
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmaceutics, LCIT School of Pharmacy, Bilaspur, Chhattisgarh, India.
The present study focused on the formulation and evaluation of a microemulsion-based gel for the enhanced transdermal delivery of Trimebutine maleate hydrochloride (TPHCl). Microemulsions were prepared using an optimized combination of oil, surfactant, and co-surfactant, and were converted into gel using Carbopol® 934P. The optimized microemulsion exhibited a globule size of 52.37 nm, polydispersity index of 0.273, and zeta potential of ?19.62 mV, indicating uniformity and stability. The microemulsion gel exhibited pseudoplastic rheological behavior and showed improved Spreadability and viscosity suitable for topical application. In vitro drug release studies revealed that the microemulsion gel released 92.46% of the drug within 8 hours, significantly higher compared to 65.43% from the plain gel. Drug permeation was enhanced by increasing water content and reducing oil and Smix concentrations. FTIR analysis confirmed no drug–excipient interactions. A strong Level A IVIVC was established with a correlation coefficient (R²) of 0.9731, suggesting a reliable predictive relationship between in vitro release and in vivo absorption. Stability studies conducted over 3 months showed that formulations stored at 4 ± 2°C remained more stable than those at 40 ± 2°C/75 ± 5% RH, with only minimal changes in physicochemical parameters like particle size, zeta potential, drug content, and viscosity. In conclusion, the developed microemulsion gel demonstrated excellent physical stability, enhanced drug release, and favorable characteristics, making it a promising vehicle for the transdermal delivery of TPHCl.
A "Noval Drug Delivery System" is any of a number of sophisticated physicochemical techniques intended to optimize the therapeutic potential of pharmacologically active substances by controlling their release and distribution inside cells, tissues, and organs. Noval drug delivery system focuses on the formulation and administration techniques that effectively transport drugs in order to maximize treatment effectiveness and reduce side effects. The development of effective drug delivery systems remains one of the foremost challenges in pharmaceutical sciences. Among various routes of administration, the transdermal drug delivery system (TDDS) offers several advantages over conventional methods such as oral or injectable routes. TDDS ensures controlled and sustained drug delivery, avoids first-pass metabolism, improves patient compliance, and minimizes side effects. However, the stratum corneum, the outermost layer of the skin, poses a significant barrier to drug permeation, limiting the effectiveness of transdermal formulations for many therapeutic agents. To overcome this barrier, novel drug delivery systems such as microemulsions have gained considerable interest. Microemulsions are thermodynamically stable, isotropic mixtures of oil, water, surfactant, and co-surfactant that can enhance the solubility and permeability of drugs across biological membranes. Their small droplet size (typically less than 100 nm) and large surface area facilitate increased drug penetration through the skin. Microemulsions also improve drug loading capacity, physical stability, and bioavailability, making them a promising carrier system for transdermal delivery of both hydrophilic and lipophilic drugs. In this context, the present study aims to develop and evaluate a microemulsion gel-based transdermal delivery system for TPHCl using suitable oils, surfactants, and co-surfactants selected based on their solubilizing potential and compatibility. The microemulsion formulations were prepared using the pseudo-ternary phase diagram approach, followed by incorporation into a gel matrix using Carbopol® 934P, which enhances the formulation’s viscosity and spreadability while maintaining its structural integrity. The physicochemical characterization of the developed microemulsion and microemulsion gel formulations included evaluation of globule size, polydispersity index (PDI), zeta potential, drug content, percentage transmittance, pH, and viscosity. In vitro drug release studies were carried out to assess the release kinetics of the optimized formulations. The results of the study revealed that the optimized microemulsion gel exhibited pseudoplastic rheological behaviour, enhanced spreadability, and significantly improved drug release compared to the plain gel. The formulation was found to be stable at 4?±?2°C, indicating that lower temperatures favour the stability of the microemulsion system. Furthermore, a strong in vitro–in vivo correlation (IVIVC) was established, indicating that the in vitro release profile could reliably predict the in vivo performance of the formulation, thus aiding in rational formulation development. Therefore, this microemulsion-based gel system holds substantial promise as a novel transdermal platform for the effective delivery of TPHCl, contributing to the advancement of patient-centric therapeutic strategies. The route of drug administration significantly influences the onset, extent, and duration of therapeutic action, as well as the distribution of the drug within the body. While oral administration remains the most common and preferred route due to its convenience and patient compliance, it suffers from drawbacks such as enzymatic degradation in the gastrointestinal tract and extensive first-pass metabolism, which can reduce bioavailability. Transdermal drug delivery offers an attractive alternative by bypassing the gastrointestinal tract and hepatic first-pass effect, providing controlled and sustained systemic drug delivery with improved patient compliance. Tapentadol Hydrochloride (TPHCl) is a centrally acting analgesic indicated for moderate to severe acute and chronic pain. However, its short half-life (~4 hours) necessitates frequent dosing, which can reduce patient adherence. Developing a transdermal delivery system for TPHCl could maintain steady plasma levels over an extended period, reduce dosing frequency, and minimize systemic side effects. The primary aim of this research is to develop and optimize a transdermal microemulsion gel formulation of Tapentadol Hydrochloride that can provide sustained drug release over 24 hours, ensuring effective systemic absorption and improved therapeutic efficacy. The successful development of a TPHClmicroemulsion gel has the potential to improve pain management by offering a controlled, sustained, and non-invasive drug delivery system that enhances patient compliance and therapeutic outcomes.
