliferative and anti-angiogenic compounds and are found in plants like Angelica, Ferula and Citrus species [73]. Esculetin, scopoletin and osthole are compounds that suppress tumor proliferation by modulating cell cycle proteins, triggering the production of ROS, inhibiting the Akt/mTOR pathway and blocking metastasis. Lignans, including podophyllotoxin, enterolactone and arctigenin, cause anticancer effects through the regulation of hormonal metabolism, induction of apoptosis and inhibition of topoisomerase II. Etoposide and teniposide are anticancer drugs that have been identified as clinically significant and their key pharmacologic ingredient is podophyllotoxin. A number of lignans have estrogen-modulating effect, and are especially applicable in the breast, ovarian, and prostate cancers. These phytochemicals have structurally diverse scaffolds to facilitate exchanges with diverse cellular targets and this potential has seen them have a repertoire of therapeutic uses.
Pharmacological Mechanisms of Natural Anticancer Agents
Natural product derived anticancer agents exercise their therapeutic actions in a wide range of molecular and cellular activities that control tumor formation, progression, metastasis and therapeutic resistance. The natural compounds have a wide-spectrum anticancer activity that is less toxic as compared to conventional chemotherapeutic agents, which usually target only one molecular pathway. They are the most useful drug development and adjuvant therapy candidates due to their ability to regulate the apoptotic process, cellular division, angiogenesis, metastasis, oxidative stress, immune reaction, and epigenetics. Significant classes of natural products such as alkaloids, polyphenols, flavonoids, terpenoids and saponins are able to show extraordinary potential in reprogramming cancer cell behaviors by triggering tumor suppressor pathways, silencing oncogenes, and destabilizing the tumor microenvironment. The next section expounds on the major pharmacological mechanisms by which natural compounds act as anticancer agents [74].
Induction of Apoptosis
The process of natural anticancer agents is through apoptosis or programmed cell death whereby the normal tissues are spared and the malignant cells are destroyed. Cancer cells often avoid apoptosis through modulation of intrinsic and extrinsic pathways, which allows them to proliferate out of control and resist treatment. Natural products re-establish apoptotic sensitivity by acting on major regulators of these pathways which results in caspases activation, mitochondrial impairment, fragmentation of DNA and cell death. The pro-survival response regulating the intrinsic pathway is mitochondrial integrity and is determined by the ratio of pro-apoptotic (Bax, Bak, Bad) and anti-apoptotic (Bcl-2, Bcl-xL, Mcl-1) proteins. Natural compounds, including curcumin, berberine, quercetin, resveratrol, and betulinic acid, catalyze the intrinsic pathway by altering this balance in support of pro-apoptotic signals. They cause mitochondrial outer membrane permeabilization (MOMP), release of cytochrome c and caspase-9 and subsequently executioner caspases (caspase-3 and -7). Polyphenols EGCG and luteolin increase mitochondrial ROS production, disrupting the membrane potential, and initiating Bax translocation. A terpenoid such as artemisinin derivatives amplify iron-dependent oxidative stress leading to mitochondrial swelling and apoptosis. Alkaloids like camptothecin and vincristine cause the damage of DNA, which triggers mitochondrial apoptosis that involves p53. All of these together indicate intrinsic pathway as one of the primary targets of natural anticancer agents [75]. Extrinsic pathway occurs through the action of death receptors on the cell surface which include Fas, TRAIL-R1/DR4, TRAIL-R2/DR5 and TNF receptors. The natural products sensitize the cancer cells to extrinsic apoptosis through the increase of the death receptors, the ligand binding affinity, and the caspase-8 activation. It has been demonstrated that curcumin, resveratrol and saponins can enhance the FasL and TRAIL expression, thereby supporting the death receptor-mediated apoptosis. Moreover, some flavonoids prevent FLIP, which is a negative regulator of caspase-8, thus, promoting the formation of DISC (death-inducing signaling complex). Bryostatin and fucoidan are marine compounds that also activate the extrinsic pathway through regulation of immune-mediated apoptotic signalling. It is this combined action on intrinsic and extrinsic pathways that enable natural molecules to work especially well in the circumventing of apoptosis resistance- a characteristic of cancer progression [76].
