
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Channabasweshwar Pharmacy College, Kava Road, Basweshwar Chowk, Latur- 413512
Glecaprevir is a direct acting antiviral agent and Hepatitis C virus (HCV) NS3/4A protease inhibitor that targets the viral RNA replication. In combination with Pibrentasvir, Glecaprevir is a useful therapy for patients who experienced therapeutic failure from other NS3/4A protease inhibitors. It demonstrates a high genetic barrier against resistance mutations of the virus. In cell cultures, the emergence of amino acid substitutions at NS3 resistance-associated positions A156 or D/Q168 in HCV genotype 1a, 2a or 3a replicons led to reduced susceptibility to Glecaprevir. Pibrentasvir is a direct acting antiviral agent and Hepatitis C virus (HCV) NS5A inhibitor that targets the the viral RNA replication and viron assembly. In combination with Glecaprevir, pibrentastiv is a useful therapy for patients who experienced therapeutic failure from other NS5A inhibitors. In cell cultures, the emergence of amino acid substitutions at known NS5A inhibitor resistance-associated positions in HCV genotype 1a, 2a or 3a replicons led to reduced susceptibility and resistance to pibrentasvir.
Glecaprevir is a direct acting antiviral agent and Hepatitis C virus (HCV) NS3/4A protease inhibitor that targets the the viral RNA replication. In combination with Pibrentasvir, Glecaprevir is a useful therapy for patients who experienced therapeutic failure from other NS3/4A protease inhibitors. It demonstrates a high genetic barrier against resistance mutations of the virus. In cell cultures, the emergence of amino acid substitutions at NS3 resistance-associated positions A156 or D/Q168 in HCV genotype 1a, 2a or 3a replicons led to reduced susceptibility to Glecaprevir. [1] The combinations of amino acid substitutions at NS3 position Y65H and D/Q168 also results in greater reductions in Glecaprevir susceptibility and NS3 Q80R in genotype 3a patients also leads to Glecaprevir resistance. Mavyret is also indicated for HCV genotype 1-infected patients who have been previously treated with regimens either containing an NS5A inhibitor or an NS3/4A protease inhibitor, but not both. [2,3]
Structure-
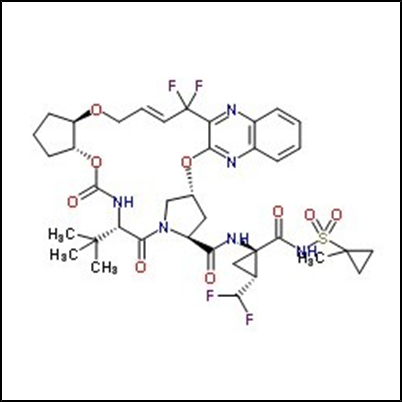
Fig. No. 1 Glecaprevir
Mechanism of action: Glecaprevir is an inhibitor of the HCV NS3/4A protease, which is a viral enzyme necessary for the proteolytic cleavage of the HCV encoded polyprotein into mature forms of the NS3, NS4A, NS4B, NS5A, and NS5B proteins. These multifunctional proteins, including NS3, are essential for viral replication. The N-terminal of NS3 protein confers serine protease activity, while the C-terminus of NS3 encodes a DExH/D-box RNA helicase which hydrolyzes NTP as an energy source to unwind double-stranded RNA in a 3′ to 5′ direction during replication of viral genomic RNA. [4,5] NS4A is a cofactor for NS3 that directs the localization of NS3 and modulates its enzymatic activities. Glecaprevir disrupts the intracellular processes of the viral life cycle through inhibiting the NS3/4A protease activity of cleaving downstream junctions of HCV polypeptide and proteolytic processing of mature structural proteins. [6,7]
Side Effects:
Difficulty in breathing; swelling of your face, lips, tongue, or throat, slow heartbeats, weak or shallow breathing; or low red blood cells--pale skin, weakness, rapid heart rate, trouble concentrating. Common side effects may include headache or feeling tired. [8]
Solubility- DMSO
Half-life- The elimination half-life (t1/2) is approximately 6 hours [9]