Transdermal drug delivery systems (TDDS) offer several advantages over conventional routes of administration, including:
Despite these benefits, the skin’s structure presents a significant barrier to transdermal drug delivery. The primary obstacle is the stratum corneum, the outermost layer of the epidermis, composed of corneocytes embedded in a lipid matrix arranged as lamellar bilayers. This layer effectively prevents excessive water loss from the body and blocks the penetration of most topically applied drugs, except those that are both lipid-soluble and of low molecular weight. Consequently, transdermal administration of many drugs remains challenging. Pharmaceutical research has addressed these challenges by developing various passive and active technologies to enhance skin penetration. Passive techniques include the use of penetration enhancers, supersaturated systems, microemulsions, and vesicles, while active methods comprise iontophoresis, electroporation, phonophoresis, microneedles, jet injectors, suction ablation, and thermophoresis.
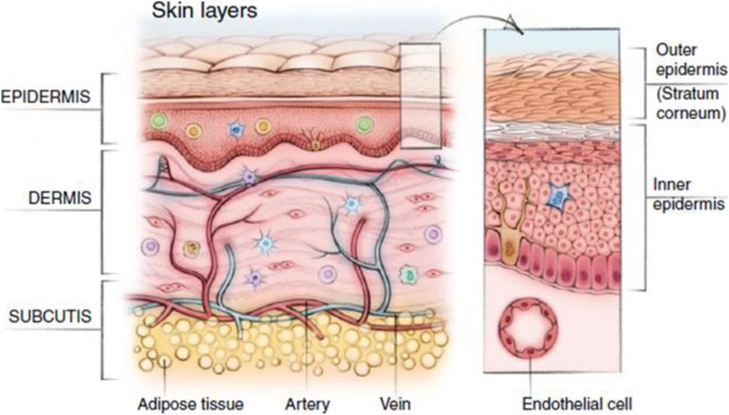
Figure 1: -Structure of Human Skin
Role of Microemulsions Transdermal Drug Delivery System
Microemulsions are promising systems for transdermal drug delivery due to their:
Multiple mechanisms contribute to the enhanced skin penetration observed with microemulsions. Small droplet size (<200 nm) and high surface area facilitate drug transport. Additionally, components such as surfactants and oils can disrupt the lipid organization of the stratum corneum or increase drug solubility within the skin layers, thereby improving partitioning. Studies have shown that specific oils like oleic acid alter lipid conformation, and certain surfactants increase uptake of components into the skin. Microemulsions also increase skin hydration, which further enhances permeability.
Tapentadol Hydrochloride was obtained as a gift sample from a Sigma Aldrich. Labrasol (Caprylocaproyl macrogol-8 glycerides) and Propylene Glycol Monocaprylate (Type I NF) were procured from commercial suppliers and were of analytical grade. All other excipients, solvents, and reagents used were of analytical or pharmaceutical grade and used without further purification. Double distilled water was used throughout the study.
Microemulsion Gel Preparation
The optimized microemulsion formulation was incorporated into a gel base to improve application and skin retention. Polymers such as carbomers or cellulose derivatives were used to impart suitable viscosity and stability to the gel. The gel matrix also facilitates sustained drug release and enhances patient compliance by providing a non-greasy, easy-to-apply vehicle.