Cell cycle arrest
Cancer cells display uncontrolled proliferation due to dysregulation of cell cycle checkpoints. Natural anticancer agents exert anti-proliferative effects by regulating cyclins, cyclin-dependent kinases (CDKs), and checkpoint proteins such as p21, p27, and p53. By disrupting the orderly progression of the cell cycle, natural products effectively suppress tumor growth.
G0/G1 phase inhibition
Numerous natural substances decrease the G0/G1 transition with the down-regulation of cyclin D, cyclin E, CDK2, CDK4 and CDK6 and the up-regulation of p21 and p27. The better-known G0/G1 inhibitors include flavonoids (quercetin, kaempferol, apigenin, etc.), that prevent the initial stage of the cell cycle and decrease multiplication. Curcumin and resveratrol regulate the expression of p53, inhibiting the cyclin D and up-regulating the CDK inhibitor transcription. The alkaloids such as berberine prevent the phosphorylation of retinoblastoma (Rb) thus inhibiting the release of E2F transcription factors that allows S-phase entry. By such measures, the natural products are successful in preventing the proliferation of cancer cells at the initial level of the cell cycle [77].
S-phase modulation
The natural compounds disrupt the DNA synthesis, replication fork advancement, and the metabolism of nucleotides, thus preventing the advancement of S-phase. Topoisomerase I is inhibited with camptothecin derivatives, leading to broken DNA strands and S-phase arrest. The curcumin interferes with the DNA polymerase process and reproduction apparatus, and the saponins cripple the nucleotide production. EGCG and resveratrol prevent DNA methyltransferase activity, which changes chromatin structure and has an indirect impact on DNA replication. Natural products cause S-phase arrest through the interference with DNA integrity and replication control, which is why they play a role in the cytotoxicity of fast-growing cancer cells [78].
G2/M blockade
G2/M arrest inhibits the entry of mitosis in the cell, which is a critical phase to the cancer cell. Paclitaxel is a diterpenoid which stabilizes microtubules and inhibits mitotic spindle formation resulting in G2/M arrest and apoptosis. Other terpenoids like derivatives of artemisinin also suppress the mitotic action of cyclin B1/CDK1. Flavonoid luteolin and genistein disruption of tubulin polymerization, interference with Aurora kinases and Plk1, which are important mitotic progression regulators. Alkaloids such as vincristine and vinblastine cause destabilization of the microtubules inducing metaphase arrest. These mechanisms enable the natural compounds to have great anti-proliferative properties by attacking the cancer cells that divide rapidly selectively [79].
Inhibition of metastasis and invasion
The spread of the cancer cells to other body organs, known as metastasis, is considered to cause more than 90 percent of mortality related to cancers. Natural compounds suppress metastasis by attacking epithelial-mesenchymal transition (EMT), extracellular matrix degradation, cell motility, as well as tumor microenvironment interactions. Curcumin and resveratrol are polyphenols that inhibit EMT with the effect of downregulating Snail, Slug, Twist, and ZEB1, and upregulating E-cadherin. Flavonoids prevent the work of matrix metalloproteinases (MMP-2, MMP-9) which decrease the breakdown of the extracellular matrix and invasion. Terpenoids inhibit the expression of integrins and interrupt the rearrangement of the cyto skeleton that is required in motility. Saponins suppress FAK/Src signaling preventing the migration of cancer cells. Natural compounds inhibit metastasis too through a specific action on chemokine receptors (CXCR4), VEGF- mediated vascular permeability, and hypoxia-mediated pathways including HIF-1a. This multi-targeted control of metastasis makes natural products work as the promising agents of preventive treatment of tumor spread and recurrence [80].