Pibrentasvir is a direct acting antiviral agent and Hepatitis C virus (HCV) NS5A inhibitor that targets the viral RNA replication and Viron assembly. In combination with Glecaprevir, Pibrentasvir is a useful therapy for patients who experienced therapeutic failure from other NS5A inhibitors. In cell cultures, the emergence of amino acid substitutions at known NS5A inhibitor resistance-associated positions in HCV genotype 1a, 2a or 3a replicons led to reduced susceptibility and resistance to Pibrentasvir. [10] These resistance-associated amino acid substitutions included Q30D/deletion, Y93D/H/N or H58D +Y93H in genotype 1a replicons, F28S + M31I or P29S + K30G in genotype 2a replicons, and Y93H in genotype 3a replicons. Individual NS5A amino acid substitutions that reduced susceptibility to pibrentasvir include M28G or Q30D in a genotype 1a replicon and P32-deletion in a genotype 1b replicon. [11] Pibrentasvir is available as an oral combination therapy with Glecaprevir under the brand name Mavyret. This fixed-dose combination therapy was FDA-approved in August 2017 to treat adults with chronic hepatitis C virus (HCV) genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis, including patients with moderate to severe kidney disease and those who are on dialysis. [12]
Structure-
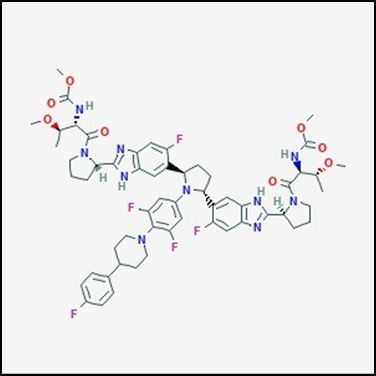
Fig. No. 2 Pibrentasvir
Mechanism of action: NS5A is a phosphoprotein that plays an essential role in replication, assembly and maturation of infectious viral proteins. The basal phosphorylated form of NS5A, which is maintained by C-terminal serine cluster, is key in ensuring its interaction with the viral capsid protein, or the core protein. By blocking this interaction, pibrentasvir inhibits the assembly of proteins and production of mature HCV particles. NS5A also interacts with viral and cellular proteins to form the HCV replicase complex, and supports the RNA replication of HCV. [13,14]
Side Effects- Diarrhea, headache, itching skin, lack or loss of strength, nausea, unusual tiredness or weakness.
Half-life: The elimination half-life (t1/2) is approximately 13 hours [15]
MATERIALS AND METHOD-
MATERIALS-
Glecaprevir and Pibrentasvir, Combination of Glecaprevir and Pibrentasvir (Mavyret) tablet dosage forms, distilled water, Acetonitrile, phosphate buffer, ammonium acetate buffer, glacial acitic acid, methanol, potassium dihydrogen phosphate buffer, tetra hydrofuran, tri ethyl amine, ortho-phosphoric acid etc.
METHODS-
Preparation of buffer:
0.01N Ammonium Formate buffer: Accurately weighed 0.66gm of Ammonium Formate in a 1000ml of Volumetric flask add about 900ml of milli-Q water added and degas to sonicate and add 1ml of OPA finally make up the volume with water then PH adjusted to 4.2 with dil. Orthophosphoric acid solution.
Standard Preparation:
Accurately Weighed and transferred 20mg of Glecaprevir and 8mg of Pibrentasvir working Standards into a 50ml clean dry volumetric flask, add 3/4th volume of diluent, sonicated for 5 minutes, and make up to the final volume with diluents. (400µg/ml of Glecaprevir and 160µg/ml Pibrentasvir)
Standard Working Preparation:
1ml from the above stock solution was taken into a 10ml volumetric flask and made up to 10ml. (40µg/ml of Glecaprevir and 16µg/ml Pibrentasvir)
Sample Preparation:
5 tablets were weighed ,1 tablet equivalent weight (100mg/40mg) powdered was transferred into a 250mL volumetric flask, 50mL of diluent added and sonicated for 25 min, further the volume made up with diluent and filtered. (400µg/ml of Glecaprevir and 160µg/ml Pibrentasvir) [16]
Sample Working Preparation:
From the filtered solution 1 ml was pippeted out into a 10 ml volumetric flask and made upto 10ml with diluent. (40µg/ml of Glecaprevir and 16µg/ml Pibrentasvir)
Validation: Validation of analytical procedures was performed for Glecaprevir and Pibrentasvir using the following parameters.
