
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Raghu College of Pharmacy, Visakhapatnam.
The oral route of administration is currently the gold standard in pharmaceuticals, valued for its safety, patient compliance, and economic advantages, which contribute to its high patient compliance. MDTs' format enables them to disintegrate or dissolve rapidly in the mouth, through the incorporation of super disintegrants, such as croscarmellose sodium, sodium starch glycolate, and crospovidone, which act through mechanisms including swelling, wicking (capillary action), and deformation recovery, eliminating the need for water and making it easier to take medications. Such tablets disintegrate or dissolve in saliva in less than 60 seconds. The formulation's popularity and utility led to the creation of multiple MDT technologies. MDTs are a valuable alternative for patients with dysphagia, offering improved reliability and ease of use for children, the elderly, and mentally ill patients. The Pharmaceutical market has seen a rise in fast-dissolving products, with many new launches in the past 2 to 3 years. This growth is also reflected in the increasing use of fast-dissolving technology for new drug development. This article provides an overview of key attributes, benefits, limitations, formulation considerations, technological approaches, assessment methods, and prospects.
The oral route is the most effective route, despite all the other advanced drug delivery systems. This is due to enhanced patient compliance and convenience for the administrator. Tablets are a frequently prescribed oral dose type that have properties like their firmness, convenience of production, and accessibility. Along with these advantages, tablets have some major disadvantages, such as difficulty in swallowing for geriatric and pediatric patients and conditions like dysphagia. Hence, mouth-dissolving tablets were developed.
Mouth dissolving tablets are defined as “The solid dosage forms that rapidly disintegrate and dissolve into saliva in the oral cavity, resulting in solution without the need for water for administration”. The technology involved in mouth-dissolving tablets makes the tablets disintegrate in the mouth without chewing, and additional water intake has drawn a greater deal of attention. Mouth dissolving tablets are also known as: fast melting tablets, rapid dissolve tablets, rapid melt tablets, fast dispersing tablets, freeze-dried wafers, and quick disintegrating tablets. These were approved by the Food and Drug Administration and classified as orally disintegrating tablets. The term Oro-dispersible tablet for MDT was recognized by the European pharmacopoeia, which means that, tablet dissolves and disintegrates in less than 3 minutes in the mouth before swallowing. A tablet disintegrates into smaller granules and melts in the mouth, resulting in a hard, solid, to gel-like structure. This allows easy swallowing by the patients. The disintegration time for the mouth-dissolving tablets varies from about seconds to minutes. Mouh dissolving tablets have gained popularity due to their Self-administration capabilities, absence of need for water, and rapid disintegration and dissolution.
ADVANTAGES1,2
DISADVANTAGES1,2:
FORMULATION ACCEPTS OF MDTs3,4:
Ingredients used in the formulation of MDTs must allow quick release of the drug, resulting in faster dissolution. This includes both active pharmaceutical ingredient (drug) and excipient (additives).
There are no particular limitations for the active pharmaceutical ingredient. Researchers have formulated mouth dissolving films with different drug categories such as analgesics, anti-allergics, anxiolytics, anti-bacterial agents, cardiovascular agents, neuroleptics, etc.
The characteristics that may render unsuitable of drug delivery as MDTs are:
Superdisintegrants involve four different mechanisms by which a tablet is broken down into smaller particles. The mechanisms involved are:
Examples of superdisintegrants include carboxymethyl cellulose, sodium carboxymethyl cellulose, sodium starch glycolate, crospovidone, microcrystalline cellulose, and modified corn starch.
PRE FORMULATION STUDIES5,6:
Db =MVb
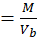
Where M is the mass of powder
Vb is the bulk volume of the powder.
It is expressed in g/ml.
Dt = MVt
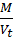
Where M is the mass of powder
Vt is the tapped volume of the powder.
The powder was allowed to flow through the funnel. The angle of repose was calculated by measuring the height and radius of the pile of powder.
tan (θ) = hr