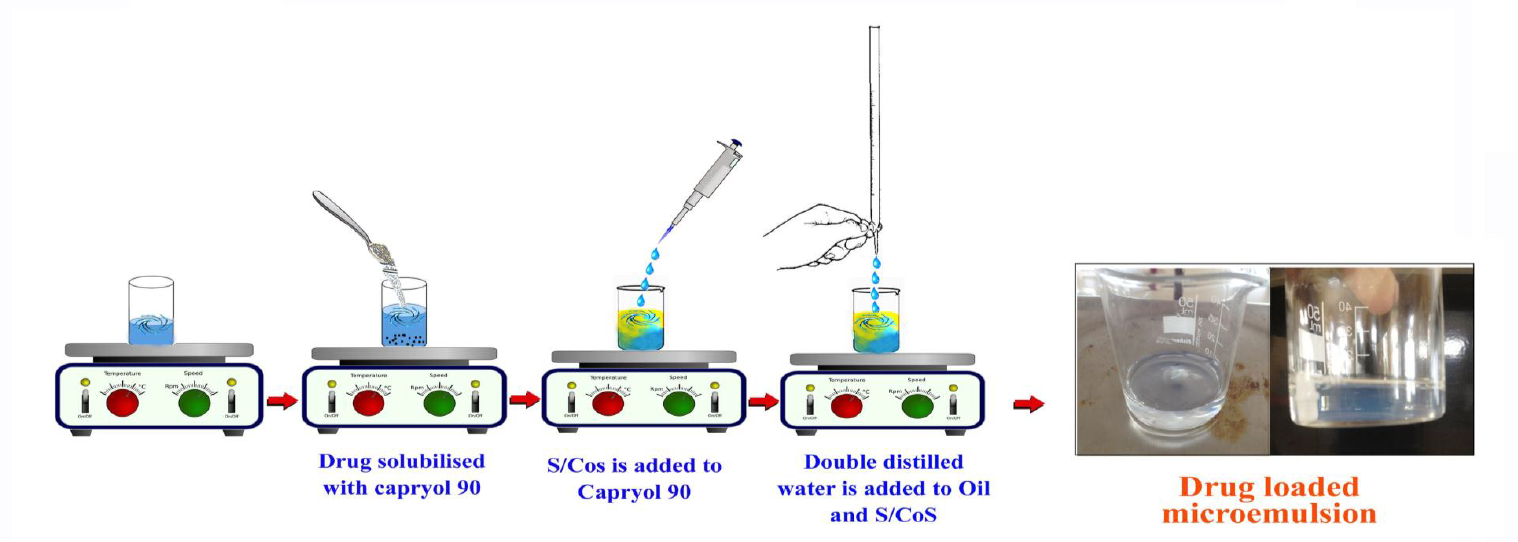
Based on preliminary solubility studies, a combination of Capryol 90 (oil phase), Labrasol (surfactant), and Transcutol P (co-surfactant) was selected for microemulsion formulation. The surfactant to co-surfactant weight ratio (Smix) was fixed at 1:1 for this set of formulations.
Characterization of Microemulsions
1.Percent Drug Content
One milliliter of microemulsion was lysed with methanol, diluted with pH 7.4 phosphate buffer, and analysed spectrophotometrically at 272 nm to determine drug content.
2.PH Measurement
The pH of microemulsions was measured using a calibrated Elico pH meter standardized with standard buffers.
3.Particle Size, Polydispersity Index (PDI), and Zeta Potential (ζ)
Particle size and distribution are crucial parameters for microemulsions, typically ranging between 100–200 nm. Smaller globule sizes increase the total surface area, enhancing drug encapsulation. Particle size was determined using photon correlation spectroscopy (Malvern Nano ZS90) at 25°C and 90° scattering angle. Zeta potential was measured to assess surface charge and predict stability. The Helmholtz–Smoluchowski equation was used to calculate zeta potential from electrophoretic mobility. Surface charge influences clearance and tissue disposition after transdermal delivery. Typically, at physiological pH, fat emulsions exhibit a negative zeta potential between -30 mV and -60 mV, indicating colloidal stability. Microemulsions were diluted 1:100 with distilled water before measurement.
4.Centrifugation Stress Testing
To evaluate physical stability, microemulsions were centrifuged at 5000 and 10000 rpm for 30 minutes. Samples were observed for phase separation, inversion, aggregation, creaming, or cracking.
5.Transmission Electron Microscopy (TEM)
Microemulsion morphology was studied by TEM (FEI, TECNAI T20, USA). Samples were diluted (1 mL microemulsion in 9 mL distilled water), stained with 2% phosphotungstic acid, placed on copper grids, dried at 25°C, and examined under TEM. Dilutions were prepared at 60°C to investigate percolation.
6.In Vitro Drug Release Studies Using Franz Diffusion Cell
In vitro release was conducted using vertical Franz diffusion cells (4.52 cm² diffusion area, 28 mL capacity). Microemulsions containing 5 mg of drug were placed in donor compartments; receptor compartments contained pH 7.4 phosphate buffer, maintained at 32 ± 0.5°C with stirring at 100 rpm. A cellulose dialysis membrane (cut-off 14 kDa), soaked overnight in receptor medium, separated donor and receptor compartments. At predetermined intervals (1, 2, 4, 6, 8, 10, 12, 24 hours), 1 mL samples were withdrawn and replaced with fresh buffer to maintain sink conditions. Drug content was analysedspectrophotometrically at 272 nm. Transdermal flux (J) was calculated from the slope of cumulative drug permeated per unit area vs. time. Permeability coefficient (Kp) was calculated using Equation 3.2:
7.Permeability Coefficient (Kp)
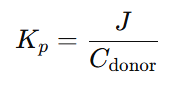
Where:
8.In Vitro Release Kinetics
Release data were fitted to Zero order ,First orderkinetic models to elucidate the drug release mechanism.