Specificity: Checking of the interference in the optimized method. We should not find interfering peaks in blank and placebo at retention times of these drugs in this method. So this method was said to be specific. [17]
Precision:
Sample Preparation:
5 tablets were weighed ,1 tablet equivalent weight(100mg/40mg) powdered was transferred into a 250mL volumetric flask, 50mL of diluent added and sonicated for 25 min, further the volume made up with diluent and filtered. (400µg/ml of Glecaprevir and 160µg/ml Pibrentasvir)
Sample Working Preparation:
From the filtered solution 1 ml was pippeted out into a 10 ml volumetric flask and made upto 10ml with diluent. (40µg/ml of Glecaprevir and 16µg/ml Pibrentasvir) [18]
Repetability-
The Repetability was determined by preparing Sample solutions of Glecaprevir (20ppm) and Pibrentasvir (16ppm) and the solutions were injected six times and the % RSD for the area of six standard injections results should not be more than 2%. [19]
Linearity:
Linearity solutions are prepared such that 0.25, 0.5, 0.75, 1, 1.25, 1.5ml from the Stock solutions of Glecaprevir and Pibrentasvir are taken in to 6 different volumetric flasks and diluted to 10ml with diluents to get 10ppm, 20ppm, 30ppm, 40ppm, 50ppm, 60ppm of Glecaprevir, and 4ppm, 8ppm, 12ppm, 16ppm,20ppm, 24ppm of Pibrentasvir. [20]
Standard Preparation:
Accurately Weighed and transferred 20mg of Glecaprevir and 8mg of Pibrentasvir working Standards into a 50ml clean dry volumetric flask, add 3/4th volume of diluent, sonicated for 5 minutes, and make up to the final volume with diluents. (400µg/ml of Glecaprevir and 160µg/ml Pibrentasvir) [21]
Accuracy:
Sample Preparation:
5 tablets were weighed ,1 tablet equivalent weight(100mg/40mg) powdered was transferred into a 250mL volumetric flask, 50mL of diluent added and sonicated for 25 min, further the volume made up with diluent and filtered. (400µg/ml of Glecaprevir and 160µg/ml Pibrentasvir)
Standard Working Preparation:
1ml from the above stock solution was taken into a 10ml volumetric flask and made up to 10ml. (40µg/ml of Glecaprevir and 16µg/ml Pibrentasvir) [22]
Robustness:
Flow minus (1.0ml/min), Flow plus (1.2ml/min), mobile phase minus, mobile phase plus, temperature minus (25°C) and temperature plus(35°C) was maintained and samples were injected in duplicate manner. System suitability parameters were not much effected and all the parameters were passed. %RSD was within the limit. [23]
LOD sample Preparation:
0.25ml each from two standard stock solutions was pipetted out and transferred to two separate 10ml volumetric flasks and made up with diluents. From the above solutions 0.3ml each of Glecaprevir and Pibrentasvir, solutions respectively were transferred to 10ml volumetric flasks and made up with the same diluents
LOQ sample Preparation:
0.25ml each from two standard stock solutions was pipetted out and transferred to two separate 10ml volumetric flask and made up with diluent. From the above solutions 0.9ml each of Glecaprevir and Pibrentasvir, solutions respectively were transferred to 10ml volumetric flasks and made up with the same diluent. [24]
Degradation studies:
Oxidation:
To 1 ml of stock solution of Glecaprevir and Pibrentasvir, 1 ml of 20% hydrogen peroxide (H2O2)was added separately. The solutions were kept for 30 min at 600c. For HPLC study, the result and solution was diluted to obtain 20µg/ ml & 16µg/ml solution and 10µl were injected in to the system and the chromatograms were recorded to assess the stability of sample.
Acid Degradation Studies:
To 1 ml of stock solution Glecaprevir and Pibrentasvir, 1 ml of 2N Hydrochloric acid was added and refluxed for 30mins at 600c.The result ant solution was diluted to obtain 20µg/ml & 16µg/ml solution and 10µl solutions were injected into the system and the chromatograms wererat corded to assess the stability of sample.
Alkali Degradation Studies:
To 1 ml of stock solution Glecaprevir and Pibrentasvir, 1 ml of 2N sodium hydroxide was added and refluxed for 30mins at 600c. There solution was diluted to obtain 20µg/ml&16µg/ml solution and 10µl were injected into the system and the chromatograms were recorded to assess the stability of sample.