Anti-angiogenic mechanisms
The growth and spread of tumors require angiogenesis, a process where new blood vessels are generated by the body using the existing ones. Natural compounds have the anti-angiogenic effects related to proliferation of endothelium, its migration, and tube formation. Curcumin, resveratrol, and EGCG prevent VEGF expression and inhibit VEGFR2 phosphorylation that prevents downstream PI3K/Akt and MAPK signaling required for endothelial cell survival. Terpenoids ursolic acid and andrographolide prevent angiogenesis by suppressing HIF-1a, MMPs and fibroblast growth factors. The fucoidan molecules derived in the sea prevent angiogenesis through the inhibition of the binding of heparan sulfate proteoglycans that bind VEGF. Saponins block endothelial nitric oxide synthase (eNOS) to decrease vascular permeability and angiogenic reactions. Natural agents interfere with angiogenic signaling which deprives tumors of oxygen and nutrients, retarding the development of disease [81].
Modulation of signaling pathways
Natural compounds exert profound anticancer effects by modulating intracellular signaling pathways crucial for survival, proliferation, metabolism, and metastasis. Their ability to regulate multiple signaling cascades simultaneously distinguishes them from single-target drugs.
PI3K/Akt/mTOR pathway
Activation of this pathway is common in cancer that fosters cancer growth and survival, angiogenesis, and resistance to therapy. Natural products interfere with PI3K activation, decrease Akt phosphorylation, and inhibit mTOR signaling, which are curcumin, resveratrol, quercetin, and berberine. The result is a reduction in protein synthesis, the induction of autophagy, apoptosis, as well as the reduction of metastasis. This pathway is also downregulated by the terpenoids such as betulinic acid and ginsenosides. PI3K/Akt/mTOR inhibition increases cancer cells to chemotherapy and radiotherapy, and supports the use of natural compounds as a combination therapy [82].
MAPK/ERK pathway
MAPK/ERK pathway controls the proliferation, differentiation, and stress responses. The polyphenols and flavonoids regulate MEK and ERK phosphorylation resulting in the arrest of cell cycle and apoptosis. Curcumin regulates JNK and p 38 MAPK stimulation, facilitating apoptotic pathways. ERK-regulated transcription factors like c-Myc and AP-1 are inhibited as well by alkaloids like berberine. Inhibition of MAPK lowers the proliferation of tumor cells and inhibits the malignant transformation [83].
Wnt/β-catenin pathway
Wnt signaling is aberrantly activated to promote proliferation, stemness, EMT and metastasis. Natural substances such as resveratrol, luteolin, and EGCG block b-catenin nuclear translocation, down-regulate wnt ligands and induce b-catenin degradation through activation of GSK-3b. This leads to decreased activity of cancer stem cell, reduced EMT and inhibition of metastatic behavior.
NF-κB pathway
NF-kB controls inflammation, resistance to apoptosis, angiogenesis and metastasis. Its sustained expression is a characteristic of a lot of cancers. Curcumin, resveratrol, quercetin, and other saponins prevent the nuclear translocation of NF-kB by inhibiting the IkB kinase (IKK). This lowers the transcription of the genes that are related to survival (Bcl-2, survivin), inflammation (COX-2, TNF-a, IL-6) and metastasis (MMP-9, VEGF). NF-kB inhibition is useful in overturning drug resistance, tumor progression, and tumor sensitization to chemotherapy [84].
Targeting cancer stem cells
CSCs are a special population that initiates, resists, recurs and metastasize tumors. Natural compounds attack CSCs through disabling self-renewal pathways that include Wnt/b-catenin, Notch, hedgehog, and STAT3. Curcumin decreases CSC markers, such as CD44, ALDH1, and Nanog, whereas resveratrol decreases the expression of Oct-4 and Sox2. Flavonoids prevent sphere formation and CSC proliferation by preventing Notch and Hedgehog pathways. Ginsenosides and artemisinin derivatives are terpenoids that inhibit CSC survival by regulating the production of ROS and EMT. CSCs are the primary focus of achieving long-term cancer remission, and natural compounds are excellent to prevent relapse and treatment resistance [85].