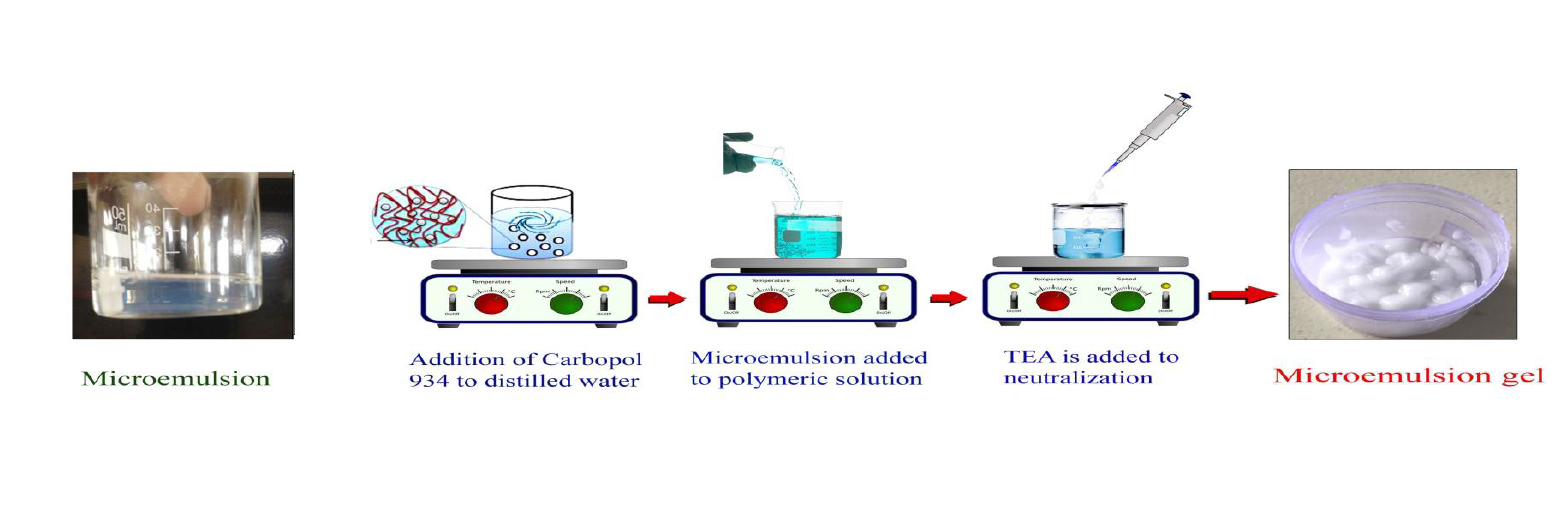
Preparation of Plain and Microemulsion-Based Gels
Although microemulsions offer excellent drug delivery properties, their low viscosity makes them difficult to apply and stabilize topically. To overcome this, Carbopol 934 NF was selected as a gelling agent due to its compatibility, ease of spreading, and favorable feel.
1.Plain Gel Preparation
Carbopol polymer dispersions (1%, 1.5%, and 2% w/v) were prepared in water with the addition of pure drug. The mixtures were stirred at room temperature until homogeneous and lump-free. Triethanolamine was added to neutralize and adjust pH to 7.0. The gels were hydrated for 24 hours at room temperature. A schematic is shown in Figure 3.7.
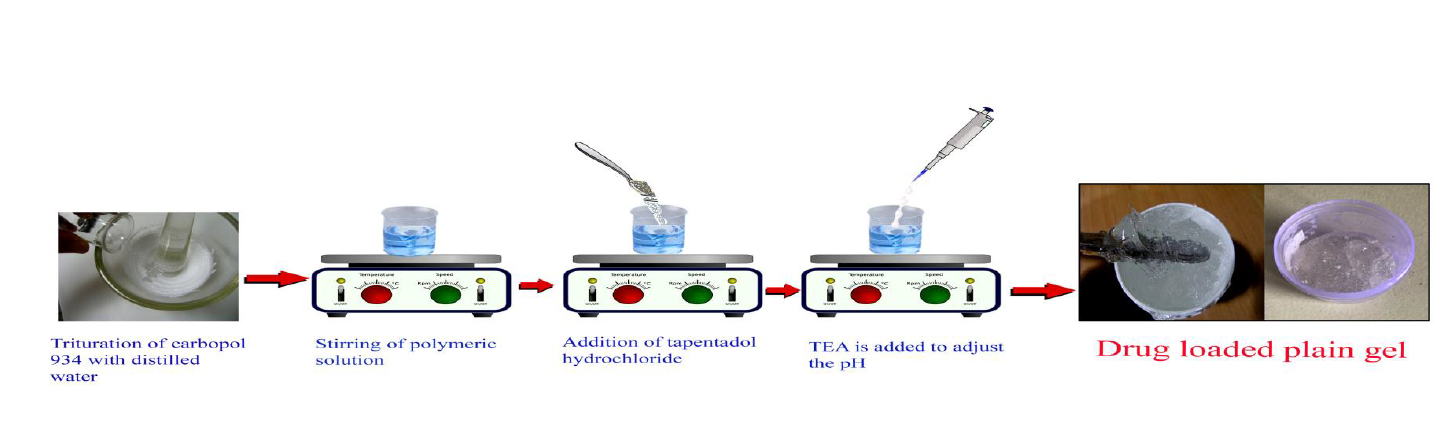
2.Microemulsion-Based Gel Preparation
For microemulsion gels, Carbopol polymer (1%, 1.5%, 2% w/v) was dissolved in freshly prepared drug-loaded microemulsion with continuous stirring. Triethanolamine was added to adjust pH between 6.0 and 7.0. The gels were subjected to physicochemical evaluation, stability studies, and diffusion/permeation testing.