Dry Heat Degradation Studies:
The standard drug solution was placed in oven at 105°C for 6h to study dry heat degradation. For HPLC study, the resultant solution was diluted to 20µg/ml&16µg/ml solution and10µl were injected into the system and the chromatograms wererat corded to assess the stability of the sample.
Photo Stability studies:
The photochemical stability of the drug was also studied by exposing the 400µg/ml&&160µg/ml solution to UV Light by keeping the beaker in UV Chamber for 1days or 200-Watt hours/m2 in photo stability chamber. For HPLC study, the resultant solution was diluted to obtain 20µg/ml&16µg/ml solutions and 10µl were injected into the system and the chromatograms were recorded to assess the stability of sample.
Neutral Degradation Studies:
Stress testing under neutral conditions was studied by refluxing the drug in water for 1 h r s at a temperature of 60ºC. For HPLC study, the resultant solution was diluted to 20µg/ml&16µg/ml solution and 10µl were injected in to the system and the chroma to grams wererat corded to assess the stability of the sample. [25,26]
RESULTS AND DISCUSSION
Method development: Method development was done by changing various, mobile phase ratios, buffers etc.
Optimized method:
Chromatographic conditions:
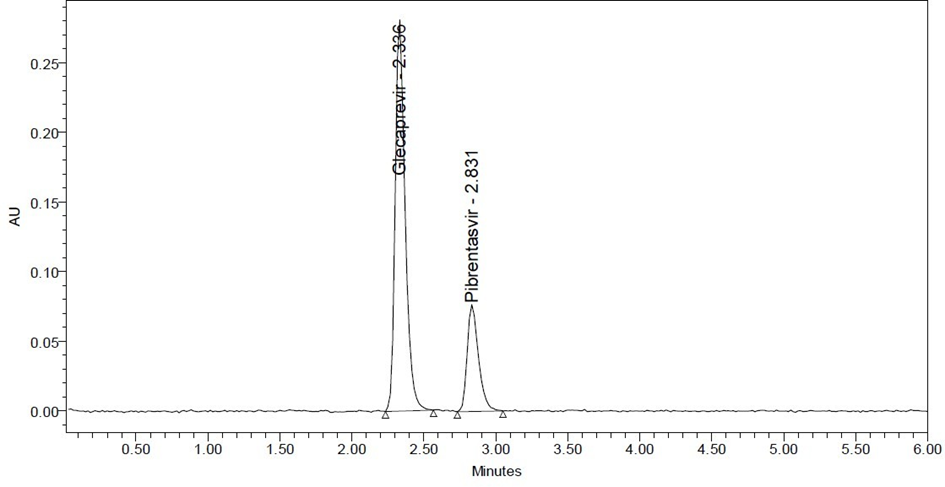
Fig. No. 3 Chromatogram
System suitability: All the system suitability parameters were within the range and satisfactory as per ICH guidelines
Table:6.1 System suitability parameters for Glecaprevir and Pibrentasvir
|
Sr. No. |
Glecaprevir |
Pibrentasvir |
|||||
|
Inj |
RT(min) |
USP Plate Count |
Tailing |
RT(min) |
USP Plate Count |
Tailing |
USP Resolution |
|
1 |
2.319 |
5736 |
1.27 |
2.830 |
6348 |
1.32 |
3.8 |
|
2 |
2.319 |
5733 |
1.28 |
2.833 |
6243 |
1.28 |
3.8 |
|
3 |
2.319 |
5503 |
1.28 |