Epigenetic regulation
Epigenetic changes such as DNA methylation, histone modification and chromatin remodeling are crucial in cancer progression. Epigenetic processes are controlled by natural compounds to inhibit DNMTs (DNA methyltransferases), HDACs (histone deacetylases), and HATs (histone acetyltransferases). Polyphenals including EGCG block DNMT1 which causes demethylation and reactivation of tumor suppressor genes. Curcumin and resveratrol regulate histone acetylation due to HDACs inhibition and HATs activity enhancement and reinstate normal gene expression patterns. Genistein changes chromatin structure by reversing methylation aberrations in hormone-dependent cancers. These are epigenetic influences which have the effect of inducing apoptosis, reduced proliferation and metastasis suppression. Notably, natural products are non-toxic epigenetic modulators having capabilities of personalized cancer treatment [86].
Oxidative Stress Modulation and ROS Generation
Reactive oxygen species (ROS) have dual effects on cancer firstly, low levels promote cell proliferation, and secondly, excess ROS forms apoptosis. Dozen of natural products cause regulated oxidative stress in cancer cells, and they take advantage of the weakened antioxidant response in cancer cells [87]. Polyphenols include quercetin, resveratrol and curcumin, which control the production of ROS, which activates apoptosis through mitochondrial pathways. Artemisinin is a terpenoid that reacts with iron to form ROS, leading to the damage of DNA and ferroptosis. The effect of the saponins and alkaloids is amplifying the production of ROS by suppressing the action of antioxidant enzymes like glutathione peroxidase and catalase. Other natural products, on the other hand, are antioxidants, and they inhibit the development of cancer by cancelling the effect of free radicals as well as blocking the oxidative injury of DNA. This two-way ROS-modulating feature indicates the situation-dependent therapeutic excellence of natural compounds [88].
Challenges, Research Gaps, and Limitations
Although natural product-derived compounds hold enormous therapeutic potential in cancer management, they are limited to application in mainstream oncology by a number of scientific, technological, clinical, and regulatory concerns. Despite the promising anticancer effects of many phytochemicals and natural metabolites in cell culture and in animal models, very few of them reach the successful phase of clinical approval. This translational disconnect indicates bioavailability, pharmacokinetics, mechanistic, standardization, toxicity-profiling, and extensive-scale production limitations. The need to overcome these barriers is essential to making natural compounds productive as potent, safe and evidence-based anticancer agents. Poor bioavailability is one of the significant problems because it impacts most of the phytochemicals, such as curcumin, resveratrol, quercetin, EGCG, and most terpenoids. They tend to be insoluble in water, have low absorption, undergo rapid metabolism and have low systemic half-lives and as a result, they do not reach sufficient levels in the tumor sites. Several methods have been suggested to enhance pharmacokinetics such as encapsulation strategies, nanoformulations, liposomes, phospholipid complexes, and structural modification, although most are in preclinical phase and need to undergo the extensive clinical test run. Also, individual differences in metabolism can have an influence on the biological activity of natural substances, which makes it more complicated to optimize the dose and have an individual approach to treatment. The other major weakness is irregular quality, standardization and reproducibility of natural extracts. The plant compounds may be diverse depending on the geographical origin, mode of farming, season of harvesting, mode of extracting and storing. The slightest changes in phytochemical make-up may lead to dramatic changes in biological activity. Lack of internationally agreed standards of quality control may usually lead to variation between batch to batch, and so consistency in therapy becomes hard to maintain. Efforts to extract plant materials in a standardized way or to purify active ingredients are still hampered by the complexity of plant matrices and the possibility of synergistic interactions which can be lost in the purification process. The mechanistic knowledge about a large number of natural compounds is still lacking. Though many studies have shown anticancer efficacies, including apoptosis generation, angiogenesis inhibition, and signaling pathway regulation, comparatively few studies have provided an in-depth explanation of either molecular targets or pharmacodynamic networks. Cancer is extremely heterogeneous, whereby there is a combination of genetic and epigenetic aberrations; hence, the effect of natural compounds can be very different among cancer types and patients. Absence of mechanistic clarity also tends to interfere with rational drug design, optimization of therapeutic combinations and prediction of possible off-target interactions. Another challenge is toxicological profiling. Despite the common belief in natural compounds safety, some of them may lead to toxicity, interactions with drugs of herbs or organ-specific effects with therapeutic doses. As an example, a high level of saponins can interfere with cell membranes and certain alkaloids can have a neurotoxic or mutagenic effect. Moreover, natural products have the ability of altering the metabolism of chemotherapeutic agents by modulation of cytochrome P450 enzymes resulting in decreased effect or inhibited toxicity. Extensive toxicological research - such as long term exposure analysis, pharmacokinetic analysis and safety analysis are necessary but not always available.