Rheological Studies
The rheological behaviour of gels with various Carbopol concentrations was studied using a Brookfield cone and plate rheometer (LV-DV III) at 25 ± 0.1ºC. Samples (0.5 g) were tested at rotational speeds from 0.5 to 100 rpm using spindle #4. Viscosity (η), shear stress (τ), and shear rate were recorded to determine flow behaviour. Newtonian fluids show a linear relationship between shear stress and shear rate. Non-Newtonian (pseudoplastic) gels, which exhibit shear thinning, are preferred for topical application due to better drug release and Spreadability. Rheological properties were also assessed for six-month-old gels stored in HDPE bottles under ambient conditions to evaluate stability.
Characterization of Gels (Plain Gel and Microemulsion Gel)
The optimized microemulsion gel formulations were subjected to various physicochemical evaluations, including percent drug content, pH, particle size, zeta potential, spreadability, and comparative in vitro and ex vivo permeation studies.
1.Determination of Particle Size, PDI, and Zeta Potential (ζ)
Particle size, polydispersity index (PDI), and zeta potential (ζ) of the tested formulations (microemulsion gel and plain gel) were measured using a Malvern Nano ZS90 (Malvern, UK) as described previously in Section.
2.Determination of Percent Drug Content (%)
An accurately weighed amount of the gel formulation was lysed with methanol and diluted with pH 7.4 phosphate buffer. The drug concentration was then determined spectrophotometrically at 272 nm, following the method outlined in Section.
3.Determination of Spreadability
Spreadability of the gels was measured using a wooden block and glass slide apparatus, as developed by Multimer et al. One gram of gel was placed on a fixed glass slide, and a second glass slide was placed over it, weighted with 100 g for 5 minutes. After removing the weight, the time taken for the upper slide to slip off the gel was recorded in seconds.
4.Spreadability (S) was calculated using the formula:
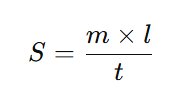
The primary aim of this study was to design, prepare, and optimize a transdermal gel formulation containing TPHCl loaded microemulsions using Carbopol 934 NF as the gelling agent. The microemulsions were formulated by carefully selecting appropriate excipients. Their compositions were determined through pseudo-ternary phase diagrams constructed using aqueous titration at room temperature. Two Smix ratios, 2:1 and 3:1, were chosen based on the microemulsion regions observed with different surfactant and co-surfactant mixtures.
Physicochemical Characterization of Microemulsions
1.Drug Content Analysis
The drug loading efficiency of the microemulsions ranged from 97.1 ± 0.8% to 99.5 ± 0.8% for the 2:1 Smix formulations and from 97.3 ± 0.8% to 99.1 ± 0.9% for the 3:1 Smix ratios. These values demonstrate a consistent and uniform distribution of TPHCl within the nanoglobules, indicating minimal drug loss during preparation and storage.
2.Particle Size and Distribution
The mean droplet size for microemulsions with a 2:1 Smix ratio was between 148.2 ± 0.4 nm and 354.1 ± 1.1 nm, with polydispersity index (PDI) values ranging from 0.187 ± 0.1 to 0.464 ± 0.7. Similarly, the 3:1 Smix formulations exhibited droplet sizes from 112.6 ± 1.5 nm to 361.6 ± 1.1 nm and PDI values from 0.132 ± 0.5 to 0.612 ± 0.4. The reduction in globule size correlated with increased surfactant and co-surfactant concentration, likely due to enhanced solubilization and decreased interfacial tension. Lower PDI values in both formulations suggest a homogenous size distribution, with the 3:1 ratio demonstrating slightly better uniformity.
3.Zeta Potential
The zeta potential values for the 2:1 Smixmicroemulsions ranged from -4.6 ± 1.2 mV to -19.1 ± 0.9 mV, while the 3:1 ratio formulations showed values between -9.1 ± 1.4 mV and -28.4 ± 0.7 mV. Smaller droplets exhibited higher magnitude zeta potentials, reflecting improved electrostatic stabilization and physical stability, as particles with higher zeta potential resist aggregation. The negative charge was attributed to the presence of surfactants, which reduce surface tension and stabilize micelles. Overall, the 3:1 Smix ratio formulations showed greater stability potential.
4.PH Evaluation
The pH values for all microemulsion samples remained close to 7.0 ± 0.1, consistent with the pH of distilled water used in their preparation. This neutral pH range is compatible with skin and suggests a low risk of irritation upon topical application.
5.Transmission Electron Microscopy
TEM images confirmed that the microemulsion droplets were predominantly spherical or slightly elliptical, with uniform size distribution. The absence of droplet coalescence after significant dilution indicated excellent thermodynamic and physical stability. The particle sizes observed by TEM were consistent with the dynamic light scattering results, typically around 100 nm.