θ = tan-1 hr
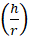
Where θ is the angle of repose.
h is the height in cms
r is the radius in cms.
Table 1: Relationship between angle of repose and powder flow
|
Sr No |
Angle of Repose |
Type of Flow |
|
1. |
<20 |
Excellent |
|
2. |
20 - 30 |
Good |
|
3. |
30-40 |
Passable |
|
4. |
>34 |
Very poor |
I =Dt-DbDt
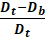
Where, Dt is the tapped density of the powder
Db is the bulk density of the powder
Table 2: Relationship between % compressibility and flow ability
|
% Compressibility |
Flow ability |
|
5 – 12 |
Excellent |
|
12 – 16 |
Good |
|
18 – 21 |
Fair Passable |
|
23 – 35 |
Poor |
|
33 – 3 |
Very poor |
|
< 40 |
Very very poor |
Hausner ratio = DtDb
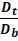
Where, Dt is the tapped density.
Db is the bulk density.
A lower Hausner ratio (<1.25) indicates better flow properties than a higher one (>1.25).
V= Vb – Vp
Where, Vb = Bulk volume (volume before tapping)
Vp = The volume (volume after tapping)
Porosity = Vb-VpVp
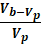
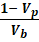
Porosity is frequently expressed in percentage. It is given by:
% porosity = ( 1- VpVb
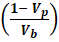
EVALUATION TESTS FOR MOUTH DISSOLVING TABLETS7,8:
Table 3: Uniformity of weight according to USP Specification
|
Sr. No |
Average weight of tablets (mg) |
Maximum % difference allowed |
|
1. |
130 or less |
10 |
|
2. |
130 – 324 |
7.5 |
|
3. |
More than 324 |
5 |
% Friability = W1- W2W1
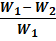
Where, W1 = weight of tablet before test
W2= weight of tablet after test
In – vivo disintegration test: In – vivo disintegration test is carried out on 2 or 3 tablets in the mouth, and the time taken to disintegrate the complete tablet is noted.
In – vitro disintegration test: In – vitro disintegration test is measured by dropping a tablet in the beaker containing 50 ml of Sorenson’s buffer pH 6.8. Three tablets from each formulation are randomly selected, and in vitro dispersion is carried out.
R= 100 X Wa-WbWb
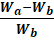
Where,
Wb is the weight of the tablet before water absorption
Wa is the weight of the tablet after water absorption
TECHNOLOGIES USED FOR THE FORMULATION OF MOUTH-DISSOLVING TABLETS9:
There are several technologies used in the formulation of mouth dissolving tablets, namely (1) Conventional technologies. (2) Patented technologies.
Table 4: Different Conventional and Patented technologies
|
Conventional Technologies |
Patented Techcnologies |
|
Freeze Drying |
Zydis technologies |
|
Tablet Molding |
Orasolv technologies |
|
Direct Compression |
Durasolv technologies |
|
Sublimation |
Wowtab technologies |
Conventional technologies for mouth-dissolving tablets:
There are many conventional technologies for the formulation of MDTs. They are:
Patented technologies for mouth-dissolving tablets:
PATENTS ON MOUTH-DISSOLVING TABLETS10:
There is an account of some recent patents in the field of mouth-dissolving tablets, as shown in Table 5.
Table 5: Patents on mouth-dissolving tablets
|
Patented Technology |
Process Involved |
Patent Owner |
Patent Number |
Drugs (Brand Names) |
Drug Release |
Advantages |
Disadvantages |
|
Zydis |
Lyophilization |
R.P. Scherer Inc. |
U.S. Pat. Nos. 4,305,502, 4,371,516, 4,642,903 and 5,738,875 |
Loratidine (Claritin Reditab and Dimetapp Quick Dissolve) |
Dissolves in 2- 10 sec |
Quick dissolution, Self-preserving, and increased bioavailability |
Expensive process, poor stability at higher temperatures and humidity |
|
Orasolv |
Compressed Tablets |
Cima Labs Inc. |
U.S.?Patent No.?5,178,878 |
Paracetamol (Tempra Quicklets), Zolmitriptan (Zolmig Repimelt) |
Disintegrate in 5-45 sec |
Taste-masking is twofold: quick dissolution |
Low mechanical strength |
|
Flashdose |
Cotton-candy process |
Fuisz Technology Ltd. |
U.S. Pat. Nos. 5,587,172, 5,616,344, and 5,622,719 |
Tramadol HCl (Relivia Flash dose) |
Dissolves within 1 min |
High surface area for dissolution |
High temperature required to melt the matrix can limit the use of heat-sensitive drugs, sensitive to moisture and humidity. |
|
Wowtab |
Compressed Moulded Tablets |
Yamanouchi Pharma Technologies, Inc. |
US Patent 5,466,464 and US Patent 5,576,014 |
Famotidine (Gaster D) |
Disintegrate in 5-45 sec |
Adequate dissolution rate and hardness |
No significant change in bioavailability |
|
Advatab |
Microcaps and diffuscap CR Technology |
Eurand International |
U.S. Pat. No. 6,139,865 |
AdvaTab cetrizine, AdvaTab Paracetamol |
Disintegrates in less than 30 seconds |
Rapid Disintegration Without Water and Taste Masking Capability |
Limited API Compatibility, Moisture Sensitivity |
|
Durasolv |
Molding |
Cima Labs Inc. |
US 6,024,981 |
Hyoscyamine Sulphate (NuLev) |
Disintegrate in 5-45 sec |
Higher mechanical strength than Orasolv, Good rigidity |
Inappropriate with a larger dose |
|
Ziplets |
Molding |
Eurand |
WO?99/44580 |
Ibuprofen (Cibalgina Due Fast) |
Disintegrate in 20 seconds in the mouth |
Good mechanical strength, satisfactory properties can be obtained at high dose (450 mg) and high weight (850 mg) |
As the soluble component dissolves, the rate of water diffusion into tablet is decreased because of the formation of a viscous concentrated solution |
|
Lyoc |
Multiparticulate Compressed Tablets |
Farmlyoc |
US 4,616,047 and US 5,843,347 |
Phloroglucinol Hydrate (Spasfon Lyoc) |
Disintegrates in less than 10 seconds |
Fast disintegration, patient compliance |
Low drug load, expensive manufacturing |
MARKETED PRODUCTS OF MOUTH DISSOLVING TABLETS
Some of the marketed products of mouth dissolving tablets are illustrated in Table 6.
Table 6: Marketed products of mouth-dissolving tablets
|
Drugs |
Product |
Super- disintegrants |
Manufactured By |
Uses |
Images |
|
Olanzapine |
Zyprexa Zydis |
Crosspovidone |
Eli Lilly and Company |
It is an atypical antipsychotic primarily used to treat schizophrenia and bipolar disorder. |
|
|
Ondansetron |
Zofran ODT |
Crosscaramellose |
Glaxo Smith Kilne (GSK) |
It is used to prevent nausea and vomiting caused by cancer medicines(chemotherapy) or radiation therapy. |