Sustainability and supply are additional issues that limit the natural anticancer agent development pipeline. Most plants or marine life and microorganisms generating potent bioactive metabolites are rare, slow growing or ecologically sensitive. The harvesting done on large scale can harm the ecosystems, can disorganize the biodiversity, or can be ethically questionable. Semi-synthesis, total synthesis, plant tissue culture and microbial expression system are potential solutions but they need high levels of technological investment and optimization. The second issue is the complicated interaction between natural molecules and the tumor microenvironment. Although, various phytochemicals regulate the process of inflammation, immune response, stromal and oxidative stress, the dynamic nature of the TME makes it hard to predict responses. The pharmacological effects of natural compounds can be affected by cancer-associated fibroblasts, immune cells, hypoxic areas, and elements of the extracellular matrix. This complexity requires refined models, e.g. 3D organoids, co-culture systems and precision oncology platforms to gain insight into context-dependent responses. Lastly, natural anticancer agents cannot be commercially developed due to regulatory and intellectual property issues. Multicomponent extracts cannot be patented and this decreases the motivation to invest in pharmaceuticals. Moreover, there are different approval pathways across the world, and natural products might be classified as dietary supplement, as herbal medicine or pharmaceutical where each has different safety and efficacy data requirements. Such regulatory complexities delay the process of translation between laboratories and the clinic. Summarizing, even though natural products have enormous potential to promote cancer treatment, the best way to make the products achieve their full potential is to overcome huge obstacles in the form of bioavailability, mechanistic explanation, quality control, toxicology, sustainable sourcing and clinical validation. The only way that these research gaps can be bridged will be through interdisciplinary collaboration, high level of analytical technologies, new delivery systems and solid regulatory frameworks. By this kind of work, natural product-derived compounds can eventually be useful, readily available, and safer substitutes or supplements to traditional cancer therapies.
FUTURE PERSPECTIVES
The future of natural product-based cancer therapeutics is actually very bright as a result of the rapid increase in technology, better mechanistic insights as well as the increasing interest on the usage of multi-targeted therapeutic responses. The natural compounds with their structural diversity and pleiotropic biological activity provide incomparable source of safer and more effective anticancer strategies as cancer is a highly heterogeneous and adaptive disease. Nevertheless, to fully leverage their therapeutic capabilities, new approaches, interdisciplinary teams, and tactical partnership with contemporary drug development platforms will have to be introduced. The combination of novel drug delivery systems, especially nanotechnology-based preparations, is one of the most important future directions. Nanocarriers, including liposomes, polymeric nanoparticles, dendrimers and lipid-based vesicles, have been demonstrated to enhance the solubility, stability, bioavailability and targeted delivery of natural compounds, which have a poor pharmacokinetic profile. Nanoformulated curcumin, resveratrol, paclitaxel and artemisinin analogs exhibit increased therapeutic effect and lowered systemic toxicity in preclinical trials. Precision and therapeutic index can be further increased by the development of smart delivery systems, which in response to tumor-specific stimuli can be pH, enzymes, redox gradients, or temperature. The natural products research will also be transformed by the advancements in computational biology, artificial intelligence, and system pharmacology. Machine learning can be used for predicting drug-target interactions, optimization of molecular structure, and synergistic interactions with conventional treatments. Network pharmacology provides information about multi-target processes, so it is possible to discuss the identification of important signaling hubs that are controlled by phytochemicals. Molecular docking, virtual screening, and pharmacophore modeling with AI are supremely useful in the discovery of new bioactive molecules in large natural product collections. The technologies will be used to solve the historical difficulties in the natural product drug discovery, including the complexity of the structure and inability to elucidate the mechanism. The other significant direction of the future is the targeting of cancer stem cells and the tumor microenvironment. Numerous natural products have been shown to prevent CSC renewal, prevent EMT, and regulate the immune-inhibitory tumor environment. The combination therapies which prevent recurrence and metastasis and resistance can be developed in further exploration. Such compounds as curcumin, resveratrol, ginsenosides, and flavonoids demonstrate high potentials in shifting TME components, such as macrophages, fibroblasts towards an anti-tumor phenotype. There will also be more interest in the epigenetic modification of cancer. The promising results of DNMT or HDAC-inhibitors in natural compounds are a source of future safer epigenetic drugs. The future research must target epigenetic signature profiling to phytochemicals, which can be used to target epigenetic therapy with an individual tumor profile.