Figure 2: Transmission Electron Microscopy (100 nm)
6.In Vitro Drug Release from Microemulsions
Drug release studies are crucial for evaluating the performance of the drug delivery system and ensuring batch consistency. The cumulative drug release profiles of all microemulsion formulations were recorded up to 24 hours. The 3:1 Smix ratio microemulsions exhibited a gradual and sustained release pattern, with drug release increasing steadily for the first 12 hours before plateauing. Percent cumulative drug release ranged from 46.2 ± 0.5% to 92.6 ± 0.2% for the 2:1 Smix ratio formulations, while the 3:1 ratio formulations released between 52.1 ± 0.2% and 98.42 ± 0.1% over the same period. The ME3 formulation (2:1 Smix) and ME5 formulation (3:1 Smix) demonstrated the highest drug release, indicating efficient drug delivery from these systems.
Figure 3: In Vitro Drug Release from Microemulsions
7.Influence of Composition on Microemulsion Properties
It was noted from the data that the droplet size decreased from ME1 to ME3 but increased from ME4 to ME9 within the 2:1 Smix formulations. This trend was explained by the relative concentration of surfactant/co-surfactant and oil phase: increased Smix from ME1 to ME3 lowered the interfacial tension, thus reducing droplet size. In contrast, increasing oil concentration and decreasing Smix from ME4 onwards led to larger droplets due to increased interfacial tension. A similar pattern was observed for the 3:1 Smix formulations, where the balance between surfactant mixture and oil content dictated globule size and stability.
8.Stability Considerations
Microemulsions with smaller droplet sizes showed higher zeta potential values, indicating enhanced physical stability and reduced tendency to aggregate. This improved stability is beneficial for maintaining formulation consistency during storage and application. The negative surface charge contributed to preventing flocculation and droplet coalescence, supporting the overall robustness of the system.
Microemulsion-Based Gel Evaluation Parameter
These gels retained suitable pH and displayed consistent rheological behaviour, making them suitable for transdermal application. In vitro and ex vivo studies of the gels showed sustained drug release and enhanced permeation compared to control formulations, demonstrating the potential of microemulsion-based gels as effective carriers for TPHCl delivery through the skin.
1.In vitro Drug Release Kinetics
Model-dependent methods were applied to mathematically describe the drug release profile as a function of parameters related to the pharmaceutical formulation. These methods assist in determining the release kinetics and mechanisms of drug delivery systems. Several factors affect drug release, including drug type and dose, excipient types and amounts, preparation methods, environmental conditions during release, as well as the geometry and dimensions of the dosage form.
2.In Vitro Drug Release Studies Using Franz Diffusion Cell