2.834 |
6165 |
1.28 |
3.8 |
|
4 |
2.325 |
5540 |
1.31 |
2.838 |
6500 |
1.36 |
3.8 |
|
5 |
2.325 |
5528 |
1.31 |
2.838 |
6442 |
1.35 |
3.7 |
|
6 |
2.329 |
5509 |
1.28 |
2.840 |
6435 |
1.30 |
3.7 |
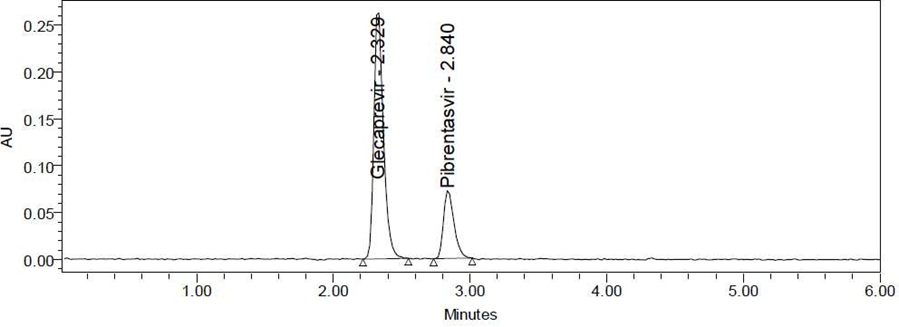
Fig. No. 4 System suitability Chromatogram
Specificity:
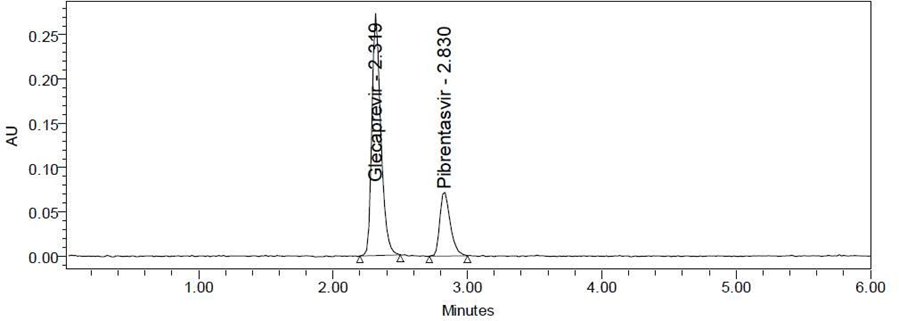
Fig. No. 5 Chromatogram of Specificity
Linearity:
Table 6.2 Linearity table for Glecaprevir and Pibrentasvir.
|
Glecaprevir |
Pibrentasvir |
||
|
Conc. (μg/mL) |
Peak area |
Conc. (μg/mL) |
Peak area |
|
0 |
0 |
0 |
0 |
|
10 |
316003 |
4 |
93507 |
|
20 |
604701 |
8 |
178990 |
|
30 |
905871 |
12 |
264691 |
|
40 |
1214419 |
16 |
354203 |
|
50 |
1512848 |
20 |
444145 |
|
60 |
1788020 |
24 |
533073 |
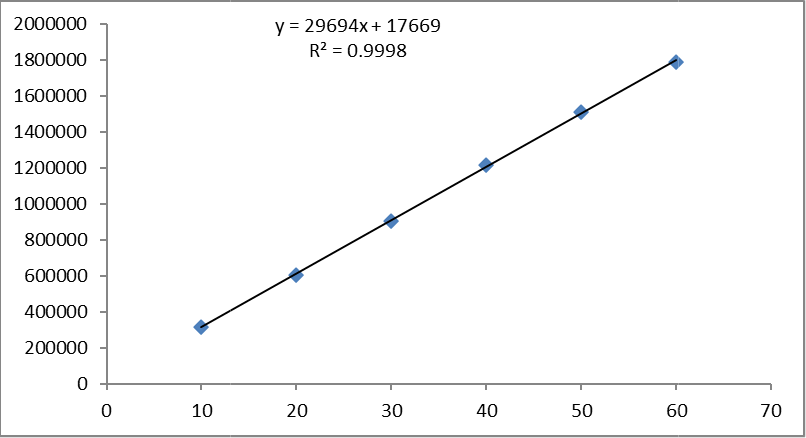
Fig No. 6 Calibration curve of Glecaprevir
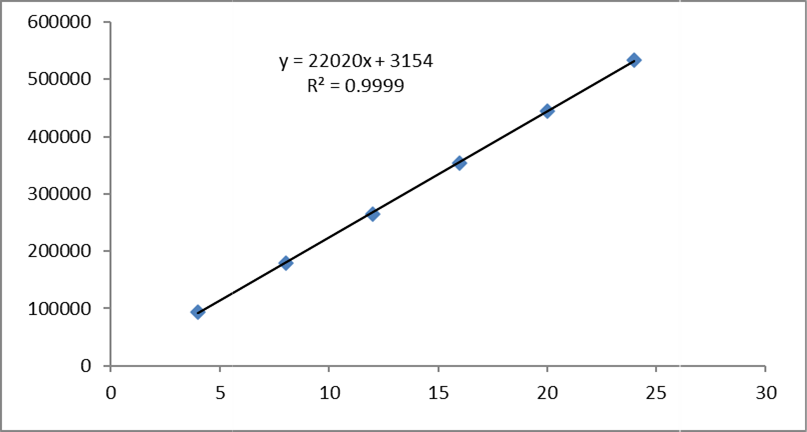
Fig No. 7 Calibration curve of Pibrentasvir
Precision:
System Precision:
Table 6.3 System precision table of Glecaprevir and Pibrentasvir
|
Sr. No. |
Area of Glecaprevir |
Area of Pibrentasvir |
|
1. |
1272446 |
351901 |
|
2. |
1264155 |
356085 |
|
3. |
1280709 |
350354 |
|
4. |
1274175 |
353291 |
|
5. |
1272807 |
359668 |
|
6. |
1277556 |
354469 |
|
Mean |
1273641 |
354295 |
|
S. D |
5615.6 |
3297.7 |
|
%RSD |
0.4 |
0.9 |
Repeatability:
Table 6.4 Repeatability table of Glecaprevir and Pibrentasvir
|
S. No |
Area of Glecaprevir |
Area of Pibrentasvir |
|
1. |
1291028 |
357532 |
|
2. |
1285991 |
350816 |
|
3. |
1277226 |
352495 |
|
4. |
1270937 |
352835 |
|
5. |
1285462 |
352134 |
|
6. |
1288698 |
355537 |
|
Mean |
1283224 |
353558 |
|
S. D |
7622.1 |
2486.3 |
|
%RSD |
0.6 |
0.7 |
Intermediate precision (Day_Day Precision):
Table 6.5 Intermediate precision table of Glecaprevir and Pibrentasvir
|
Sr. No |
Area of Glecaprevir |
Area of Pibrentasvir |