CONCLUSION
The natural product-based anticancer agents still remain one of the most useful and effective assets in the field of oncology, as they provide a treasury of structurally diverse compounds that can be used to drive various hallmarks of cancer. Contrary to the traditional chemotherapeutic drugs that often interact with a specific molecular process, natural compounds are pleiotropic in nature, influencing apoptosis, cell cycling, angiogenesis, metastasis, oxidative stress, inflammation, and epigenetics at the same time. It is especially beneficial in the context of treating the complexity, adaptability, and heterogeneity of cancer, which in most cases makes single-pathway blocking agents inadequate or susceptible to resistance. Years of preclinical studies indicate that alkaloids, flavonoids, terpenoids, polyphenols, saponins, glycosides, and other metabolites found in plants have potent anticancer effects with well-defined molecular targets. The basis of several successful chemotherapeutic agents, such as paclitaxel, vincristine, etoposide, and camptothecin derivatives, can be attributed to many of these compounds and are still key constituents of a current treatment regimen. At the same time, dietary phytochemicals such as curcumin, resveratrol, EGCG, and quercetin are still promising to be used as chemopreventive agents and adjuvant therapy due to their good safety and ability to be used in combination with conventional therapy. Although it has this potential, there are a number of limitations that prevent smooth translation of natural compounds into clinical oncology. Issues related to low bioavailability, rapid metabolism, inconsistent standardization of extract, lack of mechanistic understanding, and lack of large-scale clinical trials are still a major obstacle. Besides, sustainability concerns, toxicological loopholes, and regulatory challenges contribute to additional barriers on the wide-scale therapeutic usage. These drawbacks will have to be overcome by unified principle of extraction, increased delivery systems, strict pharmacokinetic and toxicological testing, and more robust clinical data. In the future, nanotechnology, systems pharmacology, synthetic biology, metabolic engineering, and AI-assisted drug discovery, are promising vastly to overcome these difficulties. These technologies will increase the accuracy, solubility, and clinical scalability of natural products and allow natural products to be produced at large scale. Further investigations of cancer stem cells, interactions between tumor microenvironment, and epigenetic control can also be used to find out new therapeutic strategies based on natural product pharmacology. To sum up, natural product-derived compounds will continue to play an indispensable role in the treatment of cancer. They have the potential to become mainstream, evidence-based anticancer therapeutics, complementary agents can become mainstream, improved standardization, and strategic scientific innovation can improve them to meet unmet clinical needs and enhance overall cancer treatments in the world.
REFERENCES








Vinayak Dasharath Gaikwad, Samruddhi S Patil, Navin C Pant, Subhash Chand, Chandresh Maurya, Harpreet Kaur, Vaseem Fateh, Kalpana Sonwani, Natural Product Derived Compounds in Cancer Therapy: Pharmacological Mechanisms and Clinical Applications, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 12, 4154-4177. https://doi.org/10.5281/zenodo.18098900
 10.5281/zenodo.18098900
10.5281/zenodo.18098900