In vitro drug release was performed as described in Section 3.5.6. A cellulose dialysis membrane ,pre-soaked overnight in receptor medium, served as the barrier. Formulations equivalent to 5 mg drug content were placed in the donor compartment of vertical Franz diffusion cells with an effective diffusion area of 4.52 cm². Phosphate buffer pH 7.4 (28 mL) was used in the receptor compartment, maintained at 32 ± 0.5°C and stirred at 100 rpm. Aliquots (1 mL) were withdrawn at specified intervals (1, 2, 4, 6, 8, 10, 12, and 24 hours), replaced immediately with fresh buffer, and analysed at 272 nm using UV spectroscopy. All experiments were performed in triplicate.
3.Drug and Excipient Interaction Studies by FTIR Analysis
Fourier-transform infrared spectroscopy (FTIR) was conducted using a Bruker FTIR spectrophotometer with OPUS software. Samples included pure drug (tapentadol), excipients (labrasol, capryol 90, transcutol P), their physical mixtures with the drug, and microemulsion formulations. Spectra were recorded over the range 400–4000 cm?¹ using ATR mode.
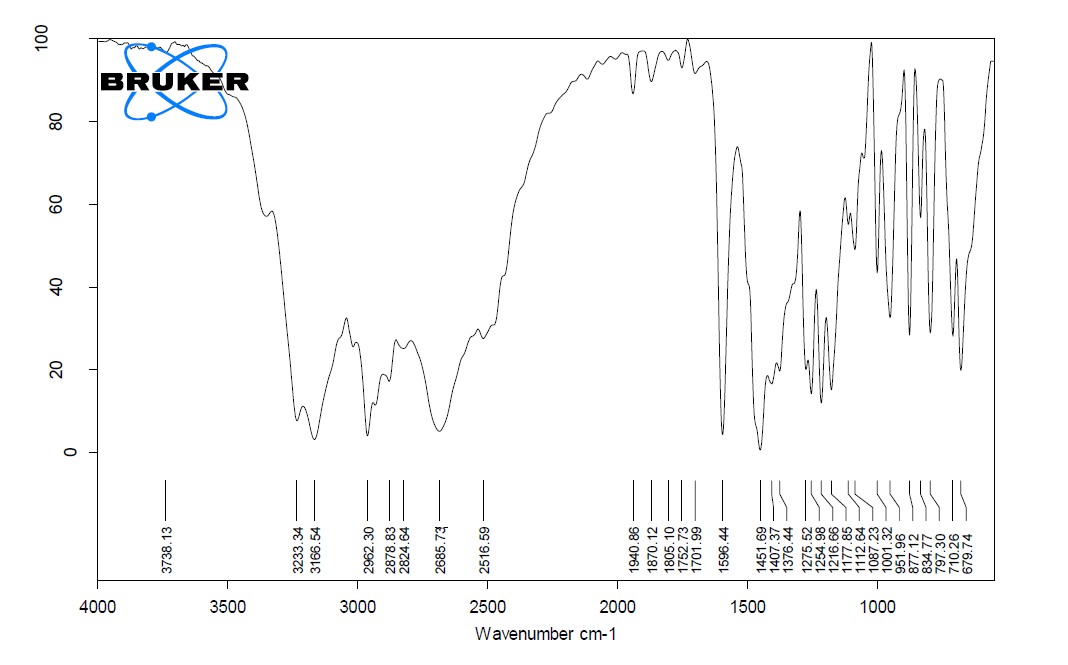
Figure 4: FTIR of Pure Drug
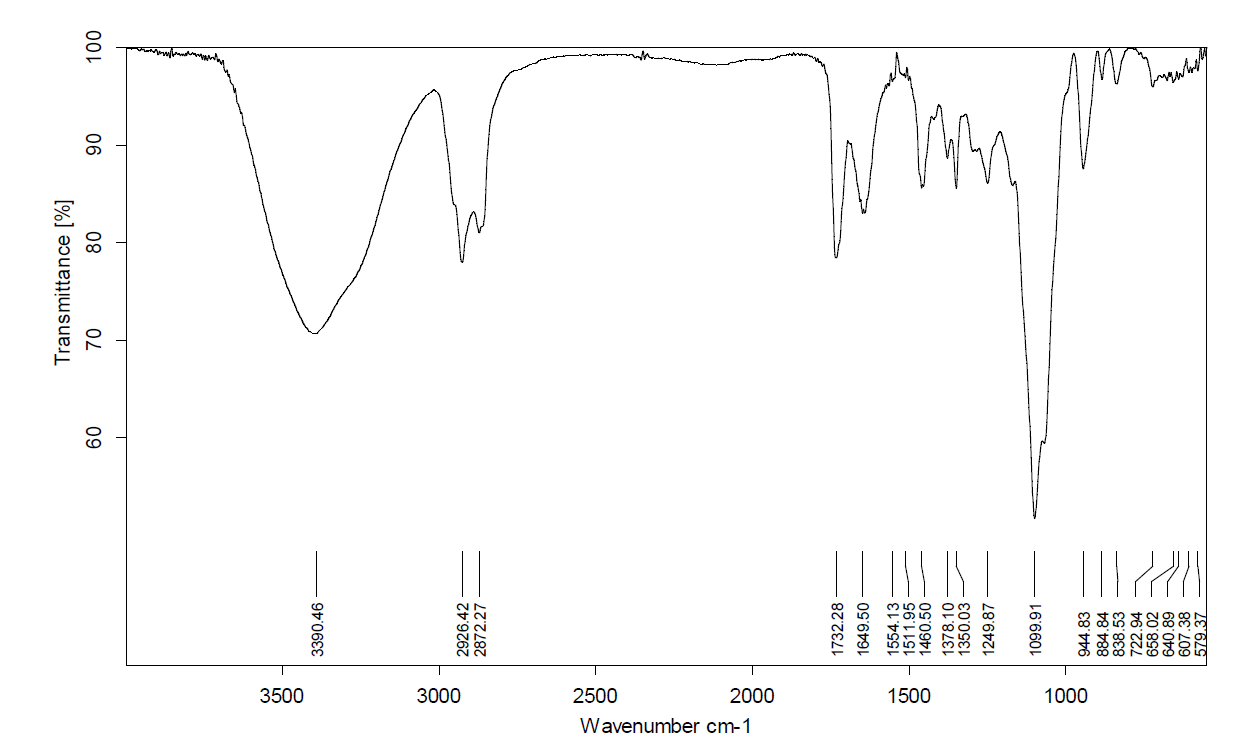
Figure 5: FTIR of Formulation
4.Drug and Excipient Interaction Studies by DSC Analysis:
Differential scanning calorimetry (DSC) was performed using a Mettler Toledo STARe (Model DSC 822e). Samples included the physical mixture, TPHCl-loaded microemulsion, and pure drug (control). Samples were heated from 25°C to 250°C at 10°C/min under nitrogen purge (20 mL/min). An empty aluminium pan was used as reference.
Figure 6: DSC Graph of Pure Drug
Figure 7: DSC Graph of Formulation
5.Stability Studies of Selected Formulations
Stability studies were conducted on formulations microemulsion gel, stored in amber containers at 4°C and 25 ± 2°C / 75 ± 5% RH for 3 months. Samples were withdrawn at 1-, 2-, and 3-months intervals and evaluated for parameters including percent drug content, globule size, and zeta potential. Stability studies conducted on the optimized microemulsion and microemulsion gel demonstrated that both formulations maintained acceptable drug content, particle size, and zeta potential over a period of three months under refrigerated conditions (4 ± 2°C). However, at accelerated stability conditions (40 ± 2°C/75 ± 5% RH), a gradual decrease in drug content and increase in particle size was observed, indicating some degree of instability at elevated temperature and humidity. In comparison with previous studies the present microemulsion and microemulsion gel formulations of TPHCl show consistent trends in enhanced permeation and sustained release profiles similar to other microemulsion systems reported for topical and transdermal drug delivery. The findings confirm that microemulsion gel systems can effectively enhance drug permeation and provide sustained release, making them promising carriers for topical delivery of lipophilic and poorly soluble drugs.