|
|
Alprazolam |
Niravam |
Crospovidone |
Schwarz Pharma |
It is used as a CNS Depressant and is used to treat anxiety and panic disorders |

|
|
Donepezil |
Aricept ODT |
Crosspovidone |
Eisai Inc. |
It is used to treat dementia and Alzheimer’s disease. |
|
|
Rizatriptan benzoate |
Maxalt MLT |
Crospovidone, Carboxymethylcellulose calcium,
|
Merck and Co. |
It is used in the acute treatment of migraine with or without aura. |
|
|
Aripiprazole |
Abilify Discmelt |
Croscarmellose sodium, Crospovidone |
Otsuka America/ Bristol-Myers Squibb |
It is used to treat schizophrenia, bipolar disorder, depression, and Tourette’s disorder. |
|
|
Lansoprazole |
Prevacid SoluTab |
Crospovidone, Microcrystalline cellulose, Hydroxypropyl cellulose |
Takeda Pharmaceuticals |
It is used to treat duodenal and gastric ulcers, erosive esophagitis, and gastroesophageal reflux disease |

|
|
Metoclopramide hydrochloride |
Reglan ODT |
Crospovidone, microcrystalline cellulose |
Oaknet Healthcare Pvt Ltd |
It is used to treat nausea, vomiting, indigestion, and heartburn. |

|
|
Carbidopa levodopa |
Parcopa |
Crospovidone, Microcrystalline cellulose |
Schwarz Pharma |
It is used to treat symptoms of Parkinson's disease. |

|
|
Risperidone |
Risperdal M-Tab |
Microcrystalline cellulose, Hypromellose, |
Janssen |
It is used to treat schizophrenia, bipolar disorder, and irritability. |
|
CONCLUSION11,12:
The MDTs have potential advantages over conventional oral dosage forms as they have improved patient compliance, rapid onset of action, and bioavailability. Various drugs that have limited bioavailability, high permeability, or that degrade rapidly in the stomach can be successfully formulated in the form of MDTs, as these tablets are absorbed through the oral cavity. MDTs are developed for most of the drugs, like anti-coagulants, anti-gout, anti-thyroid, anti-neoplastic agents, etc. Various manufacturing and evaluation techniques for MDTs are available due to advancements in technology. The research process is still going on for some categories of drugs that are not available in such dosage forms, i.e., MDTs.
REFERENCES





Kalyani Kondapalli, Yelumarthi Renuka, M. Yamini, Nalli Likhitha, Sanapala Usharani, Mouth Dissolving Tablets: A Comprehensive Review on Technologies, Research, Patents, and Marketed Products, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 9, 2768-2779. https://doi.org/10.5281/zenodo.17189781
 10.5281/zenodo.17189781
10.5281/zenodo.17189781