|
1. |
1124371 |
344899 |
|
2. |
1130669 |
345057 |
|
3. |
1137355 |
341716 |
|
4. |
1131084 |
343478 |
|
5. |
1123641 |
347010 |
|
6. |
1135778 |
347907 |
|
Mean |
1130483 |
345011 |
|
S. D |
5653.8 |
2262.9 |
|
%RSD |
0.5 |
0.7 |
Accuracy:
Table 6.6 Accuracy table of Glecaprevir
|
% Level |
Amount Spiked (μg/mL) |
Amount recovered (μg/mL) |
% Recovery |
Mean % Recovery |
|
50% |
20 |
20.01 |
100.06 |
100.43% |
|
20 |
19.97 |
99.83 |
||
|
20 |
20.00 |
100.01 |
||
|
100% |
40 |
40.54 |
101.34 |
|
|
40 |
40.67 |
101.67 |
||
|
40 |
40.30 |
100.76 |
||
|
150% |
60 |
59.97 |
99.95 |
|
|
60 |
60.23 |
100.39 |
||
|
60 |
59.92 |
99.87 |
Table 6.7 Accuracy table of Pibrentasvir
|
% Level |
Amount Spiked (μg/mL) |
Amount recovered (μg/mL) |
% Recovery |
Mean % Recovery |
|
50% |
8 |
8.09 |
101.15 |
101.17% |
|
8 |
8.14 |
101.78 |
||
|
8 |
8.12 |
101.53 |
||
|
100% |
16 |
16.12 |
100.78 |
|
|
16 |
16.21 |
101.29 |
||
|
16 |
16.26 |
101.66 |
||
|
150% |
24 |
24.29 |
101.21 |
|
|
24 |
24.25 |
101.06 |
||
|
24 |
24.01 |
100.06 |
Sensitivity:
Table 6.8 Sensitivity table of Glecaprevir and Pibrentasvir
|
Molecule |
LOD |
LOQ |
|
Glecaprevir |
0.28 |
0.85 |
|
Pibrentasvir |
0.22 |
0.67 |
Robustness:
Table 6.9 Robustness data for Glecaprevir and Pibrentasvir.
|
Sr. no. |
Condition |
%RSD of Glecaprevir |
%RSD of Pibrentasvir |
|
1 |
Flow rate (-) 1.0ml/min |
0.5 |
0.4 |
|
2 |
Flow rate (+) 1.2ml/min |
0.9 |
0.9 |
|
3 |
Mobile phase (-) 64B:36A |
0.3 |
1.1 |
|
4 |
Mobile phase (+) 73B:27A |
0.3 |
0.6 |
|
5 |
Temperature (-) 25°C |
0.9 |
0.4 |
|
6 |
Temperature (+) 35°C |
0.9 |
1.0 |
Degradation Studies:
Degradation studies were performed with the formulation and the degraded samples were injected. Assay of the injected samples was calculated and all the samples passed the limits of degradation.