Figure 8: Comparative assay f Microemulsion gel with Plan gel
6.Appearance: A visually appealing formulation is crucial for patient acceptance. The microemulgel should be clear, homogenous, and free from phase separation or particulate matter. Visual inspection under adequate lighting conditions is typically employed for this assessment.
7.PH Measurement: Maintaining a pH compatible with skin (approximately 5.5–6.5) is essential to prevent irritation. The pH of the microemulgel is measured using a calibrated digital pH meter. Adjustments are made using TEA to achieve the desired Ph.
8.Spreadability: This parameter assesses the ease of application of the gel on the skin. It is evaluated by measuring the diameter of the gel spread between two glass plates under a specified weight. Optimal spreadability ensures uniform application and enhances patient compliance.
9.Viscosity: Viscosity influences the gel's stability, drug release rate, and ease of application. It is measured using a rotational viscometer at controlled temperatures. The viscosity should be sufficient to maintain the gel's integrity while allowing easy application.Viscosity was optimized for easy application and retention on the skin
10.Drug Content Uniformity: Ensuring uniform distribution of the active drug within the gel matrix is critical for consistent therapeutic effects. Drug content is quantified by dissolving a known quantity of the gel in a suitable solvent, followed by spectrophotometric analysis. The content should be within ±5% of the labelled claim.
11.Stability Studies: Stability testing under various environmental conditions (e.g., temperature, humidity) is conducted to assess the formulation's shelf life. Parameters such as appearance, pH, viscosity, and drug content are monitored over time to ensure product integrity.
12.In Vitro Drug Release and Permeation Studies: These studies evaluate the rate and extent of drug release from the microemulgel and its permeation through biological membranes. Franz diffusion cells are commonly used for such assessments, providing data critical for predicting in vivo performance.
13.Zeta Potential: Zeta potential indicates the surface charge and stability of the microemulsion. Values >±30 mV suggest good stability.
14.PH: The gel's pH was adjusted to 5.5–6.5, ideal for skin compatibility.
CONCLUSION
Novel drug delivery systems (NDDS) are designed to deliver drugs at a controlled rate, precisely targeting the required bio-site and sustaining drug action for prolonged periods, overcoming limitations of conventional dosage forms such as low bioavailability, first-pass metabolism, and lack of site specificity. Among NDDS, microemulsion-based gels have emerged as promising carriers for transdermal delivery due to their improved drug solubilization, controlled release, enhanced stability, and biocompatibility. The present study demonstrated the successful formulation of a microemulsion and its incorporation into a gel using pharmaceutically acceptable components—propylene glycol monocaprylate as oil phase, Labrasol as surfactant, and Transcutol P as co-surfactant. The optimized microemulsions exhibited nanosized globules with low polydispersity index and negative zeta potential, indicating good stability and uniformity. In vitro skin permeation studies revealed a sustained and controlled release of the drug (TPHCl) over 24 hours, with microemulsion gels showing significantly higher permeation and skin retention compared to plain gels. Fourier-transform infrared spectroscopy and differential scanning calorimetry confirmed the absence of chemical interactions between the drug and formulation components, ensuring compatibility and stability. Transmission electron microscopy further confirmed the spherical and uniform nature of microemulsion droplets. The microemulsion gel demonstrated pseudoplastic rheological behaviour, suitable viscosity, and excellent Spreadability, making it a patient-friendly topical formulation. Stability studies indicated that the microemulsion gels were physically and chemically stable under refrigerated conditions, though temperature played a significant role in their overall stability. Overall, the developed microemulsion-based gel system proved to be an effective transdermal delivery vehicle, capable of enhancing drug permeation, improving bioavailability, and providing sustained drug release. This approach holds significant potential for improving therapeutic outcomes and patient compliance in the transdermal administration of drugs with poor oral bioavailability and significant gastrointestinal side effects.
ACKNOWLEDGMENTS
The authors express their sincere gratitude to the institutional research facilities and laboratories that supported this study. Special thanks to LCIT School of Pharmacy, Bilaspur, Chhattisgarh, India for providing access to advanced analytical instrumentation and technical assistance throughout the project. We also acknowledge the valuable guidance and encouragement from our mentors and colleagues. Finally, we appreciate our Supervisor Dr. Deepesh Lall and, Dr. Ritesh Jain Professor/Principal who made this research possible.
REFERENCES





Syed Saif Ullah*, Dr. Deepesh Lall, Dr. Ritesh Jain, Anil Kumar Manhar, Vivek Kumar Sinha, Formulation and Evaluation of Microemulsion Gel: Role of Polymers and Permeation Enhancers in Enhancing Transdermal Drug Delivery System, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 6, 3417-3433. https://doi.org/10.5281/zenodo.15718910
 10.5281/zenodo.15718910
10.5281/zenodo.15718910