Table 6.10 Degradation Data of Glecaprevir
|
Sr. No. |
Degradation Condition |
% Drug Degraded |
Purity Angle |
Purity Threshold |
|
1 |
Acid |
7.06 |
0.150 |
1.032 |
|
2 |
Alkali |
5.45 |
0.496 |
7.096 |
|
3 |
Oxidation |
4.40 |
0.701 |
0.782 |
|
4 |
Thermal |
2.17 |
0.153 |
0.276 |
|
5 |
UV |
1.30 |
0.845 |
0.816 |
|
6 |
Water |
1.30 |
0.759 |
0.797 |
Table 6.11 Degradation Data of Pibrentasvir
|
Sr. No. |
Degradation Condition |
% Drug Degraded |
Purity Angle |
Purity Threshold |
|
1 |
Acid |
7.65 |
0.533 |
1.013 |
|
2 |
Alkali |
6.44 |
0.092 |
0.581 |
|
3 |
Oxidation |
4.20 |
0.103 |
0.916 |
|
4 |
Thermal |
2.99 |
0.588 |
0.832 |
|
5 |
UV |
1.38 |
0.699 |
0.794 |
|
6 |
Water |
0.88 |
0.601 |
0.819 |
SUMMARY AND CONCLUSION-
|
Parameters |
Glecaprevir |
Pibrentasvir |
LIMIT |
|
|
Linearity Range(µg/ml) |
10-40µg/ml |
10-40µg/ml |
R< 1 |
|
|
Regression coefficient |
0.999 |
0.999 |
||
|
Slope(m) |
29694 |
22020 |
||
|
Intercept(c) |
17669 |
3154 |
||
|
Regression equation (Y= mx+c) |
y = 29694x + 17669 |
y = 22020x + 3154 |
||
|
Assay (% mean assay) |
100.55% |
99.39% |
90-110% |
|
|
Specificity |
Specific |
Specific |
No interference of any peak |
|
|
System precision %RSD |
0.4 |
0.9 |
NMT 2.0% |
|
|
Method precision %RSD |
0.6 |
0.7 |
NMT 2.0% |
|
|
Accuracy % recovery |
100.43% |
101.17% |
98-102% |
|
|
LOD |
0.28 |
0.22 |
NMT 3 |
|
|
LOQ |
0.85 |
0.67 |
NMT 10 |
|
|
Robustness |
FM |
0.5 |
0.4 |
%RSD NMT 2.0 |
|
|
MM |
0.3 |
1.1 |
|
|
|
MP |
0.3 |
0.6 |
|
|
|
TM |
0.9 |
0.4 |
|
|
|
TP |
0.9 |
1.0 |
|
CONCLUSION
Retention time of Glecaprevir and Pibrentasvir were found to be 2.336 min and 2.831 min. %RSD of the Glecaprevir and Pibrentasvir were and found to be 0.6and 0.7respectively. %Recovery was obtained as 99.85% and 100.82% for Glecaprevir and Pibrentasvir respectively. LOD, LOQ values obtained from regression equations of Glecaprevir and Pibrentasvir were 0.28, 0.85 and 0.22,0.67respectively. Regression equation of Glecaprevir is y = 29694x + 17669, y = 22020x+ 3154.of Pibrentasvir. Retention times were decreased and that run time was decreased, so the method developed was simple and economical that can be adopted in regular Quality control test.
REFERENCES





Satyam Apare, Wale Risha, Naragude Amol, Tanuja Patil, Development And Validation of Stability Indicating RP-HPLC Methods For Related Substances and Assay Analyses of Glecaprevir & Pibrentasvir Mixtures, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 7, 2327-2337. https://doi.org/10.5281/zenodo.15979049
 10.5281/zenodo.15979049
10.5281/zenodo.15979049